-
Články
Top novinky
Reklama- Vzdělávání
- Časopisy
Top články
Nové číslo
- Témata
Top novinky
Reklama- Videa
- Podcasty
Nové podcasty
Reklama- Kariéra
Doporučené pozice
Reklama- Praxe
Top novinky
ReklamaTranscriptomic Profiling of Reveals Reprogramming of the Crp Regulon by Temperature and Uncovers Crp as a Master Regulator of Small RNAs
Many bacterial pathogens cycle between environmental sources and mammalian hosts. Adaptation to the different natural habitats and host niches is achieved through complex regulatory networks which adjust synthesis of the large repertoire of crucial virulence factors and fitness determinants. To uncover underlying control circuits, we determined the first in-depth single-nucleotide resolution transcriptome of Yersinia. This revealed important novel genetic information, such as global locations of transcriptional start sites, non-coding RNAs, potential riboswitches and provided a set of virulence-relevant expression profiles, which constitute a valuable tool for the research community. The analysis further uncovered a temperature-induced global reprogramming of central metabolic functions, likely to support intestinal colonization of the pathogen. This is accompanied by a major reorganization of the CRP regulon, which involves a multitude of regulatory RNAs. The primary consequence is a fine-tuned, coordinated control of metabolism and virulence through a plethora of environmentally controlled regulatory RNAs allowing rapid adaptation and high flexibility during life-style changes.
Published in the journal: . PLoS Genet 11(3): e32767. doi:10.1371/journal.pgen.1005087
Category: Research Article
doi: https://doi.org/10.1371/journal.pgen.1005087Summary
Many bacterial pathogens cycle between environmental sources and mammalian hosts. Adaptation to the different natural habitats and host niches is achieved through complex regulatory networks which adjust synthesis of the large repertoire of crucial virulence factors and fitness determinants. To uncover underlying control circuits, we determined the first in-depth single-nucleotide resolution transcriptome of Yersinia. This revealed important novel genetic information, such as global locations of transcriptional start sites, non-coding RNAs, potential riboswitches and provided a set of virulence-relevant expression profiles, which constitute a valuable tool for the research community. The analysis further uncovered a temperature-induced global reprogramming of central metabolic functions, likely to support intestinal colonization of the pathogen. This is accompanied by a major reorganization of the CRP regulon, which involves a multitude of regulatory RNAs. The primary consequence is a fine-tuned, coordinated control of metabolism and virulence through a plethora of environmentally controlled regulatory RNAs allowing rapid adaptation and high flexibility during life-style changes.
Introduction
Pathogenic bacteria have evolved a wide range of regulatory mechanisms to adjust their virulence strategies and biological fitness in response to fluctuating environmental conditions during the infection process. Expression of virulence-associated determinants is strictly and coordinately controlled by a set of transcriptional regulators and complex signal transduction cascades are responsible for sensing and integrating the environmental information into the virulence regulons [1–4]. In addition to the transcriptional control there has been increasing recognition that post-transcriptional mechanisms are used to fine-tune the management of virulence determinants and achieve a rapid and more refined control of the bacterial response [5–9].
Post-transcriptional regulation can be accomplished by (i) RNA thermosensors and riboswitches, i.e. regulatory elements of an mRNA, that undergo a structural alteration upon a thermal shift or binding of a small molecule [10–14], and (ii) antisense and trans-encoded small regulatory RNAs of which a subset bind to their target mRNAs by the help of the RNA chaperone Hfq [15]. The regulatory outcomes generally include alterations of the translation initiation efficiency or mRNA stability. In recent years a plethora of sensory and regulatory RNA elements has been identified in many human pathogens, including the genus Yersinia [16–20], and there is increasing evidence that they serve as crucial players in regulatory circuits adjusting cellular physiology, metabolism and virulence [6,21].
Yersinia pseudotuberculosis is a common foodborne pathogen that infects wild and domestic animals, as well as humans [22]. It is a very closely related ancestor of Y. pestis, the causative agent of plague, which has evolved from Y. pseudotuberculosis about 1,500 to 20,000 years ago [23,24]. Although both pathogens are genetically very similar (>97% nucleotide identity over 75% of the protein-coding genes), they differ significantly in their pathogenesis and exhibit very different infection and disease patterns [25,26]. In contrast to Y. pestis, Y. pseudotuberculosis causes a range of mild gut-associated diseases such as enteritis, watery diarrhea and mesenterial lymphadenitis, called yersiniosis. The intestinal diseases are usually self-limiting, but in rare cases Y. pseudotuberculosis can also trigger autoimmune responses [27,28].
Unlike Y. pestis, which typically colonizes fleas, Y. pseudotuberculosis is able to survive for considerable time periods in the soil and other environmental reservoirs [29]. Based on the distinct phases of their life-style, it is not surprising that sudden temperature and nutrient changes experienced upon entry from external/vector reservoirs into a warm-blooded host are the most important signals for Y. pestis and Y. pseudotuberculosis to trigger virulence gene expression and adjust their host survival program [30]. Thermal/nutrient shifts influence expression of multiple virulence-associated processes of Yersinia, e.g. host cell interaction, intracellular persistence, host immune defense, motility, and host-adapted metabolism. Recent work on individual virulence factors has illustrated that this is achieved by a joint interplay of transcriptional and post-transcriptional mechanisms, including cis-acting RNA elements and non-coding RNAs, that allow a more refined management of the bacterial response [6]. In this study, we used transcriptional profiling by RNA-seq to determine the transcriptional landscape of Y. pseudotuberculosis YPIII, identify thermal and nutrient control mechanisms on a global level, and elucidate the magnitude and regulatory architecture of the CRP regulon linking nutritional status to virulence. The CRP protein, a crucial global transcriptional regulator that interacts with cAMP and controls a plethora of genes in Enterobacteriaceae in response to the supply of glucose or other efficiently utilizable sugars [31,32], was previously shown to play a crucial role for the pathogenicity of Yersinia [33–36]. CRP modulates a large subset of virulence-relevant genes [34,37–39], and comparative metabolome and fluxome studies further revealed that absence of CRP strongly perturbs the carbon core metabolism at the level of the pyruvate-tricarboxylic acid cycle (TCA) node [34,39]. Transcriptional profiling of Y. pseudotuberculosis in this study further revealed a comprehensive remodelling of the CRP-controlled network in response to temperature and uncovered CRP as transcriptional master regulator of regulatory RNAs.
Results and Discussion
Differential and comparative RNA-seq of Y. pseudotuberculosis YPIII
Y. pseudotuberculosis strain YPIII is a widely distributed virulent isolate which played an important role for the analysis of Yersinia infection [34,35,40–45]. Many of its virulence factors have been characterized in detail and our knowledge of virulence-relevant gene regulation and networks was mainly derived from this strain. In order to obtain a comprehensive image of the transcriptome, we used rRNA-depleted total RNA of YPIII grown to exponential or stationary phase at 25°C or 37°C resembling alterations in temperatures and nutrient limitations encountered in the different life-styles.
To produce a detailed map and identify active transcriptional start sites (TSSs) at a single nucleotide resolution, we employed a global differential RNA-seq approach by comparing sequence reads from different strand-specific barcoded cDNA libraries [46]. Libraries denoted with +TAP were generated from RNA pools treated with tobacco acid pyrophosphatase (TAP) to allow 5’ adapter ligation to primary transcripts, and-TAP labelled libraries were generated from RNA pools, which were not treated with TAP (S1 Dataset; S1 Fig), similar to what has been described previously [47,48]. From each library between 2.3–12.9 million cDNA reads were generated and mapped to the YPIII genome sequence (NC_010465) and the pYV virulence plasmid (NC_006153).
Our RNA-seq approach confirmed earlier findings that expression of the virulence plasmid pYV-encoded genes is induced at 37°C [30,45,49–52], relative to the expression of the chromosome. In this context, enhanced expression of pYV at 37°C is pronounced for stationary phase cultures (Fig. 1A; S1 Dataset). In addition, we observed that the ratio between the intergenic region (IGR) and mRNA reads was considerably higher during stationary phase compared to exponential phase (Fig. 1B), indicating a pronounced expression of trans-encoded regulatory RNAs under nutrient-deprived growth conditions. Along with this observation we found that the intracellular level of Hfq, an RNA chaperone, which is often involved in the interaction of trans-encoded sRNAs with their target mRNAs [53], is substantially increased under nutrient starvation (Fig. 1C). This differs from Escherichia coli in which the abundance of Hfq varies only slightly and is maximal during log phase [54].
