-
Články
Top novinky
Reklama- Vzdělávání
- Časopisy
Top články
Nové číslo
- Témata
Top novinky
Reklama- Videa
- Podcasty
Nové podcasty
Reklama- Kariéra
Doporučené pozice
Reklama- Praxe
Top novinky
ReklamaSusceptibility Loci Associated with Specific and Shared Subtypes of Lymphoid Malignancies
The genetics of lymphoma susceptibility reflect the marked heterogeneity of diseases that comprise this broad phenotype. However, multiple subtypes of lymphoma are observed in some families, suggesting shared pathways of genetic predisposition to these pathologically distinct entities. Using a two-stage GWAS, we tested 530,583 SNPs in 944 cases of lymphoma, including 282 familial cases, and 4,044 public shared controls, followed by genotyping of 50 SNPs in 1,245 cases and 2,596 controls. A novel region on 11q12.1 showed association with combined lymphoma (LYM) subtypes. SNPs in this region included rs12289961 near LPXN, (PLYM = 3.89×10−8, OR = 1.29) and rs948562 (PLYM = 5.85×10−7, OR = 1.29). A SNP in a novel non-HLA region on 6p23 (rs707824, PNHL = 5.72×10−7) was suggestive of an association conferring susceptibility to lymphoma. Four SNPs, all in a previously reported HLA region, 6p21.32, showed genome-wide significant associations with follicular lymphoma. The most significant association with follicular lymphoma was for rs4530903 (PFL = 2.69×10−12, OR = 1.93). Three novel SNPs near the HLA locus, rs9268853, rs2647046, and rs2621416, demonstrated additional variation contributing toward genetic susceptibility to FL associated with this region. Genes implicated by GWAS were also found to be cis-eQTLs in lymphoblastoid cell lines; candidate genes in these regions have been implicated in hematopoiesis and immune function. These results, showing novel susceptibility regions and allelic heterogeneity, point to the existence of pathways of susceptibility to both shared as well as specific subtypes of lymphoid malignancy.
Published in the journal: . PLoS Genet 9(1): e32767. doi:10.1371/journal.pgen.1003220
Category: Research Article
doi: https://doi.org/10.1371/journal.pgen.1003220Summary
The genetics of lymphoma susceptibility reflect the marked heterogeneity of diseases that comprise this broad phenotype. However, multiple subtypes of lymphoma are observed in some families, suggesting shared pathways of genetic predisposition to these pathologically distinct entities. Using a two-stage GWAS, we tested 530,583 SNPs in 944 cases of lymphoma, including 282 familial cases, and 4,044 public shared controls, followed by genotyping of 50 SNPs in 1,245 cases and 2,596 controls. A novel region on 11q12.1 showed association with combined lymphoma (LYM) subtypes. SNPs in this region included rs12289961 near LPXN, (PLYM = 3.89×10−8, OR = 1.29) and rs948562 (PLYM = 5.85×10−7, OR = 1.29). A SNP in a novel non-HLA region on 6p23 (rs707824, PNHL = 5.72×10−7) was suggestive of an association conferring susceptibility to lymphoma. Four SNPs, all in a previously reported HLA region, 6p21.32, showed genome-wide significant associations with follicular lymphoma. The most significant association with follicular lymphoma was for rs4530903 (PFL = 2.69×10−12, OR = 1.93). Three novel SNPs near the HLA locus, rs9268853, rs2647046, and rs2621416, demonstrated additional variation contributing toward genetic susceptibility to FL associated with this region. Genes implicated by GWAS were also found to be cis-eQTLs in lymphoblastoid cell lines; candidate genes in these regions have been implicated in hematopoiesis and immune function. These results, showing novel susceptibility regions and allelic heterogeneity, point to the existence of pathways of susceptibility to both shared as well as specific subtypes of lymphoid malignancy.
Introduction
Lymphoid malignancies represent clonal proliferations occurring at various stages of differentiation of B and T cells. B-cell differentiation is characterized by a canonical set of DNA modifications, including somatic hypermutation, class switching, and VDJ recombination. If aberrant, these result in lymphoid neoplasms ranging from less differentiated acute leukemia and lymphoma, to well-differentiated plasma cell malignancies [1]. Some genetic and environmental risk factors for lymphoma have been defined and antecedent autoimmune disorders increase risk for lymphoma several fold [2]. Familial clustering of lymphomas has been observed and may comprise mixed phenotypes of Hodgkin's lymphoma (HD) as well as the subsets of non-Hodgkin's (NHL) including follicular (FL), diffuse large B-cell (DLBCL), and chronic lymphocytic/small lymphocytic (CLL/SLL) [3]. While less common than B cell neoplasms, T cell malignancies are also part of the spectrum of familial lymphoma and may be seen alone or in combination with B cell neoplasms in kindreds with underlying immune deficiency or genomic instability [3].
The lack of genetic linkage to specific loci in such families has prompted the search for common susceptibility variants in the germline, which may provide evidence as to the etiology of these disorders. Genome wide association studies (GWAS) examining lymphoma susceptibility have focused on identifying risk loci associated with different subtypes of the disease, based on the a priori assumption that each of the subtypes have distinct biology and therefore, distinct pathogenesis. Thus far, a locus on 6p21.33, near PSOR1, and another region at 6p21.32, near HLA-DRB1 have been associated with FL [4], [5], [6] and Hodgkin's disease [7], [8]. A smaller study has described CDC42BPB at 14q32 to be associated with diffuse large cell lymphoma [9].
In order to test the paradigm that there are common and subtype specific germline susceptibility loci for lymphoma, we conducted a two-stage genome-wide association study (GWAS). Our stage-1 consisted of 944 cases of lymphoma, including 282 familial cases, and 4044 public shared controls. Stage-2 consisted of 1245 cases and 2596 controls. We have used a higher ratio of controls to cases to enhance power to detect association, as the use of public shared controls comes at no cost [10]. We also analyzed published data for overlap of the GWAS hits to expression quantitative trait loci (eQTL) in lymphoblastoid cell lines. Secondary analyses, such as gene set enrichment were carried out to detect enrichment of biologically relevant candidates for further study.
Results
The study design consisted of two phases, Stage-1 comprising the GWAS of lymphoma and shared controls and Stage-2 comprising 50 SNPS selected from the Stage-1 for replication.
Stage-1 results
In stage-1, we analyzed 944 cases of lymphoma, including 275 FL, and 4044 controls and documented strong evidence of association between SNPs on Chr6, with at least 9 SNPs showing PFL<1×10−7 at the HLA region (chr6 : 32.17–32.89 Mb) encompassing genes TNXB to HLA-DOB. The results of the stage-1 analysis for LYM, NHL, FL and DLBCL are shown as Manhattan plots (Figure 1) and quantile-quantile (QQ)-plots (Figure 2). FL showed the strongest enrichment of association signals; particularly on Chr6. We refrained from detailed analysis of smaller subsets, based on the power calculations performed using PGA [11] taking into account sample sizes, detectable relative risk and case to control ratios (Figure S1).Analysis of the major classifiers LYM and NHL and only the major subgroups FL, DLBCL were performed. In addition, a subset designated as NFD comprised any non-Hodgkin's lymphoma cases that were neither FL nor DLBCL. This subgroup was created to test if the associations in the larger LYM and NHL were driven primarily by the pre-dominant subgroups FL and DLBCL.
Fig. 1. Manhattan plots. 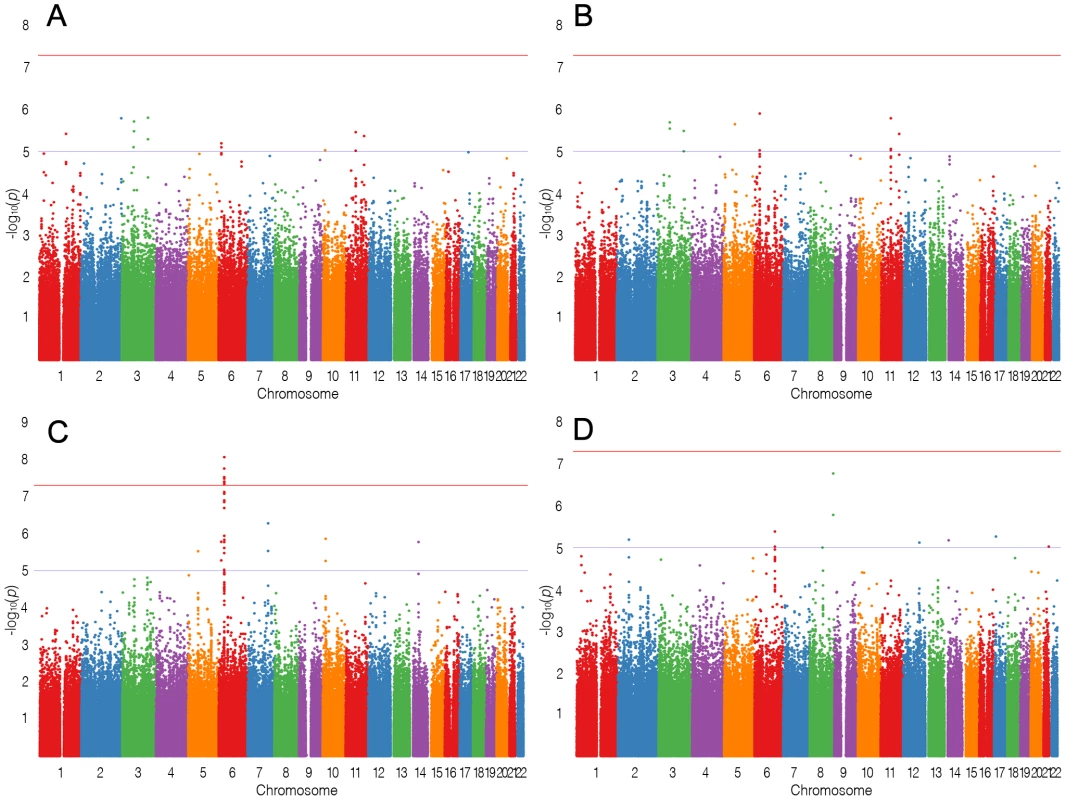
Manhattan plots for (A) LYM, (B) NHL, (C) FL, (D) DLBCL. The blue line shows suggestive association and the red line genome-wide association. X-axis labels correspond to chromosomes, Y-axis shows −log10(P) from logistic regression. Fig. 2. Quantile–quantile plots. 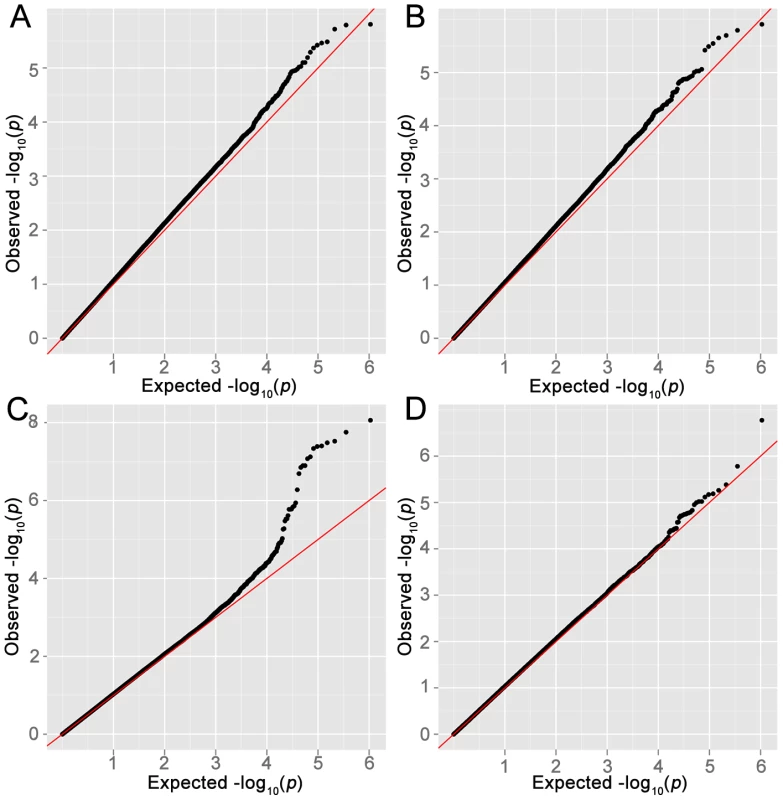
Quantile–quantile (QQ) plots for (A) LYM, (B) NHL, (C) FL, (D) DLBCL. The X-axis represents expected −log10(P) and the Y-axis represents −log10(P) from logistic regression. Genomic inflation factor λ for LYM, NHL, FL and DLBCL was 1.09, 1.07, 1.04 and 1.04 respectively. Among all analyses, the lowest p-values in the FL subset were observed on chromosome 6p. The smallest p-value was for rs2621416 (PFL = 8.69×10−9, OR 1.82) (Table S1) followed by rs9268853 (PFL = 1.76×10−8, OR = 1.74).Imputation of the stage-1 data revealed strong associations with FL for the 6p21.32 SNP rs12194148 (PFL = 1.18×10−16, 14.5 kb from rs9268853; r2 = 0.62, D′ = 1.0), suggesting a subtype specific association with the HLA locus (Figure 3C). In addition to the SNPs on chromosome 6p HLA region, we also found preliminary evidence of association of several SNPs at chromosome 3q25.2 with LYM, NHL and NFD. Another locus at 11q12.1 was defined by two SNPs with suggestive associations (P<10−5) (Table S1, Figure 3B).
Fig. 3. Regional plots from stage-1 GWAS. 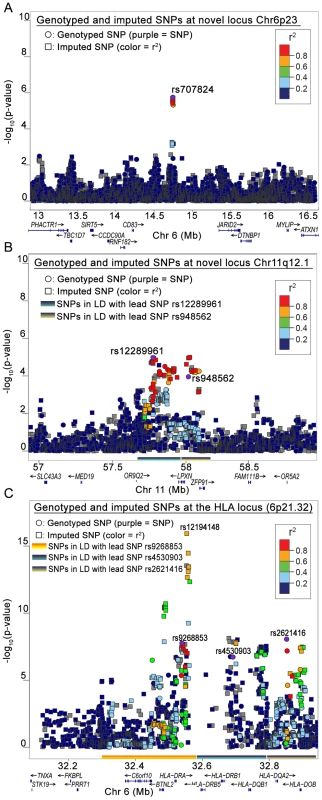
(A) Chr6p23, (B) Chr11q12.1 and (C) Chr 6p21.32. The regions corresponding to LD between lead SNPs and other SNPs are marked on the X axis of the plot. Stage-2 results
Fifty SNPs were selected from stage 1 for genotyping in a larger set of 1245 lymphomas (Table S1). After adjusting for age and Jewish ancestry, nine of 50 SNPs had P-values below the nominal alpha level of 0.05, while showing the same direction of effect as observed in stage 1 (Table S2). After adjusting for the 50 SNPs tested, rs4530903, at the HLA locus, remained significantly associated with NHL, FL, and DLCBCL. This SNP also appears to be associated with LYM, but the p-value was marginally higher than the Bonferroni corrected threshold. Two other tests were significant after multiple test correction: rs707824 on chromosome 6p23 with NHL and rs12289961 on chromosome 11q12.1 with LYM. Thus, two novel susceptibility loci replicated in stage 2. Notably, the SNPs at 11q12.1 also are nominally significant (P<0.05) in the NFD subgroup, which is different from the observation for the SNPs at 6p21.32. Based on this analysis, nine of these SNPs were advanced to a meta-analysis of both stage-1 and stage-2 data (Table 1).