Fig. 1. Global cDNA read count distribution and Hfq levels in Y. pseudotuberculosis during growth under environmental and infection-relevant conditions. 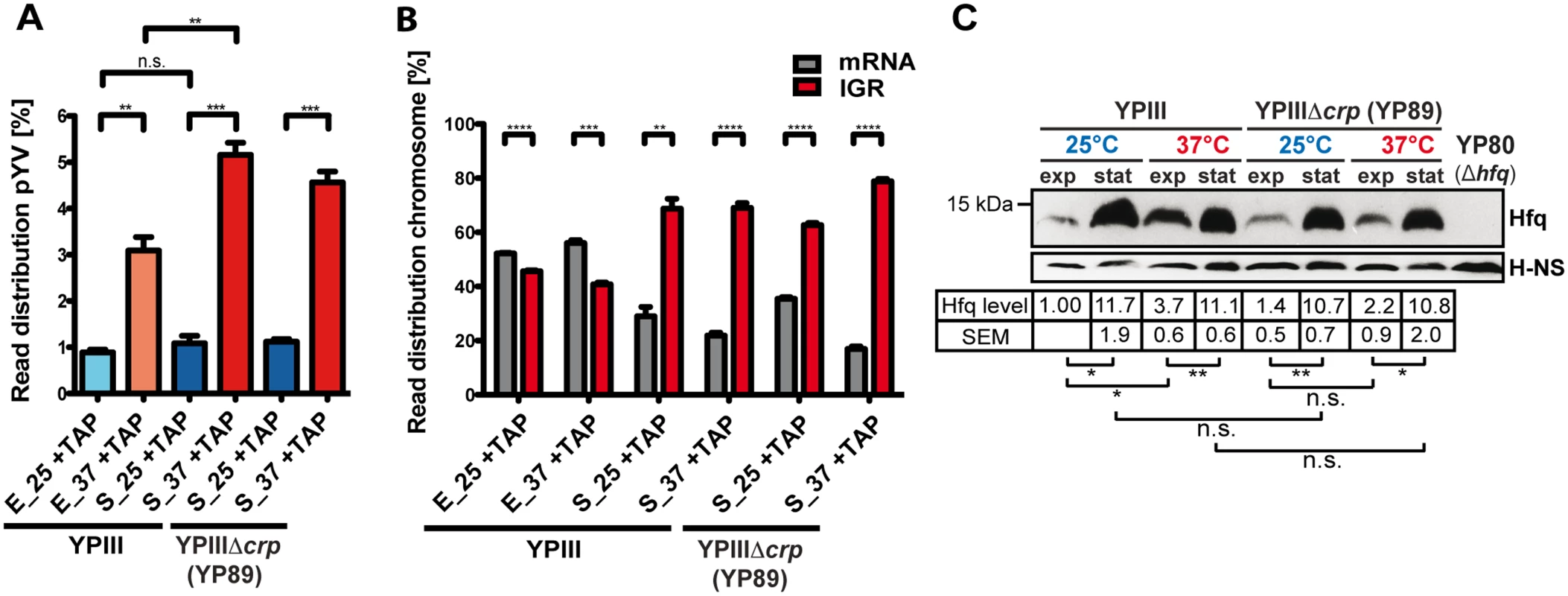
(A) Percentage of uniquely mapped reads of the virulence plasmid pYV obtained by RNA-seq of Y. pseudotuberculosis YPIII and YPIIIΔcrp cDNA libraries. Libraries were generated from TAP-treated (+TAP) rRNA depleted RNA obtained from cultures grown to exponential (E) or stationary (S) growth phase at 25°C (25) and 37°C (37) (for detailed statistics see S1 Dataset). The data represent the mean ± SEM from three independent biological replicates and were analyzed with Student’s t-test. **: P<0,01; ***: P<0,001; n.s.: not significant. (B) Percentage of uniquely mapped reads to annotated ORFs (mRNA) or intergenic regions (IGR) of the YPIII genome. IGR reads do not include 5’-UTRs of mRNAs, rRNAs and tRNAs, and mostly represent trans-encoded sRNAs. The data represent the mean ± SEM from three independent biological replicates and were analyzed with Student’s t-test. **: P<0,01; ***: P<0,001; ****: P<0.0001. (C) Growth phase- and temperature-responsive expression of Hfq in Y. pseudotuberculosis YPIII and the isogenic crp deletion strain YP89. Equal amounts of whole cell extracts of YPIII or YP89 grown to exponential (exp) or stationary phase (stat) at 25°C or 37°C were separated by SDS-PAGE prior to Western blotting using polyclonal Hfq or H-NS antibodies (loading control). As a negative control YP80 (YPIII Δhfq) was included. Relative protein amounts were quantified using ImageJ. The data represent the mean ± SEM from three independent experiments and were analyzed with Student’s t-test. *: P<0,05; **: P<0,01; n.s.: not significant. Genome-wide analysis of TSSs
Annotation and comparison of the 5’-ends in the (+) vs (-) TAP cDNA libraries revealed a redistribution of the genome coverage profile towards an elevated sharp-edged 5’ flank, as illustrated for rovA, hfq, katY and crp (Fig. 2A). By a bioinformatic approach and other criteria (see Materials and Methods), we were able to identify 1151 individual TSSs in proximity to annotated open reading frames (ORFs) under all tested growth conditions, out of which 24 were mapped on the pYV virulence plasmid (S2 Dataset). In total, for 815 ORFs a single TSS was mapped, while 155 ORFs harbor at least one alternative TSS. The highest number of alternative TSSs was identified for rpoD and gapA for which transcription occurs from at least 5 different start sites (S2 Dataset).
Fig. 2. Global identification of mRNA transcriptional start sites (TSSs). 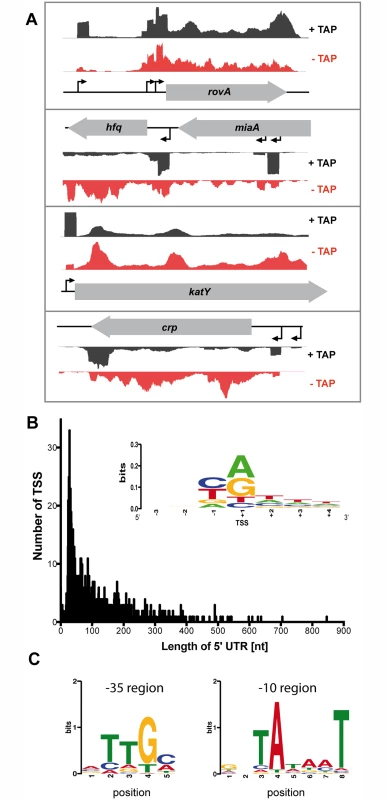
(A) Visualization of RNA-seq (+/- TAP) based cDNA sequencing reads mapped to the YPIII rovA, hfq, katY and crp gene locus using the Artemis genome browser (Release 15.0.0). (B) The 5’-UTR repertoire. The distribution and frequency of the length of 5’-UTRs is given for all mRNAs of YPIII which start upstream of the annotated translational start site. More than 40% of all 5’-UTRs are 20–60 nt in length. (Inset) Sequence conservation at the TSSs. Sequence logo computed from 1151 unaligned TSS regions (TSS is located at position +1) showing nucleotide conservation around the TSSs. The initial nucleotide of transcripts (position +1) is dominated by purines while position -1 is dominated by pyrimidines. (C) Detected conserved sequence motifs in the -35 and -10 promotor region. To validate the TSSs, we compared predicted TSSs of the RNA-seq analysis with TSSs previously published for genes/operons of Y. pseudotuberculosis or close relatives and found that the vast majority of the identified TSSs start within 2 nt or are identical (S2 Dataset). For example, the identified TSS of katY was identical with the katY TSS identified in Y. pestis [55], and the two proximal TSSs of rovA matched perfectly with published start sites [56] (Fig. 2A; S2 Dataset). Interestingly, an additional rovA TSS was mapped in our approach, which is in perfect agreement with a third more proximal TSS identified within the rovA regulatory upstream region in Y. enterocolitica [57]. For hfq three individual TSSs were identified (Fig. 2A; S2 Dataset). Two TSSs reside within the coding region of the upstream gene miaA, similar to hfq from E. coli [58]. The most proximal hfq TSS at position -90 relative to the annotated hfq translational start site is in perfect agreement with a TSS recently determined for hfq of Y. pestis and Y. pseudotuberculosis [59], yet the TSS with the highest accumulation of cDNA reads under all tested conditions is the most distant one at position -509 (Fig. 2A; S2 Dataset). We further validated newly identified Y. pseudotuberculosis TSSs by 5’ RACE and found that 14 of the identified 16 TSSs in the RNA-seq approach matched with high accuracy (S2 Dataset).
To further strengthen our genome-wide analysis of TSSs, we investigated the nucleotide preference from position -3 to +4 surrounding the identified Y. pseudotuberculosis TSSs and generated a sequence logo using the WebLogo software (Fig. 2B) [60]. Our analysis revealed a significant dinucleotide preference at positions -1 and +1 while adjacent positions did not show any nucleotide preference. Similar to what has previously been reported for E. coli, Salmonella enterica serovar Typhimurium and Klebsiella pneumoniae [61,62], the nucleotide at position -1 is dominated by a pyrimidine base (37.01% C, 35.01% T), and transcription is preferentially initiated at a purine base (position +1; 42.75% A, 35.27% G) (Fig. 2B).
We further used our global TSS map to determine a motif for the -10 and -35 regions of Y. pseudotuberculosis promoters (Fig. 2C). To compute conserved sequence motifs, we performed de-novo motif discovery within the -10 region (position -15 to -3) and the -35 region (position -45 to -25) using MEME [63]. The detected conserved sequence motifs in the -35 (TTGc/a) and -10 promotor region (TAtaaT) are highly similar to those identified in other related Enterobacteriaceae [61,62].
The 5’-UTR repertoire of Y. pseudotuberculosis
Our global TSS annotation further allowed us to define and analyze the untranslated regions (5’-UTRs) from the TSS to the start codon of the immediate downstream genes for putative cis-regulatory RNA elements, i.e. RNA riboswitches, known to control the translational efficiency of messengers [10–13]. Inspection of the 5’-UTRs of all 1151 defined transcriptional units of Y. pseudotuberculosis YPIII showed that the majority of 5’-UTRs is 20–40 nt in length (Fig. 2B; S2 Dataset) similar to other bacteria [64–66].
Notably, 155 genes are transcribed as mRNAs with 5’-UTRs longer than 200 nt, and a subset of 12 mRNAs had 5’-UTRs longer than 500 nt (S2 Dataset). Computational analysis using RibEx riboswitch explorer [67] predicted 4 known riboswitch-like elements (RLEs) among the long 5’-UTRs, e.g. cobalamin and FMN riboswitches, the yybP/ykoY element and the threonine operon leader (S2 Dataset). Moreover, a conserved RNA motif was predicted within the 217 nt 5’-UTR of moaA, encoding the molybdenum cofactor (Moco) biosynthesis protein A. Studies in E. coli demonstrated that the highly conserved RNA motif functions as Moco riboswitch [68]. RibEx predicted 17 additional RLEs; the respective candidates are mainly involved in metabolism and gene expression (S2 Dataset). For instance, the 5’-UTR of the corA mRNA of Y. pseudotuberculosis, encoding a magnesium/nickel/cobalt transporter (YPK_4002), harbors multiple RLEs. It likely represents a cation-responsive riboswitch similar to mgtA in Salmonella, encoding an Mg2+ transporter which is important for replication within macrophages [69,70].
We further report a small number of leaderless mRNAs with a 5’-UTR <10 nt (S2 Dataset). All 14 annotated leaderless mRNAs lack a classical ribosome-binding site and use an AUG as translation start codon, which was shown to be essential for stable ribosome-binding to leaderless transcripts [71]. Moreover, we identified 24 TSSs downstream of the annotated translational start site, indicating incorrect translational start site annotation. We compared the predicted sites with the start position of equivalent genes in other sequenced Y. pseudotuberculosis strains, including IP32953 and IP31758, and Y. pestis CO92 (S2 Dataset). For most of these genes, the annotated translational start site is located downstream of the translational start site given for YPIII in at least one of the other Yersinia genomes. One prominent example includes the virulence plasmid-encoded gene pYV0047 for the effector protein YopM. The annotation of the orthologous yopM gene on the PB1/+ virulence plasmid indicates a start codon that is located several nucleotides downstream of the TSS annotated for pYV (S2 Dataset). A putative ribosomal binding site (5’-AGGCA-3’) is located 6 nt upstream of the yopM translational start site annotated for the PB1/+ virulence plasmid, but it is lacking upstream of the translational start site annotated on pYV.
The repertoire of Y. pseudotuberculosis non-coding RNAs
Several trans-encoded small RNAs (sRNAs) of different Yersinia strains have been recently discovered by deep sequencing of cDNA libraries enriched for small transcripts [16–20]. Our analysis of the Y. pseudotuberculosis YPIII transcriptome now allows us to investigate expression of sRNAs in relation to other transcripts under the different growth conditions. For identification of candidate regulatory RNAs a conservative strategy was used which defines non-coding RNAs as generally small transcripts (>40 nt and <500 nt) expressed from intergenic regions or the antisense strand of defined mRNAs. Besides the stable tRNAs and rRNAs also the predicted transfer messenger RNA (tmRNA, SsrA), RNase P (M1 RNA) and signal recognition particle (SRP RNA) were found to be expressed (S3 Dataset). We further identified 78 putative trans-encoded RNAs (S3 Dataset) and 80 putative antisense RNAs (S3 Dataset). All trans-encoded sRNA candidates are encoded on the YPIII chromosome. Conspicuously, 19 putative antisense RNAs are encoded on the virulence plasmid, a rather dense population compared to the low number of antisense RNAs identified for the YPIII chromosome. A comparison between trans-encoded sRNAs identified in this and previous studies [16–20] revealed a surprisingly low overlap of 38 sRNAs. We identified 42 new putative trans-encoded sRNAs for Y. pseudotuberculosis YPIII (S3 Dataset), whereas 143 previously described sRNAs were not detectable. This discrepancy is likely to be caused by low expression under the tested growth conditions, strain differences, RNA/library preparation protocols as well as bioinformatic criteria and pipelines applied for the RNA-seq approaches. Notably, several of the previously reported sRNAs were also classified as 5’-UTRs in our study, e.g. sR028, sR041, sR050, sR066 and sR070 which were identified in very close proximity to adjacent ORFs [20] (S2 Dataset). Potentially, those putative sRNAs are expressed as a premature transcript and result from processing of the 5’-UTR. This might account at least for sR066, for which a distinct short transcript was detected by Northern blotting [20]. In total, 36 of the non-coding RNA candidates were confirmed by Northern blotting (Figs. 3A, S2). This included 12 conserved sRNAs previously identified in other Enterobacteriaceae, 2 non-validated sRNAs (Ysr100 and Ysr103) and 1 validated sRNA (Ysr164) also identified in sRNA libraries of Y. pestis Kim6+ and Y. pseudotuberculosis IP32953 [16,18], 11 newly identified sRNAs and 10 antisense RNAs. Some of the validated antisense RNAs were identified as trans-encoded RNAs in Y. pseudotuberculosis IP32953 (Ysr93, Ysr15, Ysr114) [16]. Ysr15 and Ysr93 were grouped as antisense RNAs in this study, as they are encoded antisense to genes of hypothetical proteins of YPIII, which are not annotated in the IP32953 genome, and Ysr114 was found to largely overlap with the 3’-coding region of trxB (YPK_2687). How these antisense RNAs exert their function remains to be shown. For instance, the non-coding RNA McaS of E. coli, which regulates motility and biofilm formation is trans-acting, although it is encoded antisense to an annotated ORF [72].
Fig. 3. Temperature- and growth phase-responsive protein-encoding genes and trans-encoded RNAs. 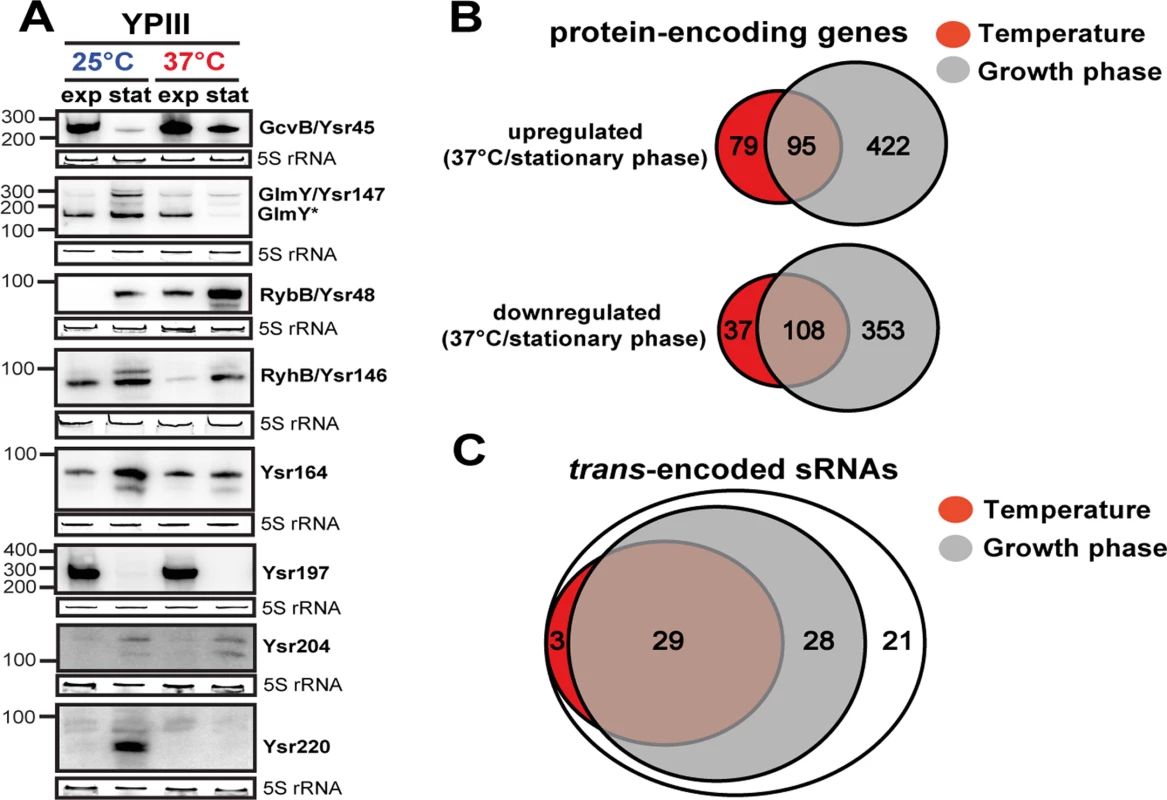
(A) Differential expression of trans-encoded sRNAs of YPIII shown by Northern blot analyses. RNA samples were prepared from bacteria grown to exponential (exp) or stationary phase (stat) at 25°C and 37°C. sRNAs were detected by specific radioactive labeled probes. 5S rRNA served as loading control. The size marker (nt) is indicated. Venn diagrams illustrating temperature and growth phase-regulated (B) protein-encoding genes and (C) trans-encoded sRNAs in Y. pseudotuberculosis YPIII. The regulons were obtained by comparative RNA-seq using DESeq from triplicate experiments (S3 and S4 Datasets). Protein- and trans-encoded RNAs which are differentially regulated by at least 4-fold (p-value ≤0.05) are included. The temperature and growth phase-responsive regulons
To improve our understanding of the complexity of the regulatory networks controlling the multiple life-styles of yersiniae, we used our RNA-seq approach to identify coordinately regulated genes in response to temperature and nutrient limitation. Comparative RNA-seq analysis using DESeq [73] from triplicate experiments revealed a total of 324 ORFs of YPIII that were differentially regulated by at least 4-fold (p-value ≤0.05) in response to temperature (Fig. 3B; S4 Dataset). Growth phase had an even higher impact on global gene expression. Nearly 25% of the genome (983 genes) was differentially expressed (Fig. 3B; S4 Dataset). In accordance with previous data [30,45,52,74–76], many of the temperature-regulated genes encode well-known (i) regulators, effectors or components of the T3SS machinery on virulence plasmid pYV (of note, the components displayed a wide range of thermoregulation, of which the lcrGVHyopBD, ypkA, yopE and yopH transcripts were highly abundant even at 25°C), (ii) classical pathogenicity factors such as the adhesins AilA, PsaA, InvA, and the CNFY toxin, or (iii) other virulence-associated traits, e.g. urease, iron sequestration systems, motility/chemotaxis machinery and genes related to oxidative stress. The majority of the virulence-associated traits is also subject to growth phase control (S4 Dataset). This overlap is not solely restricted to the virulence-related attributes. Remarkably, 47% of the temperature-regulated genes encode metabolic functions, mainly enzymes of the carbon/energy metabolism, of which most are also subject to growth phase control (S4 Dataset). This observation and validity of our approach was confirmed by the strong correlation between comparative RNA-seq and qRT-PCR data of 24 randomly selected temperature - and/or growth phase-regulated genes encoding hypothetical proteins and proteins implicated in metabolism and virulence-relevant traits (S3 Fig). In summary, this indicated a clear link between both regulons in the control of Yersinia virulence.
Among the metabolic genes which are significantly upregulated at 37°C are genes encoding numerous sugar and amino acid transporters, enzymes involved in the catabolism of various carbohydrates and amino acids, β-oxidation of fatty acids, and energy production/conversion, indicating that terminal oxidation of many different substrates is favored at 37°C to allow rapid proliferation within the host (Fig. 4; S4 Dataset). Concomitant changes in oxidative catabolism, implicating a subset of these genes, were also found during temperature transition of the conditionally virulent Y. pestis strain KIM5 [77]. Most likely, these metabolic functions are part of the core regulon of thermoregulated genes in Yersinia.