Tab. 1. Summary statistics for the analyses in all three stages. 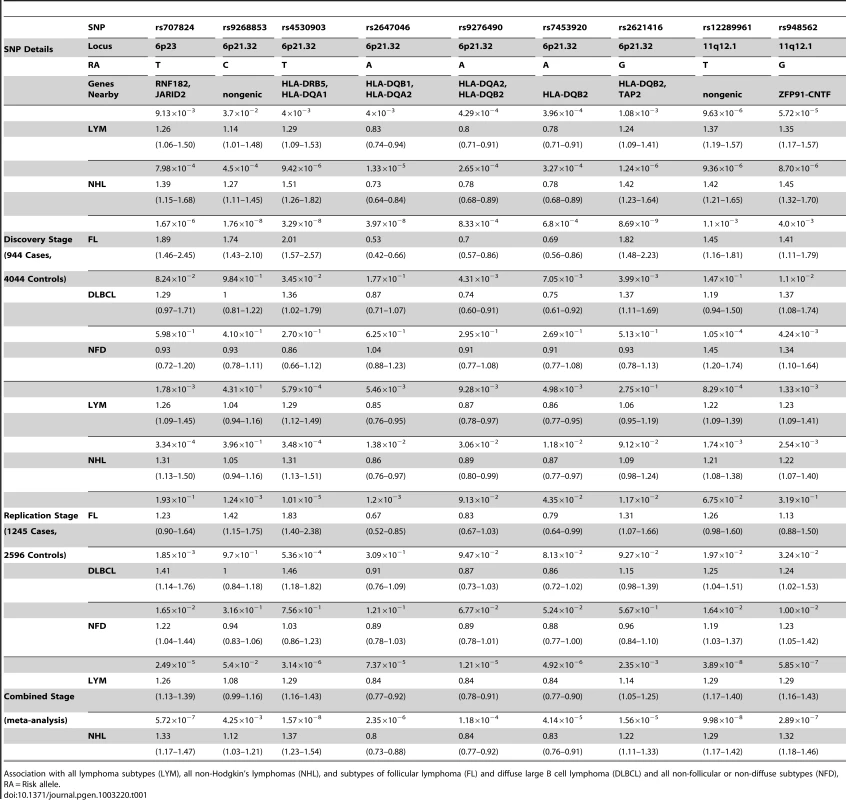
Association with all lymphoma subtypes (LYM), all non-Hodgkin's lymphomas (NHL), and subtypes of follicular lymphoma (FL) and diffuse large B cell lymphoma (DLBCL) and all non-follicular or non-diffuse subtypes (NFD), RA = Risk allele. Meta-analysis of the combined Stage-1 and Stage-2
Confirmation of the 6p21.32 HLA association in FL
To combine data from stages 1 and 2, a meta-analysis of nine SNPs for one or more subtypes of lymphoma (Table 1) was performed. We replicated the previously reported association with 6p21.32 region and FL; a novel SNP in this region, rs4530903 was associated with both FL (PFL = 2.69×10−12, OR = 1.93) as well as NHL (PNHL = 1.57×10−8, OR = 1.37). rs4530903 was correlated with a previously reported SNP, rs10484561 (r2 = 0.84, D′ = 0.95), associated with FL [6]. In addition, rs9268853 (PFL = 2.48×10−10, OR = 1.56) and rs2647046 (PFL = 3.77×10−10, OR = 0.59) were also significantly associated with FL (r2 = 0.43, D′ = 1.0). These SNPs showed very little evidence of association in DLBCL. Three of these nine SNPs mapped to two chromosomal regions not previously reported, 6p23 and 11q12.1. The SNP at 6p23, rs707824 (PNHL = 5.72×10−7, OR = 1.33) fell just below the genome wide threshold of significance.
Two novel SNPs associated with LYM and NHL at 11q12.1
Two SNPs in a novel region at 11q12.1 that were significantly associated with combined lymphoma subtypes were discovered. rs12289961, a nongenic SNP, showed evidence of association in the combined meta-analysis of the two phases (PLYM = 3.89×10−8, OR = 1.29). Another SNP, rs948562 at 11q12.1, 287 kb distal to rs12289961, showed a similar trend for association (PLYM = 5.85×10−7, OR = 1.29; PNHL = 2.89×10−7, OR = 1.32). These SNPs are weakly correlated (r2 = 0.6, D′ = 0.86) and were not highly significant for the major subtypes FL and DLBCL (Table 1). However, these SNPs were the most significant of the nine SNPs in the NFD group, suggesting that the FL and DLBCL signals were not driving these associations. Heterogeneity amongst the major subtypes FL, DLBCL, and NFD was not seen for the SNPs on chr11q12.1 and 6p23, while all SNPs except rF453920 at 6p21.32 showed heterogeneity in effect sizes (Table 2). The same trend was found when these SNPs were tested amongst cases with and without a family history of lymphoma (Table 2). These data suggest evidence of a novel mechanism of shared susceptibility to lymphoma associated with the 11q12.1 and 6p23 regions. The evidence for heterogeneity at 6p21.32 was weaker when HD was excluded from the analysis.
Tab. 2. Heterogeneity test. 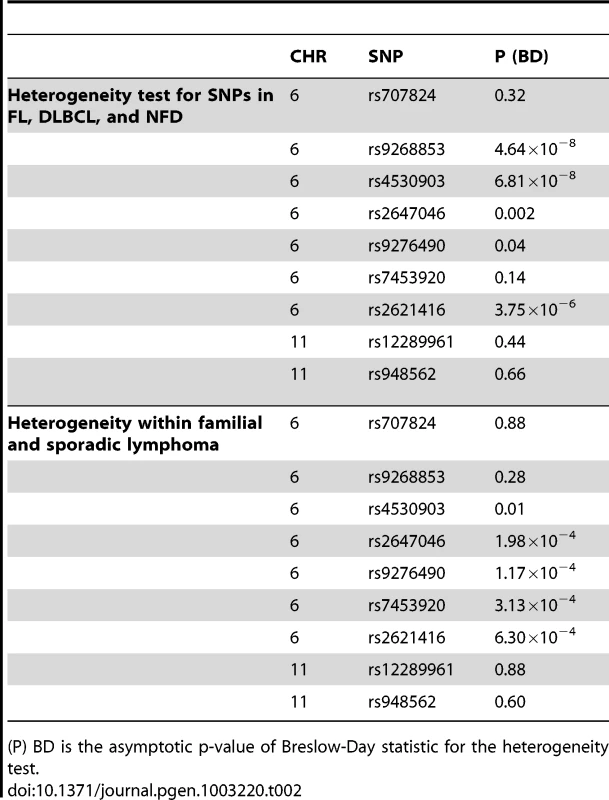
(P) BD is the asymptotic p-value of Breslow-Day statistic for the heterogeneity test. The data show the known regions of association with FL at 6p21.32 to include four more SNPs. These novel SNPs were rs2647046 (PFL = 3.77×10−10, OR = 0.59), rs9268853 (PFL = 2.48×10−10, OR = 1.56), and rs2621416 (PFL = 2.41×10−9, OR = 1.57, Figure 4). Two of these SNPs at 6p21.32, rs9268853 and rs2621416, were predominantly associated with FL and did not show an association with the aggregate set of all LYM (Table 1). SNPs rs4530903, rs707824 and rs2647046 demonstrated p-value less than 5×10−5 in NHL, likely driven by the association signal in FL and DLBCL at this locus. In addition, SNPs rs9268853, rs4530903 and rs2621416 showed association (P<0.05) with the HD subtype, while only rs2647046 showed an association in multiple myeloma (P<0.1).
Fig. 4. The Chr6p21.32 locus depicting the novel loci near the HLA II locus. 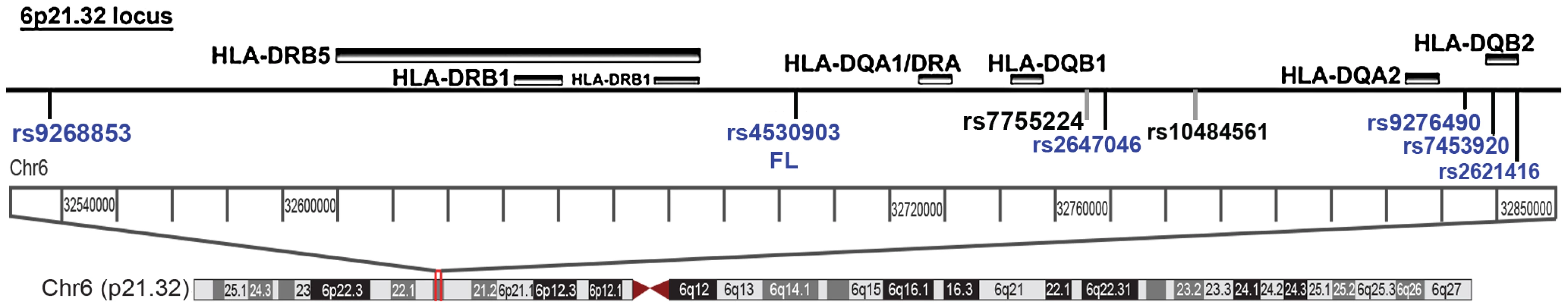
SNPs marked in blue are novel SNPs that are significant in LYM, NHL or FL. The two SNPs previously reported are rs7755224 and rs10484561 (in black). As a further demonstration of locus heterogeneity at 6p21.32 for FL, rs9268853, rs2647046 and rs2621416 are not in LD with any previously reported FL SNPs in this region (Table S3). A step-wise conditional logistic regression analysis of the FL SNPs from the phase-1 was performed. These investigations in Table 3 suggest some of the SNPs at the HLA locus are independent of the most significant SNP rs4530903. Hence, three SNPs rs4530903, rs9268853 and rs2621416 appear to be independent SNPs. The minor allele frequencies for these SNPs were comparable to the Hapmap population across the three stages.
Tab. 3. Conditional logistic regression analysis for the 6p21.32 SNPs in FL. 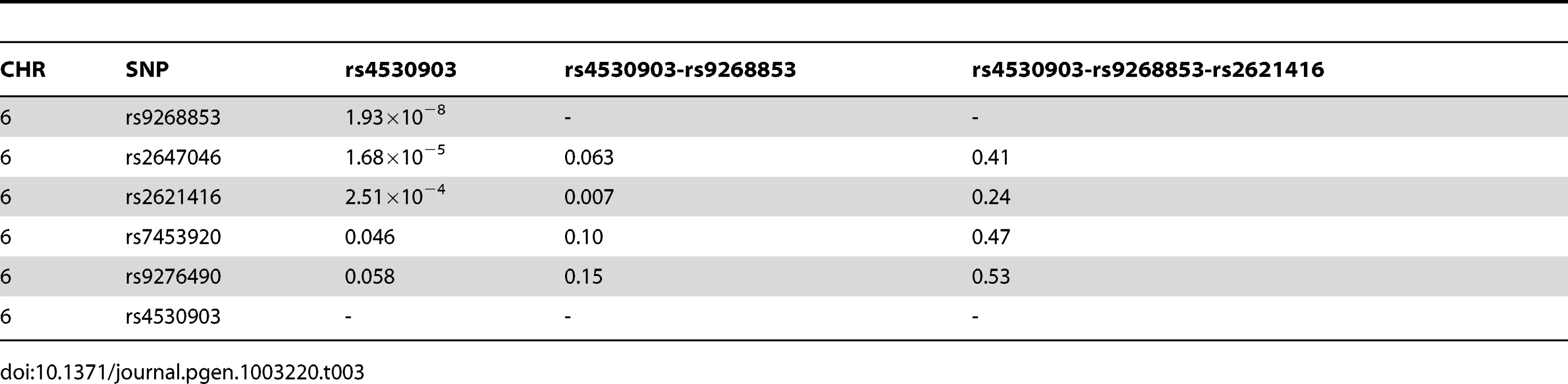
eQTL analysis
Analysis of the available data on expression quantitative trait loci (eQTL) on lymphoblastoid cell lines [12] for the lead SNPs and corresponding candidate genes from the GWAS was performed. In the cis - eQTLs-gene analysis, we identified eQTL in the two genes of interest based on the SNP P-values obtained from the association study using the database utility GENEVAR [13]. rs3129763 in HLA-DQA2 showed the strongest SNP-gene association (Figure S2, Figure S3) (P = 1.23×10−13), while rs241440 (Figure S2, Figure S4) showed association (P = 3.3×10−7) with TAP2 (Figure S2). Presence of any SNP that was in linkage disequilibrium with the expression probe was checked in HapMap data. There was a SNP rs9276442 in the expression array probe for HLA-DQA2. However, it is 124 kb upstream from the most significant eQTL SNP, rs3129763 and examination of haplotype blocks in the HapMap suggests that these two SNPs are not in LD. It is unlikely that the eQTL association is a consequence of perturbed probe binding due to the SNP. A lymphoma risk SNP, rs948562 identified at chromosome 11q12.1, was associated with expression levels of OR9Q in lymphocytes (Padj = 2.49×10−2, adjusted using non-parametric permutation, Figure S2).
Shared variants
In a global analyses of all SNPs with associations at P<1×10−3, we observed that most SNPs were exclusive to the subtypes FL and DLBCL, thus reinforcing the notion of subtype-specific etiologic pathways. We found an overlap of only two SNPs between DLBCL and FL, confirming distinct genetic susceptibility in these subtypes. Predictably, about a half of the variants (P<10−3) were shared between LYM and NHL (Figure S5A). The same trend was noted in a gene-set enrichment analysis. Within the top 100 genes enriched in LYM, NHL, FL and DLBCL, we found the majority of genes associated with specific lymphoma subtypes, with a few genes common between them. One gene, RELN, was common to all subsets and groupings (Figure S5B). Since the sample sizes for the subtypes were small, this analysis was not done for each individual subtypes of NHL. In FL, significantly associated genes were HLA-DOB, HLA-DQA2, TAP2, HLA-DRA, HLA-DQB1 and HLA-DRB1 (PFL<4×10−8) and in LYM, these were HES6, ILKAP, PER2, FOXP1, OR5 family members, and ATF6 (PLYM<1×10−5).
Discussion
The major finding of this study is the observation that some regions are most strongly associated with a particular subtype of lymphoma, e.g. 6p21.32 in FL, while others are most strongly associated with combined types of lymphoma, e.g. the novel regions on 11q12.1. Evidence favoring a model of common susceptibility loci includes observations of familial clustering of multiple subtypes of lymphoma. Several studies have now discovered pre-disposing genetic loci at the HLA region for FL, DLBCL, CLL and HD [4], [5], [6], [7], [8] and some of these reports highlight the existence of shared susceptibility loci at the individual subtype levels that were studied. Etiologically, patients with HD have a higher risk of developing NHL as a secondary malignancy [14]. Similarly, patients with NHL have a higher risk of developing HD at a later stage [15]. At a molecular level, the model of common susceptibility pathways is supported by recent studies examining the coding sequences and genomes of non-Hodgkin's lymphomas, which have demonstrated increased mutation burden in shared genes [16], [17]. In addition, recent tumor analysis has demonstrated that DLBCL and FL share somatic mutations in the same chromatin and histone modifying genes, MLL2 and MEF2B, respectively [16]. Such evidence notwithstanding, a direct test of subtype-specific association would require a very large number of cases per subtype, feasible as part of a combined consortium approach. However, as a first approximation of shared versus subtype specific susceptibilities to lymphoma, it is possible to determine if a putative locus shows heterogeneity. For the 11q12.1 region shown here to be a pan-lymphoma susceptibility locus, there was no evidence of such heterogeneity within the largest subtypes.
Of the susceptibility markers reported here, the 6p21.32 HLA II region has been previously associated with FL and NHL [4], [5], [6]. In our report, the 6p21.32 region was implicated by three SNPS; rs4530903 upstream from HLA-DRB1 and HLA-DQA1, rs2621416 upstream of HLA-DQB2, and rs9268853 downstream of HLA-DRA, HLA-DRB5 and HLA-DRB1, but upstream of BTLN2. rs2621416 and rs9268853 have also been associated with risk for ulcerative colitis [18] and rheumatoid arthritis [19] respectively, both of which increases risk for certain types of lymphoma. Allelic heterogeneity at this same locus has also been demonstrated in FL, with both protective and risk alleles described [6]. rs2647012, a previously reported SNP [6] is correlated (r2 = 1, D′ = 1) with rs2647046 in our results. None of the 6p21.32 SNPs are correlated with rs10484561, the HLA-associated SNP previously described [4]. Our data support the earlier findings of allelic heterogeneity at this region, with a slightly stronger magnitude of the effect size.