Fig. 4. Temperature-dependent reprogramming of the primary metabolism of Y. pseudotuberculosis. 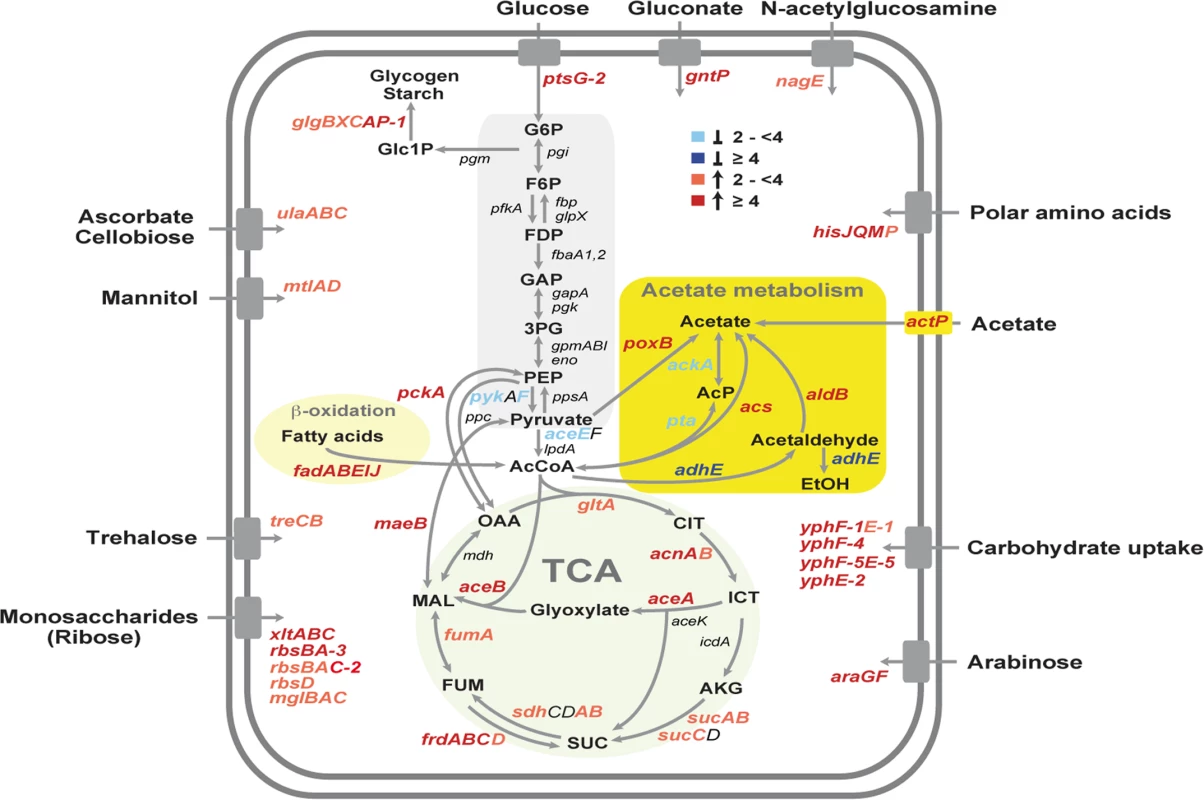
Glycolysis, acetate metabolism, β-oxidation of fatty acids, the TCA cycle and transport systems are illustrated and the genes, designated according to the annotated YPIII genome, which encode equivalent enzymes of the presented pathways are given. Genes which are differentially expressed in response to temperature are colored. The blue color shows genes induced at 25°C (≥4, dark blue, ≥2 light blue), and the red color represents genes upregulated at 37°C (≥4, dark red, ≥2 light red). The values of the expression changes are listed in S4 Dataset. One other striking observation which has not been observed in Y. pestis, is the existence of a thermoregulated ‘acetate switch’ (Fig. 4). This physiological switch occurs when bacteria transit from a program of rapid growth that produces acetate from acetogenic carbon sources, e.g. glucose, serine (dissimilation), to a program of slower growth facilitated by the import and utilization (assimilation) of acetate [78]. Similar to E. coli grown on high concentration of acetate, Y. pseudotuberculosis upregulates genes encoding enzymes with assimilation functions (PoxB, ActP, Acs), the glyoxylate bypass, the TCA cycle and gluconeogenesis (MaeB, PckA) at 37°C, while genes encoding PykF, AceEF, the Pta-AckA pathway and AdhE, converting acetyl-CoA to acetate or ethanol, were repressed (Fig. 4; S4 Dataset). A stronger induction of the acetate switch and the glyoxylate pathway was also observed under nutrient limiting condition during stationary phase, although expression of multiple enzymes of the TCA cycle remained unchanged (S4 Dataset). To support this observation, we compared the intracellular amount of two key metabolites of the acetate switch, acetate and acetyl-CoA, in Y. pseudotuberculosis grown at 25°C or 37°C, and found that the level of both metabolites was significantly reduced during growth at 37°C (S4 Fig).
Why flipping the switch in response to temperature? One reason could be that bacteria are primed for growth in the intestine. The mammalian intestinal tract is rich in short-chain fatty acids, of which acetate predominates. These volatile fatty acids are produced by the intestinal microbiota through consumption of available polysaccharides from diet or the host-secreted mucus and could constitute an important carbon/energy source for Y. pseudotuberculosis within the digestive tract [78,79]. Temperature-induced reprogramming to facilitate utilization of polysaccharide-derived degradation products is further supported by the fact that expression of several transport and catabolic genes of simple sugars are also thermally induced (Fig. 4). It is also possible that the acetate metabolism of Yersinia is influenced by the low availability of oxygen in the intestine. Studies with E. coli have shown that the absence of oxygen inhibits the expression of many TCA cycle enzymes which affects the pyruvate-acetate-acetyl-CoA flow [78].
Our global and quantitative RNA-seq approach further revealed a strikingly large number of growth phase - and/or temperature responsive trans-encoded sRNAs (74%; Fig. 3A, C; S3 and S4 Datasets). The vast majority (86%) of the growth phase-controlled sRNAs are present at higher abundance during stationary phase of which about half also respond to temperature (Fig. 3A, C; S3 Dataset). Among them are most conserved sRNAs, e.g. RybB and many novel sRNAs, e.g. Ysr220. However, there are also prominent exceptions to this trend, e.g. GcvB and Ysr197, which are strongly repressed during stationary phase (Fig. 3A; S3 Dataset). The large number of temperature - and growth phase-responsive sRNAs is likely to contribute an important additional control layer to adjust and fine-tune the pathogen’s fitness and virulence in the response to the dramatic changes associated with the transmission of yersiniae between heterothermic environments and homeothermic mammals. The physiological relevance of these sRNAs for pathogenesis is further supported by a previous study, reporting the importance of the RNA chaperone Hfq for the virulence of Y. pseudotuberculosis [80].
Reprogramming of the CRP regulon by temperature
One crucial global regulator previously shown to link the nutritional status of Yersinia to virulence is CRP. CRP controls the transcription of multiple genes and operons in Enterobacteriaceae in response to certain carbon sources (in particular simple sugars), which are scarse during stationary phase. Loss of CRP was found to affect transcription of many genes involved in the primary metabolism and pathogenicity in Y. pestis, Y. enterocolitica and Y. pseudotuberculosis and led to a strong attenuation of their virulence [33,34,36–38,81]. This tight link between catabolite repression and virulence motivated us to include a crp deficient mutant of Y. pseudotuberculosis YPIII (YP89) into our transcriptomic approach to (i) compare the influence of CRP at temperatures mimicking environmental/vector or mammalian host niches, and (ii) address global impact of CRP on the expression of sRNAs.
Our analysis revealed 741 CRP-dependent genes (S5 Dataset) of which 228 were previously identified by a microarray approach (62% overlap) which addressed expression solely at 25°C during late stationary phase [34]. Strikingly, only a small proportion of genes (142 genes, 19%) is shared between the CRP regulons determined at 25°C or 37°C, as exemplified by the acs and mdh genes, while the majority varies significantly. For instance, aceBAK transcript abundance is negatively affected by CRP at 37°C, but not at 25°C (S5 Dataset; S5 Fig). This is also reflected by the finding that the composition of temperature-regulated genes is dramatically altered in the crp mutant. Loss of crp had only a minor influence on the expression of the pYV-encoded virulence genes, whereas the majority of the temperature-dependent chromosomal genes (191 genes, 75%), including numerous metabolic, virulence and stress adaptation genes, did not respond to temperature in the absence of CRP (S6 Dataset). In contrast, 247 genes are solely temperature-regulated in the crp deletion strain, the majority of which is negatively affected by CRP. In summary, our data reveal a profound reprogramming of the CRP regulon by temperature (S6 Dataset). How this is accomplished is unclear, but the large number of transcriptional regulators which are members of the two distinct CRP regulons (S5 Dataset; S5 Fig) indicates that the switch is supported by CRP-dependent, thermo-controlled regulators such as RovA [34,82,83]. This global virulence regulator is only expressed and positively controlled by CRP at 25°C, but not at 37°C due to the fact that RovA is a protein thermometer which is partially defolded and rapidly degraded at 37°C [82].
CRP, a master regulator of sRNAs in Y. pseudotuberculosis
The accumulation of regulatory RNAs during stationary phase suggested that they might also be part of the CRP regulon. Our investigation of transcriptomic changes revealed 53 trans-encoded RNAs of Y. pseudotuberculosis (65%) which are up - or down-regulated in the absence of crp at 25°C and/or 37°C (S3 and S5 Datasets). Many conserved trans-encoded RNAs, but also multiple newly identified sRNAs were identified as members of the CRP regulon, and their intracellular level is strongly affected by temperature. CRP-dependent regulation of all 15 randomly selected trans-encoded sRNAs was confirmed by Northern blotting (Fig. 5A). Furthermore, we validated one newly identified trans-encoded RNA Ysr206, as well as two antisense RNAs Ysr232 and Ysr114, which were exclusively detected in the crp mutant (Fig. 5A). Both antisense RNAs constitute the most abundant antisense RNAs identified by our approach (S3 Dataset). The huge subset of CRP-regulated non-coding RNAs strongly indicates that CRP is a master regulator of regulatory RNAs in Y. pseudotuberculosis. Recent identification of four CRP-dependent riboregulators in Y. pestis (CyaR, sR065, sR066, sR084 encoded on pPCP [20]) further indicate that this might also account for other Yersinia species. Furthermore, CRP-mediated control seems to account for the accumulation of many sRNAs during stationary phase, and it also explains downregulation of a subset of sRNAs (e.g. GcvB), which are negatively affected by the regulator (Figs. 3A, 5A).
Fig. 5. CRP-dependent non-coding RNAs of Y. pseudotuberculosis. 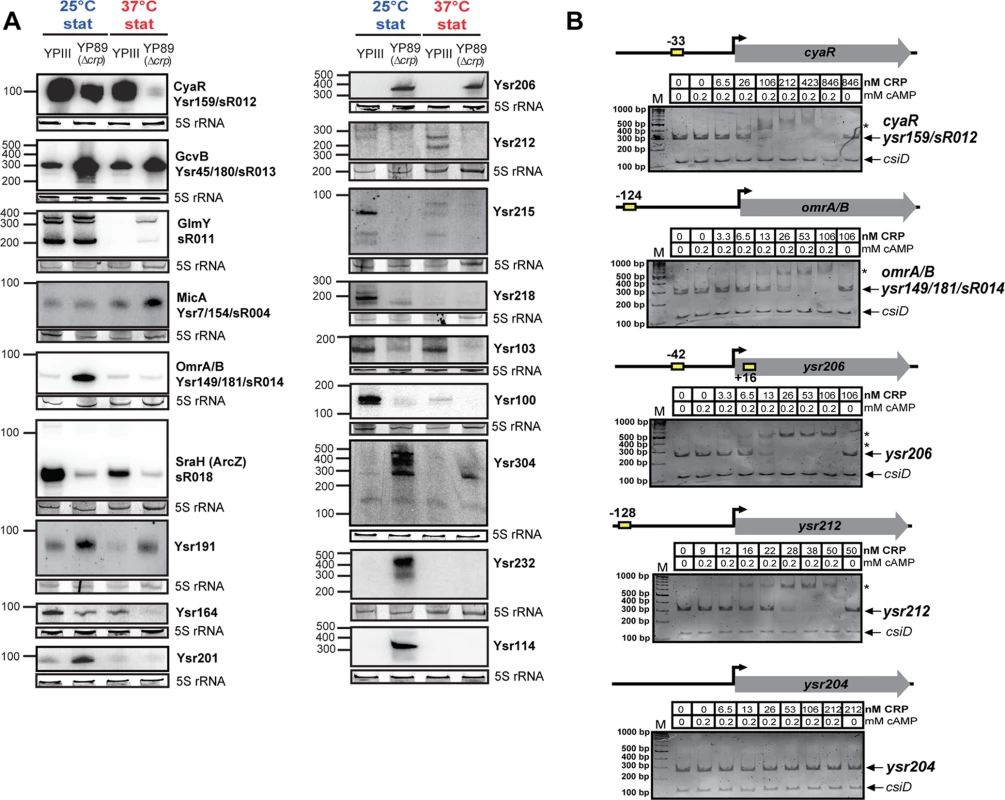
(A) Northern blot analyses of selected CRP-regulated non-coding RNAs. RNA samples were prepared from YPIII and YP89 (YPIII Δcrp) bacteria grown to stationary growth phase (stat) at 25°C and 37°C. 5S rRNA served as loading control. The size marker (nt) is indicated. (B) Interaction of CRP with the regulatory regions of selected CRP-regulated sRNA genes. Individual DNA fragments with the predicted CRP-binding site(s) (yellow boxes; S3 Dataset) used for electrophoretic mobility shift assays are illustrated. An individual sRNA promoter fragment (ysr204) for which no CRP-binding site was predicted was included as negative control. Respective DNA fragments were incubated with increasing concentrations of CRP and 0.2 mM cAMP. As a negative control, cAMP was omitted in samples with the highest CRP concentration (right lane). The CRP-DNA complexes were separated on 4% polyacrylamide gels. The position of specific higher molecular weight complexes is marked with an asterisk. A molecular weight standard (M) was loaded, and the corresponding molecular weights are indicated. A csiD PCR fragment amplified from E. coli served as an internal negative control. CRP interacts with the regulatory upstream region of multiple Yersinia sRNAs
A manual search and a string-based prediction of CRP binding motifs (TGTGA-N6-TCACA) within the regulatory region of all 54 CRP-dependent sRNA genes identified one or more putative CRP binding sites within 18 of these sRNA genes (S3 Dataset). Direct and specific CRP binding in a cAMP-dependent manner was found for cyaR, omrA, sraH, Ysr100, 191, 206, 212, 215, 218, 226, and a manually identified sRNA, Ysr304 (Figs. 5B, S6). Cooperative interactions were observed with the regulatory region of Ysr206 and Ysr226, for which more than one CRP binding site was predicted (Figs. 5B, S6; S3 Dataset). The validity of our data was reinforced by the fact that CRP did not interact with the regulatory regions of ysr204, rybB and ysr232 for which no CRP binding site was predicted (Figs. 5B, S6). To confirm these results we also selected two CRP-dependent promoter regions of sRNA genes (sR018 and ysr212), which have previously been identified to be CRP-dependent (Figs. 5B, S6; S3 Dataset) and performed DNase I footprinting experiments. Incubation of the fragments with increasing CRP concentrations revealed that CRP binds specifically to the promoter regions at the predicted CRP-binding sites (S7 Fig). This demonstrates that a large proportion of the Yersinia sRNAs are under direct control of CRP. Catabolite repression of the remaining sRNAs might be mediated indirectly by other transcriptional regulators (e.g. RpoN for GlmY [84], and RpoE for RybB [85,86] identified as part of the CRP regulon; S5 Dataset; S5 Fig).
Regulation of CRP in response to growth phase and temperature
The tight link between catabolite repression and thermal control in Yersinia, in particular when one considers the expression of non-coding RNAs, suggested that synthesis and/or activity of CRP itself might be subjected to thermal and growth phase control. Indeed, we found that expression of the cAMP-producing adenylate cyclase gene cyaA was 2.8-fold increased during stationary phase (S4 Dataset). Moreover, CRP levels are strongly increased during stationary phase, whereby the amount of the global regulator is slightly but significantly higher at 25°C compared to 37°C (Fig. 6A). Interestingly, the crp transcript levels quantified by RNA-seq do not increase during transition from exponential to stationary growth (S4 Dataset), indicating that environmental control in response to growth phase occurs on the post-transcriptional level. Post-transcriptional control of CRP was also reported in Y. pestis CO92 in the context of the control of the plasminogen activator protease Pla [36]. In Y. pestis, loss of the RNA chaperone Hfq was found to decrease CRP levels. The molecular mechanism is not yet known, but it was found to involve the crp 5’-UTR [36]. We further tested whether loss of Hfq has also an influence on the amount of CRP in Y. pseudotuberculosis (Fig. 6B). However, no significant difference of CRP levels was detectable between the YPIII wild-type, the isogenic hfq mutant and the in trans-complemented hfq mutant during stationary phase at 25°C and 37°C (Fig. 6B). One the other hand, the intracellular level of Hfq, shown to be CRP-dependent in other related Enterobacteriaceae [87], remained unchanged in the absence of crp (Fig. 1C). Together, this demonstrated that CRP acts as a global sRNA regulator in Y. pseudotuberculosis YPIII in an Hfq-independent manner.
Fig. 6. CRP, a growth phase and temperature-responsive master regulator in Y. pseudotuberculosis. 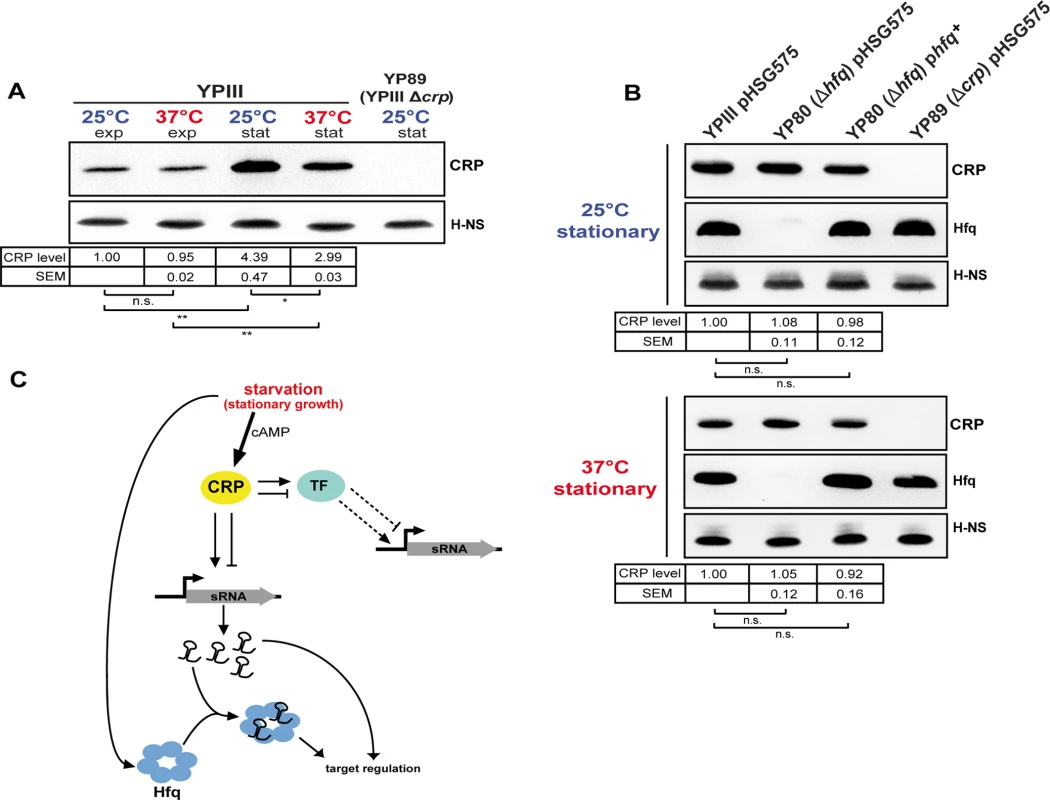
(A) CRP protein levels in response to growth phase and temperature. Equal amounts of whole cell extracts of YPIII grown to exponential (exp) or stationary phase (stat) at 25°C and 37°C were separated by SDS-PAGE prior to Western blotting using polyclonal CRP antibodies. H-NS served as loading control. YP89 (YPIII Δcrp) was included as negative control. Relative protein amounts were quantified using ImageJ. The data represent the mean ± SEM from three independent experiments analyzed with Student’s t-test. *: P<0,05; **: P<0,01; n.s.: not significant. (B) Effect of the RNA chaperone Hfq on CRP. Equal amounts of whole cell extracts of the wild-type strain YPIII, the isogenic hfq mutant (YP80) and the complemented hfq mutant (YPIII pBW131 (phfq+)) grown to stationary phase at 25°C and 37°C were separated by SDS-PAGE prior to Western blotting using polyclonal CRP or Hfq antibodies. Plasmid pHSG575 served as the empty vector control. YP89 (YPIII Δcrp) was included as negative control. H-NS served as loading control. Relative protein amounts were quantified using ImageJ. The data represent the mean ± SEM from three independent experiments analyzed with Student’s t-test. n.s.: not significant. (C) Current model of growth phase- and temperature-dependent sRNA expression involving the transcriptional regulator CRP and the RNA chaperone Hfq. During stationary growth when glucose is limiting intracellular CRP and cAMP levels increase, leading to accumulation of active CRP protein. Active CRP induces the expression of numerous sRNAs by direct binding to the regulatory region of the sRNA gene, thereby stimulating or repressing transcription. Further, expression of those sRNAs which are not directly regulated by CRP might involve one or more of the CRP-dependent transcription factors (TF), whose expression is differently affected by temperature. During stationary growth the RNA chaperone Hfq accumulates, similar to CRP, and affects sRNA abundance on the post-transcriptional level. Conclusions
Many years of research on the pathogenicity mechanisms of Yersinia provided great insights into the plethora of virulence and fitness factors required for the successful establishment of an infection. However, we are far from understanding the precise choreography and concerted action of transcriptional and post-transcriptional factors controlling Yersinia virulence. In this work, we successfully applied RNA-seq to obtain a valuable data resource that provides detailled information about the expression of the majority of genes of the enteric pathogen Y. pseudotuberculosis. The high-resolution transcriptome presented here constitutes the first single nucleotide resolution TSS map of a member of the genus Yersinia. In addition to creating a valuable tool for the Yersinia research community, it provides a framework for better analysis of identified sensory and regulatory non-coding RNAs and for studying the complex regulatory mechanisms and networks that allowed pathogenic yersiniae to become successful human pathogens. The complex expression and processing patterns show that many RNA-mediated regulations are yet to be discovered in Yersinia. The ongoing analysis of strain - and species-specific differences of these regulatory elements will further help us to identify how they manipulate the transcriptional output of the pathogens and contribute to phenotypic variations and/or specific adaptation to certain hosts.