The novel regions reported here include 6p23 and 11q12.1, represented by SNPs mapping near genes with biologically plausible ties to lymphoid development. The novel SNP at 6p23, rs707824, is upstream of JARID2, encoding Jumonji, which co-localizes with the polycomb repressive complex 2 and H3K27me3 on chromatin and plays a role in self-renewal and differentiation of embryonic stem cells [20]. JARID2 is regulated by miR-155 where very high levels decrease endogenous JARID2 mRNA levels [21]. High levels of miR-155 are observed in different types of B-cell lymphomas (DLBCL, HD and latency type III EBV-positive Burkitt lymphoma), and transgenic mice expressing miR155 at the late pro-B-cell stage of differentiation developed B-cell tumors. JARID2/Jumonji-deficient mice have widespread developmental defects including abnormalities of hematopoiesis [22]. rs707824 is located downstream of CD83. CD83 antigen, also known as B-cell activation protein, is expressed on dendritic cells and is thought to have roles in the modulation of antigen presentation and CD4+ T cell generation [23].
The 11q12.1 region reported here was marked by two SNPs, rs948562, located within the non-coding gene ZFP91, and rs12289961. rs12289961 at 11q12.1 is 230 kb upstream of the LPXN (leupaxin) locus, originally identified binding to alpha4 integrins and playing a role in integrin-mediated cell adhesion [24]. LPXN was found to be a member of a fusion protein with RUNX1 in human acute leukemia where wild-type LPXN was shown to transform NIH 3T3 cells [25]. Particularly relevant to its putative role suggested here in B-cell lymphomagenesis, LPXN is preferentially expressed in hematopoietic cells and plays an inhibitory role in B-cell antigen receptor signaling and B-cell function [26].
eQTL analysis showed that there was overlap between the most significant SNPs in the GWAS and lymphoblastoid cell lines cis-eQTL candidate genes, such as HLA-DQA2 and TAP2. HLA-DQA2 plays a pivotal role in the immune system by presenting peptides derived from extracellular proteins. Gene set enrichment analysis showed interesting candidates related to lymphomagenesis and hematopoietic cell development in the top 20 significant genes. The one variant common in all gene enrichment analyses was RELN, which has been shown to be recurrently mutated in acute lymphocytic leukemia [27].
Based on patterns of inheritance of multiple subtypes of lymphoid neoplasms in families, as well as from the GWAS data reported here, there is evidence to suggest that multiple phenotypes of lymphoma may be associated with shared common genetic predispositions. The candidate genes uncovered in this GWAS suggest that in addition to the genes involved in immune regulation, such as HLA and JARID2, those involved in B-cell development (e.g. LPXN) are logical targets for further studies. It is possible that the GWAS associations with multiple phenotypes reported here have resulted from the ascertainment utilized, since the study was enriched with a familial subset of samples. However, we included only one individual from each kindred, precluding a spurious association of a single SNP with multiple phenotypes in the same family. SNPs that show shared susceptibility, including some of those discovered here, may yet have strongest association with specific lymphoma subtypes. While this study reports associations within combined smaller subtypes, e.g. mantle cell and marginal zone lymphoma, larger sample sizes will be required to delineate whether these and other associations are shared or subtype specific.
Thus, we have described two novel lymphoma-susceptibility regions, one at 11q12.1 and another putative susceptibility locus at 6p23, and further characterized the 6p21.32 (HLA class II) association signal observed in a prior GWAS of FL. While genetic susceptibility to lymphoma has been viewed as subtype specific, here we propose an alternate model. Based on our analysis of the overlap between genotypes and phenotypes (Figure S5), we predict that the shared loci associated with multiple subtypes of lymphoma will be less frequent than subtype-specific susceptibilities. Finally, the effect sizes observed in this report (0.59–1.93) are somewhat higher than those previously reported, e.g. for breast and colon cancer, but well below thresholds required for clinical utility [28]. As in other cancer genome-wide association studies, the novel loci reported here harbor interesting genes in pathways that regulate hematopoiesis, offering potential new insights into the pathogenesis of lymphoid neoplasms.
Methods
Ethics statement
All cases were ascertained through Memorial Sloan-Kettering Cancer Center IRB-approved protocols, or a protocol approved by the IRB at the Dana Farber Cancer Institute or Hadassah Hebrew University (Table S4). These protocols either required informed consent for identified use of specimens for research into the genetic basis of lymphoma, or allowed research use of specimens permanently de-identified prior to genotyping.
Sample selection for stage-1 and stage-2
The stage-1 of our study was comprised of 944 unrelated probands. This ascertainment was enriched to included 282 cases of familial lymphoproliferative syndrome, defined as two or more lymphoid cancers in the same lineage. These kindreds were characterized by mixed phenotypes of lymphoid malignancy (Figure S6), and kindreds contained from 2 to 5 affected relatives. In addition, stage-1 contained 107 cases of lymphoma with a first degree relative affected by a lymphoid malignancy, and 347 cases of early onset (age of diagnosis <45 years) lymphoma. Stage 2 was comprised of 1245 unrelated lymphoma probands from a prevalent ascertainment at MSKCC and unselected for specific histology or family history of lymphoma. Lymphomas were categorized according to a modification of the 2008 World Health Organization classification system; primary reports were obtained in all cases and reviewed by two of the authors (KO and AZ). Because of the presence of multiple subtypes in kindreds with familial lymphoma, all subtypes of B and T cell lymphoma, including Hodgkin's disease and plasma cell neoplasms were included in both stage 1 and 2, although it was recognized that sizes of these subgroups would be too small to allow subset analysis. The sample distribution of histologic subsets of lymphoma mirrors the prevalence of the disease subtypes in the US population.
Genotyping for Stage-1 and quality control of data
Genotyping of the cases was performed utilizing the Affymetrix 6.0 SNP array. For control data, Bipolar and GENEVA Diabetes Study (NHS/HPFS) data were downloaded from dbGAP (accession phs000017.v3 http://1.usa.gov/xrXL1D and phs000091.v2 http://1.usa.gov/yevUOY). Affymetrix SNP 6.0 CEL files were arranged according to the batches in which data were originally genotyped. Data were initially quality checked for the gender and Mini-DM thresholds. Only CEL files that passed a Mini-DM >85% were used in the full Birdseed [29] genotyping of the 906,000 SNPs. The mean heterozygosity of each sample was computed (26.8) and samples with low or high heterozygosity were excluded. Samples that passed >95% Birdseed calls were further processed to generate PLINK [30] formatted files, using only calls that had copy number state two and a confidence score >0.9. This was performed using the utility Birdsuite to PLINK from Broad Institute. Hapmap controls were removed. In addition, any sample that showed abnormal copy number profile states in Birdsuite were excluded (CN0%, CN1%, CN2%, CN3% and CN4%). Particular attention was paid to any samples that had the CLL/SLL phenotype in the copy number variability screen, to exclude samples with somatic mosaicism caused by circulating tumor cells. Individuals from dbGaP marked as controls in the data-manifest were retained for further study. Samples with genetic or cryptic relatedness were excluded by using the relationship score-matrix (PI_HAT<0.1) in the entire dataset. Data was filtered for multi-mapping, mitochondrial and monomorphic SNPs on the Affymetrix 6.0 SNP Chip. Individuals and SNPs were filtered for 95% genotyping rate and departures from Hardy-Weinberg equilibrium [31]. SNPs were also removed if they failed differential missing or haplotype-based differential missing tests as implemented in PLINK. Finally, the data was matched against previously called genotyping data from dbGAP for a subset of SNPs and their allele frequencies. Analyses were carried out on 944 cases and 4044 controls on 530,583 SNPs. Principal component analysis was carried out to test for population match in both cases and controls (Figure S7). Association was performed using case-control status with each phenotype specifically defined, along with age and the first four eigenvectors from the output of EIGENSTRAT [32] program using logistic regression.