We further obtained a detailed snapshot of the abundance of coding and non-coding transcripts and their sites of initiation taken at different virulence-relevant conditions. The comparative analysis of these data revealed global reprogramming of the transcriptional units controlled by CRP when growth conditions change from moderate environmental to human body temperatures, which appear to prime the bacteria for growth in the digestive tract. This includes a large set of catabolic and energy production genes that facilitate consumption of various carbon sources, e.g. dietary fibers-derived simple sugars and uncovered the existence of a thermo-controlled ‘acetate switch’ which may allow preferential growth on acetate, a predominant endproduct of the intestinal microbiota. This observation is supported by another study of our group, which demonstrated that absence of CRP perturbs in particular the fluxes of the carbon core metabolism at the level of the pyruvate-acetyl-CoA-TCA cycle node [88].
The thermally induced expression switch of virulence-relevant metabolic genes is most likely to be mediated by CRP-dependent transcriptional regulators and small non-coding RNAs (Fig. 6C). In fact, we found that CRP affects the abundance of more than 50% of all identified sRNAs, most of which are under thermal control and induced during stationary phase. Based on our data we postulate that the transcription factor CRP acts as master regulator of non-coding RNAs in Yersinia. The identified CRP-dependent sRNAs are likely to contribute to virulence and host adaptation by modulating metabolic pathways, which are important for efficient host colonization by Yersinia. Furthermore, use of non-coding RNAs to link the nutritional status of Yersinia to virulence may allow the pathogen to increase its energy budget upon nutrient limitation by replacing proteinaceous regulators by regulatory RNA elements. Considering the wide range of reservoirs, including the different niches in the human body, a global switching between regulons and regulatory elements seems advantageous to rapidly and efficiently readjust gene expression during transmission between the different challenging external reservoirs and host environments.
We anticipate that a comprehensive detailed analysis of the global impact of transcriptional regulators important for bacterial fitness and virulence, coupled with the identification of post-transcriptional control mechanisms by sensory and regulatory RNA elements during growth in environmental reservoirs, transmission vectors and during the infection of animal hosts and humans will herald a new exciting era for future research. This will not only help us to elucidate the complex regulatory network adjusting gene expression to the rapidly changing demands during the process of an infection, it will also allow us to identify crucial control mechanisms which can be further exploited as potential drug targets.
Materials and Methods
DNA manipulation and plasmid construction
All DNA manipulations, restriction digestions, ligations and transformations were performed using standard genetic and molecular techniques [89]. The plasmids used in this work are listed in S1 Table. Oligonucleotides used for PCR and sequencing were purchased from Metabion and are also listed in S1 Table. Plasmid DNA was isolated using Qiagen plasmid preparation kits. DNA-modifying enzymes and restriction enzymes were purchased from New England Biolabs. PCRs were performed in a 100 μl volume for 29 cycles using Phusion High-Fidelity DNA polymerase (New England Biolabs). Purification of PCR products was routinely performed using the QIAquick kit (Qiagen). The constructed plasmid was sequenced by the in-house facility. To generate plasmid pBW131, the entire hfq gene along with 847 nt upstream region and 42 nt downstream region was amplified by PCR using primers V75 and V80 and cloned into the BamHI site of pHSG575.
Bacterial strains and RNA isolation
Y. pseudotuberculosis YPIII and the isogenic crp mutant YP89 were grown in LB medium to exponential phase (OD600 0.5) and stationary phase (16 h) at 25°C and 37°C (S8 Fig). Total bacterial RNA was isolated by a hot phenol extraction protocol [89], DNA was digested using the TURBO DNase (Ambion), purified with phenol:chlorophorm:isopropanol and the quality was assessed using the Agilent RNA 6000 Nano Kit on the Agilent 2100 Bioanalyzer (Agilent Technologies) (S9 Fig). From 8 μg of total RNA the rRNA was depleted using MICROBExpress (Ambion). 1 μg of rRNA depleted RNA was treated with TAP for +TAP libraries (Epicentre Biotechnologies) (S1 Fig).
Strand-specific library preparation and Illumina sequencing
Strand-specific RNA-seq cDNA library preparation and barcode introduction was based on RNA adapter ligation as described previously [90], omitting double strand specific nuclease normalization (DSN). In brief, the rRNA-depleted +/-TAP treated RNA was fragmented by sonication to a median size of 200 nt. The fragments were 5’-phosphorylated and ligated to 3’ - and 5’-RNA-adapter oligonucleotides (S1 Fig). After reverse transcription, cDNA libraries were PCR amplified (15 cycles). Quality of the libraries was validated using Agilent 2100 Bioanalyzer (Agilent Technologies) following the manufacturer’s instruction. Cluster generation was performed using the Illumina cluster station. Single-end sequencing on the HiSeq2500 and Genome Analyzer IIx followed a standard protocol. The fluorescent images were processed to sequences and transformed to FastQ format using the Genome Analyzer Pipeline Analysis software 1.8.2 (Illumina). The sequence output was controlled for general quality features, sequencing adapter clipping and demultiplexing using the fastq-mcf and fastq-multx tool of ea-utils [91].
Read mapping, bioinformatics and statistics
Quality of the sequencing output was analyzed using FastQC (Babraham Bioinformatics). All sequenced libraries were mapped to the YPIII genome (NC_010465) and the pYV plasmid (accessions NC_006153) using Bowtie2 (version 2.1.0) [92] with default parameters. After read mapping, SAMtools [93] was employed to filter the resulting bam files for uniquely mapped reads (both strands), which were the basis for downstream analyses. In summary, between 36–95% and 63–97% of the cDNA reads in the +TAP and-TAP libraries could be mapped to the YPIII sequence, of which 3–22% of +TAP reads and 2–23% of-TAP reads mapped uniquely to the genome.
Identification of transcription start sites (TSSs)
In a preliminary step, sample libraries were normalized to million uniquely mapped reads and for every base the coverage and the number of reads starting at the respective position were calculated. Then, biological replicates were combined/merged by averaging coverage and read starts data. For detection of TSSs that are associated with mRNAs an adapted method of Schlüter et al. [94] was applied on YPIII +TAP and YP89 +TAP libraries. Initially, the genome was scanned for potential TSSs. A TSS was defined as the starting position of at least 10 cDNA reads which map to the same genomic position at their 5’-end. Moreover, the read coverage of these reads had to be at least 2-fold higher than the coverage of overlapping reads. In the next step, the potential TSSs were extended in downstream direction to transcripts of continuous coverage and classified according to the minimal transcriptional unit (MTU) model [94]. The MTU model extends the given annotation of protein coding genes by virtual 5’ - and 3’-UTRs of lengths 54 nt and 20 nt for genes where 5’-UTRs are not annotated. Transcripts were retained if they could be assigned to one of three classes: mTSS (TSS of messenger RNA), lmTSS (TSS of leaderless mRNAs), rmTSS (TSS of mRNAs to be reannotated). A TSS was associated with an mRNA and classified as mTSS, if the TSS transcript started upstream of the corresponding gene and overlapped with its MTU. lmTSS represent the TSS of leaderless transcripts. In this case the 5’-UTR of the transcript is <10 nt. In some cases TSS candidates could not be identified upstream of genes, but in close proximity downstream to the start codon. TSS transcripts with a maximal distance of 70 nt were classified as rmTSS and indicate genes, which need to be reannotated. Following classification, adjacent TSS with distance ≤ 2 nt were clustered and the TSS with the highest number of read starts counted was reported as the primary TSS.
Recently, an automated approach for the detection of TSS in dRNA-seq data was proposed by Schmidtke et al. [95], which exploits the difference of read starts distributions between a library from untreated total RNA (-) and a library enriched for primary transcripts (+). Based on the assumption that the number of mapped reads in a defined genomic interval follows a Poisson distribution, the Skellam distribution is used to detect genomic positions, where the difference of observed read starts significantly exceeds the difference of expected read starts. These positions are then defined as TSS. We further applied TSSAR [96], an implementation of the dRNA-seq TSS detection method, on our RNA-seq data. Different to the standard dRNA-seq experimental setup, the background distribution of read starts was obtained from-TAP libraries, which are specifically depleted for cDNAs derived from fragments containing the 5’-end of primary transcripts, while the corresponding replicate of the +TAP sample represents the unbiased library (S1 Fig). p-values of TSSAR TSS predictions were added to the set of manually curated TSS and in case of matching positions (S2 Dataset). The newly identified TSSs were labelled to following conventions, x_TSS_n, where “x” indicates the YPIII genomic replicon (i.e. YPK, chromosome; YPKp, pYV virulence plasmid).