Control data for stage-2: New York Cancer Project
Controls for the replication were gathered from the New York Cancer Project (NYCP), which is a study of 18,000 New York City residents that allows researchers to better understand how factors such as environment, lifestyle, diet, family health history, and genetics affect the development of cancer and an array of other life threatening diseases. The data include age, gender, history of cancers (including lymphoma) and ethnicity [33]. All subjects consented to use of samples to study the genetics of any disease state. Only samples with self-declared European ancestry were used for stage-2. Since individuals of Ashkenazi Jewish ethnicity formed a subset of both ascertainments, ethnicity was used as one of the covariates in the analysis in stage-2. Genotyping for stage-2 was carried out by designing multiplexed PCR using Sequenom iPLEX assays and analyzed using MassARRAY [34]. Genotypes were called using TYPER 4.0.2 software.
Imputing SNPs at Chr6 and Chr11 loci
The dataset (BED, BIM, FAM) was split to each chromosome, then subset using gtool [35] to create .gen and .sample files. Imputation was done using pre-phasing and best-guess imputing using IMPUTE2 [36] with the references used being 1000 genomes and Hapmap3 populations for genome build v36. Best practices for imputation of the data were followed The dataset (BED, BIM, FAM) was split to each chromosome, then subset using gtool [35] to create .gen and .sample files. Imputation was done using pre-phasing and best-guess imputing using IMPUTE2 [36] with the references used being 1000 genomes and Hapmap3 populations for genome build v36. Best practices for imputation of the data were followed [37]. The dosage output was filtered for confidence scores and analyzed using PLINK, filtered on INFO and plotted using locuszoom [38]. Haplotypes were viewed in Haploview [39]. The dosage output was filtered for confidence scores and analyzed using PLINK, filtered on INFO and plotted using locuszoom [38]. Haplotypes were viewed in Haploview [39].
Selection of SNPs into Stage-2
SNPs were ranked on p-value in both major types and subtype specific analyses. Each index-ranked SNP (within top 100 SNPs) was graded based on a custom script used to generate scatterplots from Birdsuite, which were inspected and graded on the cluster separation and skew. In order to prioritize the SNPs that were to be replicated, SNPs were given a negative grade if they were singletons (i.e. neighboring SNPs not showing low p-values). A positive grade was given if a given SNP showed low p-value (P<5×10−4) in any other type or subtype. Only SNPs with good scatterplots were selected for the iPLEX design. Analysis was performed by logistic regression using the same criteria as stage-1, however, instead of the PCA, self-reported ethnicity information was used. Only Caucasian samples were used in the replication study. A meta-analysis of the stage-1 and stage-2 data was performed using the results of the logistic regression. For test of heterogeneity specifically for the 6p21.32 locus, the combined dataset consisting of stage-1 and stage-2 was split into three major groups namely FL, DLBCL and any other NHL subgroup designated as NFD in this report. Since we have only one control set, the control samples were randomly assigned in a fixed ratio to match the percent cases per subset without replacement. The three clusters were joined together to perform Breslow-Day test using PLINK.
Gene set enrichment analysis
We performed gene set enrichment analysis using the p-values from each of the subgroup and group analyses. The program VEGAS [40] was used to compute the gene enrichment analyses. It annotates SNPs to corresponding genes (±50 kb boundaries), produces a gene-based test statistic, and then uses simulation to calculate an empirical gene-based p-value. The Hapmap population was used as a reference. The top 10 percent of significant SNPs were chosen for the analysis with simulation performed 106 times. Venn diagram was created using Venny (http://bioinfogp.cnb.csic.es/tools/venny/index.html).
eQTL analysis in lymphoblastoid cell line
We analyzed available hapmap3 population data from lymphoblastoid cell lines [12] for eQTLs [12] using GENEVAR [13]. Two types of analyses were performed, (1) identifying cis-eQTLs in candidate genes discovered from the GWAS and (2) SNP-gene association analysis. Adjusted p-values (Padj) were derived from 10,000 permutations as implemented on the GENEVAR applet.
Supporting Information
Zdroje
1. PileriSA, MilaniM, Fraternali-OrcioniG, SabattiniE (1998) From the REAL classification to the upcoming WHO scheme: A step toward universal categorization of lymphoma entities? Ann Oncol 9 : 607–612.
2. ZintzarasE, VoulgarelisM, MoutsopoulosHM (2005) The risk of lymphoma development in autoimmune diseases: a meta-analysis. Arch Intern Med 165 : 2337–2344.
3. SiddiquiR, OnelK, FacioF, OffitK (2004) The genetics of familial lymphomas. Curr Oncol Rep 6 : 380–387.
4. CondeL, HalperinE, AkersNK, BrownKM, SmedbyKE, et al. (2010) Genome-wide association study of follicular lymphoma identifies a risk locus at 6p21.32. Nat Genet 42 : 661–664.
5. SkibolaCF, BracciPM, HalperinE, CondeL, CraigDW, et al. (2009) Genetic variants at 6p21.33 are associated with susceptibility to follicular lymphoma. Nat Genet 41 : 873–875.
6. SmedbyKE, FooJN, SkibolaCF, DarabiH, CondeL, et al. (2011) GWAS of follicular lymphoma reveals allelic heterogeneity at 6p21.32 and suggests shared genetic susceptibility with diffuse large B-cell lymphoma. PLoS Genet 7: e1001378 doi:10.1371/journal.pgen.1001378.
7. Enciso-MoraV, BroderickP, MaY, JarrettRF, HjalgrimH, et al. (2010) A genome-wide association study of Hodgkin's lymphoma identifies new susceptibility loci at 2p16.1 (REL), 8q24.21 and 10p14 (GATA3). Nat Genet 42 : 1126–1130.
8. MoutsianasL, Enciso-MoraV, MaYP, LeslieS, DiltheyA, et al. (2011) Multiple Hodgkin lymphoma-associated loci within the HLA region at chromosome 6p21.3. Blood 118 : 670–674.
9. KumarV, MatsuoK, TakahashiA, HosonoN, TsunodaT, et al. (2011) Common variants on 14q32 and 13q12 are associated with DLBCL susceptibility. J Hum Genet 56 : 436–439.
10. MukherjeeS, SimonJ, BayugaS, LudwigE, YooS, et al. (2011) Including additional controls from public databases improves the power of a genome-wide association study. Hum Hered 72 : 21–34.
11. MenasheI, RosenbergPS, ChenBE (2008) PGA: power calculator for case-control genetic association analyses. BMC Genet 9 : 36.
12. DimasAS, DeutschS, StrangerBE, MontgomerySB, BorelC, et al. (2009) Common regulatory variation impacts gene expression in a cell type-dependent manner. Science 325 : 1246–1250.
13. YangTP, BeazleyC, MontgomerySB, DimasAS, Gutierrez-ArcelusM, et al. (2010) Genevar: a database and Java application for the analysis and visualization of SNP-gene associations in eQTL studies. Bioinformatics 26 : 2474–2476.
14. DoresGM, MetayerC, CurtisRE, LynchCF, ClarkeEA, et al. (2002) Second malignant neoplasms among long-term survivors of Hodgkin's disease: a population-based evaluation over 25 years. J Clin Oncol 20 : 3484–3494.
15. TravisLB, CurtisRE, GlimeliusB, HolowatyE, Van LeeuwenFE, et al. (1993) Second cancers among long-term survivors of non-Hodgkin's lymphoma. J Natl Cancer Inst 85 : 1932–1937.
16. MorinRD, Mendez-LagoM, MungallAJ, GoyaR, MungallKL, et al. (2011) Frequent mutation of histone-modifying genes in non-Hodgkin lymphoma. Nature 476 : 298–303.
17. PasqualucciL, TrifonovV, FabbriG, MaJ, RossiD, et al. (2011) Analysis of the coding genome of diffuse large B-cell lymphoma. Nat Genet 43 : 830–837.
18. AndersonCA, BoucherG, LeesCW, FrankeA, D'AmatoM, et al. (2011) Meta-analysis identifies 29 additional ulcerative colitis risk loci, increasing the number of confirmed associations to 47. Nat Genet 43 : 246–252.