Detection of conserved sequence motifs
To investigate potential sequence conservation at and around the newly determined TSSs, a sequence logo for the -3 to +4 neighborhoods of all 1151 TSSs was generated using the WebLogo software [60]. To compute conserved sequence motifs in the -10 and -35 promotor region we performed de-novo motif discovery using the MEME software [63]. As input for motif detection in the -10 region served the subsequences starting at position -15 and ending at position -3 (relative to the TSS) of all 1151 TSSs determined in this study. For the -35 region, we used the subsequences starting at position -45 and ending -25. We ran MEME in one occurence per sequence (OOPS) mode and searched for motifs between length three and eight for the -10 region and between length three and five for the -35 region.
Discovery of novel non-coding RNAs
To identify expressed small regulatory RNAs, a global screen in all +TAP samples for unannotated trans-encoded sRNAs and cis-encoded antisense RNAs was performed. For this, transcripts were assembled from reads and subsequently classified. For non-coding RNA classification, TSS data were included in the YPIII annotation. In a first step, transcripts seeds, which correspond to genomic regions of minimal length of 40 nt and a continuous coverage of at least 30 reads, were identified. The resultant transcripts were extended on both ends until the coverage dropped to 10% of the transcript’s maximum coverage or if the coverage was lower than 5 reads. Finally, transcripts located in intergenic regions without overlapping UTRs were classified as trans-encoded sRNAs, while transcripts found on the strand opposite to a protein-coding gene were defined as cis-encoded antisense RNAs. Finally, all TSS and non-coding RNA candidates were inspected manually and reported if they passed this last filter. The novel non-coding RNAs were labelled according to the common convention (Ysr_n) with ongoing numbers (n).
Differential expression analysis and identification of CRP-dependent transcripts
To detect ORFs and trans-encoded sRNA that are differentially expressed in YPIII and the crp mutant YP89, we used DESeq (version 1.12.1) [73] for all differential expression (DE) analyses following the default analysis steps described in the package’s vignette. For each comparison HTSeq in union count mode was used to generate raw read counts required by DESeq as basis for DE analysis.
Computational prediction of CRP binding sites
In this study, a string-based prediction of conserved CRP binding site motifs was performed for trans-encoded sRNA which were classified as CRP-dependent by differential gene expression analysis. The CRP consensus sequence (TGTGA-6 nt-TCACA) was used to scan regions 200 nt upstream and 100 nt downstream of the sRNA 5’-end for putative CRP binding sites, allowing a maximum of two mismatches.
Prediction of riboswitch-like elements within 5’-UTRs
Candidate mRNAs with 5’-UTRs ≥ 200 nt were screened for riboswitch-like elements (RLEs). For this purpose the entire 5’-UTR along with 50 nt of the coding region were scanned for RLEs using the RibEx Riboswitch Explorer [67].
Data access
The high-throughput read data is deposited at the European Nucleotide Archive (ENA) with the accession no. PRJEB7268 (http://www.ebi.ac.uk/ena/data/view/PRJEB7268). A complete list of the TSSs, antisense and trans-encoded sRNAs is provided in the S2 and S3 Datasets. The comparative transcriptome analyses are given in S4–S6 Datasets. The annotation of global TSSs, reannotated ORFs, trans - and cis-encoded RNAs are provided as TXT files (S1–S7 Files).
5’ RACE and Northern blotting
Total RNA was isolated using the SV Total RNA Isolation System (Promega) as described [52]. 5’ RACE was performed using the 5’/3’ RACE Kit (Roche). For Northern blotting 10–20 μg total RNA were separated using 7 M urea/10% acrylamide/0.6 x TBE gels. The RNA was transferred onto positively charged membranes (Roche) by semi dry blotting in 1 x TBE and UV crosslinked. For radioactive labeling 20 μCi γ-ATP32 (Hartmann Analytic) were mixed with 10 U T4 Polynucleotide kinase (Fermentas) and 4 pmol oligonucleotide and incubated at 37°C for 1 h. The reaction was stopped by addition of STE buffer (100 mM NaCl, 10 mM Tris, pH 8, 1 mM EDTA) and the labeled oligonucleotides were purified using MicroSpin G-25 columns (GE Healthcare). The probes were mixed with 500 μg yeast tRNA (Invitrogen) and 250 μg salmon sperm DNA (Invitrogen) to reduce unspecific probe binding. Denaturation was carried out for 10 min at 95°C. Hybridization was performed in hybridization buffer (0.5 M Na2HPO4, pH 7.2, 1 mM EDTA pH 7.5, 7% [w/v] SDS) over night at 68°C. The blots were washed in washing buffer (40 mM Na2HPO4, pH 7.2, 1 mM EDTA, pH 7.5, 1% [w/v] SDS) and exposed to a Phosphorimaging screen and analyzed with a Typhoon FLA-9000 (GE Healthcare).
Quantitative real-time RT-PCR
qRT-PCR was performed with DNA digested total RNA (1 ng/μl) using the SensiFastNoRox Kit (Bioline). Primers employed for analyzing relative gene expression are listed S1 Table. The 5S rRNA gene was used for normalization and relative gene expression was calculated as described earlier [97]. Primer efficiencies were determined experimentally using serial dilutions of genomic Y. pseudotuberculosis YPIII DNA. Primer efficiencies are: 5S rRNA: 2.22; acnA (YPK_2030): 2.21; actP (YPK_3923): 2.17; ail (YPK_1268): 2.15; gntP (YPK_0762): 2.33; katY (YPK_3388): 2.40; sodC (YPK_3445): 1.91; uxaC (YPK_0554): 2.15; yopD (pYV0054): 2.20; yopE (pYV0025): 2.07; yopP/J (pYV0098): 1.89; yscW (pYV0075): 2.3; adhE (YPK_2072): 2.03; fliA (YPK_2380): 1.91; fliC (YPK_2381): 2.03; glnA (YPK_4189): 2.02; glpD (YPK_0152): 2.22; invA (YPK_2429): 2.10; pykF (YPK_1855): 1.91; fumC (YPK_1985): 2.12; glmS (YPK_4229): 2.21; YPK_1731 : 2.17; YPK_2197 : 2.19; YPK_2200 : 2.10.
Western blotting
Y. pseudotuberculosis YPIII, the isogenic mutants YP89 (Δcrp) and YP80 (Δhfq) were grown in LB medium to exponential phase (OD600 0.5) and stationary phase (16 h) at 25°C and 37°C (S8 Fig). For immunological detection of Hfq, H-NS and CRP, cell extracts of equal amounts of bacteria were prepared and separated on a 15% polyacrylamide SDS gel [89]. Proteins were transferred onto an Immobilon-P membrane (Millipore) and probed with polyclonal antibodies directed against Hfq, H-NS or CRP as described [56,98].
Purification of CRP-His6 and electrophoretic mobility shift assays (EMSAs)
CRP-His6 was purified as described [34]. For the EMSAs, the DNA fragment harboring the potential CRP binding site(s) was mixed with a control DNA fragment (csiD gene fragment of E. coli) in equimolar amounts. Increasing amounts of the CRP protein in 1 x binding buffer (10 mM Tris-HCl pH 7.5, 1 mM EDTA, 5 mM DTT, 5% glycerol, 10 mM NaCl, 1 mM MgCl2, 0.1 mg/ml BSA) containing 0.2 mM cAMP were added and the mixture was incubated at 30°C for 20 min. Subsequently, the reactions were separated on a 4% native polyacrylamide gel and the DNA fragments visualized with ethidium bromide [34].
DNase I footprinting
For DNase I footprinting, segments of the sR018 and ysr212 promoters harboring the predicted CRP binding site were amplified by PCR using a digoxigenin (DIG)-labelled primer and a non-labelled primer (S1 Table). PCR fragments were purified, incubated with the purified CRP protein in the presence or absence of cAMP as described for the EMSAs. The PCR products were digested with DNase I of an appropriate dilution and the resulting products were separated and visualized as described previously [56]. The protected nucleotides were identified by comparison with a sequence ladder generated with the same DIG-labelled primer used for the amplification of the fragment by PCR.
Detection of acetate and acetyl-CoA
For the detection of acetate and acetyl-CoA we used an enzymatic assay system (BioAssay Systems) and followed the instructions of the manufacturers.
Supporting Information
Zdroje
1. Green J, Rolfe MD, Smith LJ (2014) Transcriptional regulation of bacterial virulence gene expression by molecular oxygen and nitric oxide. Virulence 5 : 794–809. doi: 10.4161/viru.27794 25603427
2. Duprey A, Reverchon S, Nasser W (2014) Bacterial virulence and Fis: adapting regulatory networks to the host environment. Trends Microbiol 22 : 92–99. doi: 10.1016/j.tim.2013.11.008 24370464
3. Miller JF, Mekalanos JJ, Falkow S (1989) Coordinate regulation and sensory transduction in the control of bacterial virulence. Science 243 : 916–922. 2537530
4. Konkel ME, Tilly K (2000) Temperature-regulated expression of bacterial virulence genes. Microbes Infect 2 : 157–166. 10742688
5. Geissmann T, Marzi S, Romby P (2009) The role of mRNA structure in translational control in bacteria. RNA Biol 6 : 153–160. 19885993
6. Schiano CA, Lathem WW (2012) Post-transcriptional regulation of gene expression in Yersinia species. Front Cell Infect Microbiol 2 : 129. doi: 10.3389/fcimb.2012.00129 23162797
7. Papenfort K, Vogel J (2014) Small RNA functions in carbon metabolism and virulence of enteric pathogens. Front Cell Infect Microbiol 4 : 91. doi: 10.3389/fcimb.2014.00091 25077072
8. Heroven AK, Bohme K, Dersch P (2012) The Csr/Rsm system of Yersinia and related pathogens: A post-transcriptional strategy for managing virulence. RNA Biol 9.