19. EleftherohorinouH, HoggartCJ, WrightVJ, LevinM, CoinLJ (2011) Pathway-driven gene stability selection of two rheumatoid arthritis GWAS identifies and validates new susceptibility genes in receptor mediated signalling pathways. Hum Mol Genet 20 : 3494–3506.
20. ShenX, KimW, FujiwaraY, SimonMD, LiuY, et al. (2009) Jumonji modulates polycomb activity and self-renewal versus differentiation of stem cells. Cell 139 : 1303–1314.
21. BolisettyMT, DyG, TamW, BeemonKL (2009) Reticuloendotheliosis virus strain T induces miR-155, which targets JARID2 and promotes cell survival. J Virol 83 : 12009–12017.
22. TakeuchiT, WatanabeY, Takano-ShimizuT, KondoS (2006) Roles of jumonji and jumonji family genes in chromatin regulation and development. Dev Dyn 235 : 2449–2459.
23. CaoW, LeeSH, LuJ (2005) CD83 is preformed inside monocytes, macrophages and dendritic cells, but it is only stably expressed on activated dendritic cells. Biochem J 385 : 85–93.
24. LiuS, ThomasSM, WoodsideDG, RoseDM, KiossesWB, et al. (1999) Binding of paxillin to alpha4 integrins modifies integrin-dependent biological responses. Nature 402 : 676–681.
25. DaiHP, XueYQ, ZhouJW, LiAP, WuYF, et al. (2009) LPXN, a member of the paxillin superfamily, is fused to RUNX1 in an acute myeloid leukemia patient with a t(11;21)(q12;q22) translocation. Genes Chromosomes Cancer 48 : 1027–1036.
26. ChewV, LamKP (2007) Leupaxin negatively regulates B cell receptor signaling. J Biol Chem 282 : 27181–27191.
27. ZhangJ, DingL, HolmfeldtL, WuG, HeatleySL, et al. (2012) The genetic basis of early T-cell precursor acute lymphoblastic leukaemia. Nature 481 : 157–163.
28. StadlerZK, ThomP, RobsonME, WeitzelJN, KauffND, et al. (2010) Genome-wide association studies of cancer. J Clin Oncol 28 : 4255–4267.
29. KornJM, KuruvillaFG, McCarrollSA, WysokerA, NemeshJ, et al. (2008) Integrated genotype calling and association analysis of SNPs, common copy number polymorphisms and rare CNVs. Nat Genet 40 : 1253–1260.
30. PurcellS, NealeB, Todd-BrownK, ThomasL, FerreiraMA, et al. (2007) PLINK: a tool set for whole-genome association and population-based linkage analyses. Am J Hum Genet 81 : 559–575.
31. LaurieCC, DohenyKF, MirelDB, PughEW, BierutLJ, et al. (2010) Quality control and quality assurance in genotypic data for genome-wide association studies. Genet Epidemiol 34 : 591–602.
32. PriceAL, PattersonNJ, PlengeRM, WeinblattME, ShadickNA, et al. (2006) Principal components analysis corrects for stratification in genome-wide association studies. Nat Genet 38 : 904–909.
33. MitchellMK, GregersenPK, JohnsonS, ParsonsR, VlahovD (2004) The New York Cancer Project: rationale, organization, design, and baseline characteristics. J Urban Health 81 : 301–310.
34. JurinkeC, van den BoomD, CantorCR, KosterH (2002) The use of MassARRAY technology for high throughput genotyping. Adv Biochem Eng Biotechnol 77 : 57–74.
35. FreemanC, MarchiniJ (2007) GTOOL.
36. HowieBN, DonnellyP, MarchiniJ (2009) A flexible and accurate genotype imputation method for the next generation of genome-wide association studies. PLoS Genet 5: e1000529 doi:10.1371/journal.pgen.1000529.
37. de BakkerPI, FerreiraMA, JiaX, NealeBM, RaychaudhuriS, et al. (2008) Practical aspects of imputation-driven meta-analysis of genome-wide association studies. Hum Mol Genet 17: R122–128.
38. PruimRJ, WelchRP, SannaS, TeslovichTM, ChinesPS, et al. (2010) LocusZoom: regional visualization of genome-wide association scan results. Bioinformatics 26 : 2336–2337.
39. BarrettJC, FryB, MallerJ, DalyMJ (2005) Haploview: analysis and visualization of LD and haplotype maps. Bioinformatics 21 : 263–265.
40. LiuJZ, McRaeAF, NyholtDR, MedlandSE, WrayNR, et al. (2010) A versatile gene-based test for genome-wide association studies. Am J Hum Genet 87 : 139–145.
Štítky
Genetika Reprodukční medicína
Článek Comparative Genome Structure, Secondary Metabolite, and Effector Coding Capacity across PathogensČlánek TATES: Efficient Multivariate Genotype-Phenotype Analysis for Genome-Wide Association StudiesČlánek Secondary Metabolism and Development Is Mediated by LlmF Control of VeA Subcellular Localization inČlánek Human Disease-Associated Genetic Variation Impacts Large Intergenic Non-Coding RNA ExpressionČlánek The Roles of Whole-Genome and Small-Scale Duplications in the Functional Specialization of GenesČlánek The Role of Autophagy in Genome Stability through Suppression of Abnormal Mitosis under Starvation
Článek vyšel v časopisePLOS Genetics
Nejčtenější tento týden
2013 Číslo 1
-
Všechny články tohoto čísla
- A Model of High Sugar Diet-Induced Cardiomyopathy
- Comparative Genome Structure, Secondary Metabolite, and Effector Coding Capacity across Pathogens
- Emerging Function of Fat Mass and Obesity-Associated Protein (Fto)
- Positional Cloning Reveals Strain-Dependent Expression of to Alter Susceptibility to Bleomycin-Induced Pulmonary Fibrosis in Mice
- Genetics of Ribosomal Proteins: “Curiouser and Curiouser”
- Transposable Elements Re-Wire and Fine-Tune the Transcriptome
- Function and Regulation of , a Gene Implicated in Autism and Human Evolution
- MAML1 Enhances the Transcriptional Activity of Runx2 and Plays a Role in Bone Development
- Predicting Mendelian Disease-Causing Non-Synonymous Single Nucleotide Variants in Exome Sequencing Studies
- A Systematic Mapping Approach of 16q12.2/ and BMI in More Than 20,000 African Americans Narrows in on the Underlying Functional Variation: Results from the Population Architecture using Genomics and Epidemiology (PAGE) Study
- Transcription of the Major microRNA–Like Small RNAs Relies on RNA Polymerase III
- Histone H3K56 Acetylation, Rad52, and Non-DNA Repair Factors Control Double-Strand Break Repair Choice with the Sister Chromatid
- Genome-Wide Association Study Identifies a Novel Susceptibility Locus at 12q23.1 for Lung Squamous Cell Carcinoma in Han Chinese
- Genetic Disruption of the Copulatory Plug in Mice Leads to Severely Reduced Fertility
- The [] Prion Exists as a Dynamic Cloud of Variants
- Adult Onset Global Loss of the Gene Alters Body Composition and Metabolism in the Mouse
- Fis Protein Insulates the Gene from Uncontrolled Transcription
- The Meiotic Nuclear Lamina Regulates Chromosome Dynamics and Promotes Efficient Homologous Recombination in the Mouse
- Genome-Wide Haplotype Analysis of Expression Quantitative Trait Loci in Monocytes
- TATES: Efficient Multivariate Genotype-Phenotype Analysis for Genome-Wide Association Studies
- Structural Basis of a Histone H3 Lysine 4 Demethylase Required for Stem Elongation in Rice
- The Ecm11-Gmc2 Complex Promotes Synaptonemal Complex Formation through Assembly of Transverse Filaments in Budding Yeast
- MCM8 Is Required for a Pathway of Meiotic Double-Strand Break Repair Independent of DMC1 in
- Comparative Genomic Analysis of the Endosymbionts of Herbivorous Insects Reveals Eco-Environmental Adaptations: Biotechnology Applications
- Integration of Nodal and BMP Signals in the Heart Requires FoxH1 to Create Left–Right Differences in Cell Migration Rates That Direct Cardiac Asymmetry
- Pharmacodynamics, Population Dynamics, and the Evolution of Persistence in
- A Hybrid Likelihood Model for Sequence-Based Disease Association Studies
- Aberration in DNA Methylation in B-Cell Lymphomas Has a Complex Origin and Increases with Disease Severity
- Multiple Opposing Constraints Govern Chromosome Interactions during Meiosis
- Transcriptional Dynamics Elicited by a Short Pulse of Notch Activation Involves Feed-Forward Regulation by Genes
- Dynamic Large-Scale Chromosomal Rearrangements Fuel Rapid Adaptation in Yeast Populations
- Heterologous Gln/Asn-Rich Proteins Impede the Propagation of Yeast Prions by Altering Chaperone Availability
- Gene Copy-Number Polymorphism Caused by Retrotransposition in Humans
- An Incompatibility between a Mitochondrial tRNA and Its Nuclear-Encoded tRNA Synthetase Compromises Development and Fitness in
- Secondary Metabolism and Development Is Mediated by LlmF Control of VeA Subcellular Localization in
- Single-Stranded Annealing Induced by Re-Initiation of Replication Origins Provides a Novel and Efficient Mechanism for Generating Copy Number Expansion via Non-Allelic Homologous Recombination
- Tbx2 Controls Lung Growth by Direct Repression of the Cell Cycle Inhibitor Genes and
- Suv4-20h Histone Methyltransferases Promote Neuroectodermal Differentiation by Silencing the Pluripotency-Associated Oct-25 Gene
- A Conserved Helicase Processivity Factor Is Needed for Conjugation and Replication of an Integrative and Conjugative Element
- Telomerase-Null Survivor Screening Identifies Novel Telomere Recombination Regulators
- Genome-Wide Analysis Reveals Selection for Important Traits in Domestic Horse Breeds
- Coordinated Degradation of Replisome Components Ensures Genome Stability upon Replication Stress in the Absence of the Replication Fork Protection Complex
- Nkx6.1 Controls a Gene Regulatory Network Required for Establishing and Maintaining Pancreatic Beta Cell Identity
- HIF- and Non-HIF-Regulated Hypoxic Responses Require the Estrogen-Related Receptor in
- Delineating a Conserved Genetic Cassette Promoting Outgrowth of Body Appendages
- The Telomere Capping Complex CST Has an Unusual Stoichiometry, Makes Multipartite Interaction with G-Tails, and Unfolds Higher-Order G-Tail Structures
- Comprehensive Methylome Characterization of and at Single-Base Resolution
- Loci Associated with -Glycosylation of Human Immunoglobulin G Show Pleiotropy with Autoimmune Diseases and Haematological Cancers
- Switchgrass Genomic Diversity, Ploidy, and Evolution: Novel Insights from a Network-Based SNP Discovery Protocol
- Centromere-Like Regions in the Budding Yeast Genome
- Sequencing of Loci from the Elephant Shark Reveals a Family of Genes in Vertebrate Genomes, Forged by Ancient Duplications and Divergences
- Mendelian and Non-Mendelian Regulation of Gene Expression in Maize
- Mutational Spectrum Drives the Rise of Mutator Bacteria
- Human Disease-Associated Genetic Variation Impacts Large Intergenic Non-Coding RNA Expression
- The Roles of Whole-Genome and Small-Scale Duplications in the Functional Specialization of Genes
- Sex-Specific Signaling in the Blood–Brain Barrier Is Required for Male Courtship in
- A Newly Uncovered Group of Distantly Related Lysine Methyltransferases Preferentially Interact with Molecular Chaperones to Regulate Their Activity
- Is Required for Leptin-Mediated Depolarization of POMC Neurons in the Hypothalamic Arcuate Nucleus in Mice
- Unlocking the Bottleneck in Forward Genetics Using Whole-Genome Sequencing and Identity by Descent to Isolate Causative Mutations
- The Role of Autophagy in Genome Stability through Suppression of Abnormal Mitosis under Starvation
- MTERF3 Regulates Mitochondrial Ribosome Biogenesis in Invertebrates and Mammals
- Downregulation and Altered Splicing by in a Mouse Model of Facioscapulohumeral Muscular Dystrophy (FSHD)
- NBR1-Mediated Selective Autophagy Targets Insoluble Ubiquitinated Protein Aggregates in Plant Stress Responses
- Retroactive Maintains Cuticle Integrity by Promoting the Trafficking of Knickkopf into the Procuticle of
- Phenome-Wide Association Study (PheWAS) for Detection of Pleiotropy within the Population Architecture using Genomics and Epidemiology (PAGE) Network
- Genetic and Functional Modularity of Activities in the Specification of Limb-Innervating Motor Neurons
- A Population Genetic Model for the Maintenance of R2 Retrotransposons in rRNA Gene Loci
- A Quartet of PIF bHLH Factors Provides a Transcriptionally Centered Signaling Hub That Regulates Seedling Morphogenesis through Differential Expression-Patterning of Shared Target Genes in
- A Genome-Wide Integrative Genomic Study Localizes Genetic Factors Influencing Antibodies against Epstein-Barr Virus Nuclear Antigen 1 (EBNA-1)
- Mutation of the Diamond-Blackfan Anemia Gene in Mouse Results in Morphological and Neuroanatomical Phenotypes
- Life, the Universe, and Everything: An Interview with David Haussler
- Alternative Oxidase Expression in the Mouse Enables Bypassing Cytochrome Oxidase Blockade and Limits Mitochondrial ROS Overproduction
- An Evolutionarily Conserved Synthetic Lethal Interaction Network Identifies FEN1 as a Broad-Spectrum Target for Anticancer Therapeutic Development
- The Flowering Repressor Underlies a Novel QTL Interacting with the Genetic Background
- Telomerase Is Required for Zebrafish Lifespan
- and Diversified Expression of the Gene Family Bolster the Floral Stem Cell Network
- Susceptibility Loci Associated with Specific and Shared Subtypes of Lymphoid Malignancies
- An Insertion in 5′ Flanking Region of Causes Blue Eggshell in the Chicken
- Increased Maternal Genome Dosage Bypasses the Requirement of the FIS Polycomb Repressive Complex 2 in Arabidopsis Seed Development
- WNK1/HSN2 Mutation in Human Peripheral Neuropathy Deregulates Expression and Posterior Lateral Line Development in Zebrafish ()
- Synergistic Interaction of Rnf8 and p53 in the Protection against Genomic Instability and Tumorigenesis
- Dot1-Dependent Histone H3K79 Methylation Promotes Activation of the Mek1 Meiotic Checkpoint Effector Kinase by Regulating the Hop1 Adaptor
- A Heterogeneous Mixture of F-Series Prostaglandins Promotes Sperm Guidance in the Reproductive Tract
- Starvation, Together with the SOS Response, Mediates High Biofilm-Specific Tolerance to the Fluoroquinolone Ofloxacin
- Directed Evolution of a Model Primordial Enzyme Provides Insights into the Development of the Genetic Code
- Genome-Wide Screens for Tinman Binding Sites Identify Cardiac Enhancers with Diverse Functional Architectures
- PLOS Genetics
- Archiv čísel
- Aktuální číslo
- Informace o časopisu
Nejčtenější v tomto čísle- Function and Regulation of , a Gene Implicated in Autism and Human Evolution
- An Insertion in 5′ Flanking Region of Causes Blue Eggshell in the Chicken
- Comprehensive Methylome Characterization of and at Single-Base Resolution
- Susceptibility Loci Associated with Specific and Shared Subtypes of Lymphoid Malignancies
Kurzy
Zvyšte si kvalifikaci online z pohodlí domova
Současné možnosti léčby obezity
nový kurzAutoři: MUDr. Martin Hrubý
Všechny kurzyPřihlášení#ADS_BOTTOM_SCRIPTS#Zapomenuté hesloZadejte e-mailovou adresu, se kterou jste vytvářel(a) účet, budou Vám na ni zaslány informace k nastavení nového hesla.
- Vzdělávání