9. Harris JF, Micheva-Viteva S, Li N, Hong-Geller E (2013) Small RNA-mediated regulation of host-pathogen interactions. Virulence 4 : 785–795. doi: 10.4161/viru.26119 23958954
10. Henkin TM (2008) Riboswitch RNAs: using RNA to sense cellular metabolism. Genes Dev 22 : 3383–3390. doi: 10.1101/gad.1747308 19141470
11. Breaker RR (2011) Prospects for riboswitch discovery and analysis. Mol Cell 43 : 867–879. doi: 10.1016/j.molcel.2011.08.024 21925376
12. Kortmann J, Narberhaus F (2012) Bacterial RNA thermometers: molecular zippers and switches. Nat Rev Microbiol 10 : 255–265. doi: 10.1038/nrmicro2730 22421878
13. Serganov A, Nudler E (2013) A decade of riboswitches. Cell 152 : 17–24. doi: 10.1016/j.cell.2012.12.024 23332744
14. Coppins RL, Hall KB, Groisman EA (2007) The intricate world of riboswitches. Curr Opin Microbiol 10 : 176–181. 17383225
15. Chao Y, Vogel J (2010) The role of Hfq in bacterial pathogens. Curr Opin Microbiol 13 : 24–33. doi: 10.1016/j.mib.2010.01.001 20080057
16. Koo JT, Alleyne TM, Schiano CA, Jafari N, Lathem WW (2011) Global discovery of small RNAs in Yersinia pseudotuberculosis identifies Yersinia-specific small, noncoding RNAs required for virulence. Proc Natl Acad Sci U S A 108: E709–717. doi: 10.1073/pnas.1101655108 21876162
17. Schiano CA, Koo JT, Schipma MJ, Caulfield AJ, Jafari N, et al. (2014) Genome-wide analysis of small RNAs expressed by Yersinia pestis identifies a regulator of the Yop-Ysc type III secretion system. J Bacteriol 196 : 1659–1670. doi: 10.1128/JB.01456-13 24532772
18. Beauregard A, Smith EA, Petrone BL, Singh N, Karch C, et al. (2013) Identification and characterization of small RNAs in Yersinia pestis. RNA Biol 10 : 397–405. doi: 10.4161/rna.23590 23324607
19. Qu Y, Bi L, Ji X, Deng Z, Zhang H, et al. (2012) Identification by cDNA cloning of abundant sRNAs in a human-avirulent Yersinia pestis strain grown under five different growth conditions. Future Microbiol 7 : 535–547. doi: 10.2217/fmb.12.13 22439729
20. Yan Y, Su S, Meng X, Ji X, Qu Y, et al. (2013) Determination of sRNA expressions by RNA-seq in Yersinia pestis grown in vitro and during infection. PLoS One 8: e74495. doi: 10.1371/journal.pone.0074495 24040259
21. Papenfort K, Vogel J (2010) Regulatory RNA in bacterial pathogens. Cell Host Microbe 8 : 116–127. doi: 10.1016/j.chom.2010.06.008 20638647
22. Bockemuhl J, Roggentin P (2004) [Intestinal yersiniosis. Clinical importance, epidemiology, diagnosis, and prevention]. Bundesgesundheitsblatt Gesundheitsforschung Gesundheitsschutz 47 : 685–691. 15254824
23. Achtman M, Zurth K, Morelli G, Torrea G, Guiyoule A, et al. (1999) Yersinia pestis, the cause of plague, is a recently emerged clone of Yersinia pseudotuberculosis. Proc Natl Acad Sci U S A 96 : 14043–14048. 10570195
24. Reuter S, Connor TR, Barquist L, Walker D, Feltwell T, et al. (2014) Parallel independent evolution of pathogenicity within the genus Yersinia. Proc Natl Acad Sci U S A 111 : 6768–6773. doi: 10.1073/pnas.1317161111 24753568
25. Chain PS, Carniel E, Larimer FW, Lamerdin J, Stoutland PO, et al. (2004) Insights into the evolution of Yersinia pestis through whole-genome comparison with Yersinia pseudotuberculosis. Proc Natl Acad Sci U S A 101 : 13826–13831. 15358858
26. Wren BW (2003) The yersiniae—a model genus to study the rapid evolution of bacterial pathogens. Nature Reviews Microbiology 1 : 55–64. 15040180
27. Koornhof HJ, Smego RA Jr., Nicol M (1999) Yersiniosis. II: The pathogenesis of Yersinia infections. Eur J Clin Microbiol Infect Dis 18 : 87–112. 10219574
28. Smego RA, Frean J, Koornhof H.J. (1999) Yersiniosis I: Microbiological and clinicoepidemiological aspects of plague and non-plague Yersinia infections. Eur J Clin Microbiol Infect Dis 18 : 1–15. 10192708
29. Kapperud G (1981) Survey on the reservoirs of Yersinia enterocolitica and Yersinia enterocolitica-like bacteria in Scandinavia. Acta Pathol Microbiol Scand B 89 : 29–35. 7020335
30. Straley SC, Perry RD (1995) Environmental modulation of gene expression and pathogenesis in Yersinia. Trends Microbiol 3 : 310–317. 8528615
31. Saier MH Jr. (1998) Multiple mechanisms controlling carbon metabolism in bacteria. Biotechnol Bioeng 58 : 170–174. 10191387
32. Zheng D, Constantinidou C, Hobman JL, Minchin SD (2004) Identification of the CRP regulon using in vitro and in vivo transcriptional profiling. Nucleic Acids Res 32 : 5874–5893. 15520470
33. Zhan L, Han Y, Yang L, Geng J, Li Y, et al. (2008) The cyclic AMP receptor protein, CRP, is required for both virulence and expression of the minimal CRP regulon in Yersinia pestis biovar microtus. Infect Immun 76 : 5028–5037. doi: 10.1128/IAI.00370-08 18710863
34. Heroven AK, Sest M, Pisano F, Scheb-Wetzel M, Steinmann R, et al. (2012) Crp induces switching of the CsrB and CsrC RNAs in Yersinia pseudotuberculosis and links nutritional status to virulence. Front Cell Infect Microbiol 2 : 158. doi: 10.3389/fcimb.2012.00158 23251905
35. Avican K, Fahlgren A, Huss M, Heroven AK, Beckstette M, et al. (2015) Reprogramming of Yersinia from virulent to persistent mode revealed by complex in vivo RNA-seq analysis. PLoS Pathog 11: e1004600. doi: 10.1371/journal.ppat.1004600 25590628
36. Lathem WW, Schroeder JA, Bellows LE, Ritzert JT, Koo JT, et al. (2014) Posttranscriptional regulation of the Yersinia pestis cyclic AMP receptor protein Crp and impact on virulence. MBio 5: e01038–01013. doi: 10.1128/mBio.01038-13 24520064
37. Zhan L, Yang L, Zhou L, Li Y, Gao H, et al. (2009) Direct and negative regulation of the sycO-ypkA-ypoJ operon by cyclic AMP receptor protein (CRP) in Yersinia pestis. BMC Microbiol 9 : 178. doi: 10.1186/1471-2180-9-178 19703315
38. Kim TJ, Chauhan S, Motin VL, Goh EB, Igo MM, et al. (2007) Direct transcriptional control of the plasminogen activator gene of Yersinia pestis by the cyclic AMP receptor protein. J Bacteriol 189 : 8890–8900. 17933899
39. Bücker R, Heroven AK, Becker J, Dersch P, Wittmann C (2014) The pyruvate-tricarboxylic acid cycle node: a focal point of virulence control in the enteric pathogen Yersinia pseudotuberculosis. J Biol Chem 289 : 30114–30132. doi: 10.1074/jbc.M114.581348 25164818
40. Davis KM, Mohammadi S, Isberg RR (2015) Community behavior and spatial regulation within a bacterial microcolony in deep tissue sites serves to protect against host attack. Cell Host Microbe 17 : 21–31. doi: 10.1016/j.chom.2014.11.008 25500192
41. Nuss AM, Schuster F, Kathrin Heroven A, Heine W, Pisano F, et al. (2014) A direct link between the global regulator PhoP and the Csr regulon in Y. pseudotuberculosis through the small regulatory RNA CsrC. RNA Biol 11 : 580–593. 24786463
42. Fahlgren A, Avican K, Westermark L, Nordfelth R, Fallman M (2014) Colonization of cecum is important for development of persistent infection by Yersinia pseudotuberculosis. Infect Immun 82 : 3471–3482. doi: 10.1128/IAI.01793-14 24891107
43. Durand EA, Maldonado-Arocho FJ, Castillo C, Walsh RL, Mecsas J (2010) The presence of professional phagocytes dictates the number of host cells targeted for Yop translocation during infection. Cell Microbiol 12 : 1064–1082. doi: 10.1111/j.1462-5822.2010.01451.x 20148898
44. Frost S, Ho O, Login FH, Weise CF, Wolf-Watz H, et al. (2012) Autoproteolysis and intramolecular dissociation of Yersinia YscU precedes secretion of its C-terminal polypeptide YscU(CC). PLoS One 7: e49349. doi: 10.1371/journal.pone.0049349 23185318
45. Bolin I, Portnoy DA, Wolf-Watz H (1985) Expression of the temperature-inducible outer membrane proteins of yersiniae. Infect Immun 48 : 234–240. 3980086
46. Sharma CM, Vogel J (2014) Differential RNA-seq: the approach behind and the biological insight gained. Curr Opin Microbiol 19 : 97–105. doi: 10.1016/j.mib.2014.06.010 25024085
47. Rosinski-Chupin I, Soutourina O, Martin-Verstraete I (2014) Riboswitch discovery by combining RNA-seq and genome-wide identification of transcriptional start sites. Methods Enzymol 549 : 3–27. doi: 10.1016/B978-0-12-801122-5.00001-5 25432742
48. Soutourina OA, Monot M, Boudry P, Saujet L, Pichon C, et al. (2013) Genome-wide identification of regulatory RNAs in the human pathogen Clostridium difficile. PLoS Genet 9: e1003493. doi: 10.1371/journal.pgen.1003493 23675309
49. Bolin I, Norlander I, Wolf-Watz H (1982) Temperature-inducible outer membrane protein of Yersinia pseudotuberculosis and Yersinia enterocolitica is associated with the virulence plasmid. Infect Immun 37 : 506–512. 6749681
50. Hoe NP, Minion FC, Goguen JD (1992) Temperature sensing in Yersinia pestis: regulation of yopE transcription by lcrF. J Bacteriol 174 : 4275–4286. 1624422
51. Skurnik M, Toivanen P (1992) LcrF is the temperature-regulated activator of the yadA gene of Yersinia enterocolitica and Yersinia pseudotuberculosis. J Bacteriol 174 : 2047–2051. 1548243
52. Böhme K, Steinmann R, Kortmann J, Seekircher S, Heroven AK, et al. (2012) Concerted actions of a thermo-labile regulator and a unique intergenic RNA thermosensor control Yersinia virulence. PLoS Pathog 8: e1002518. doi: 10.1371/journal.ppat.1002518 22359501
53. Vogel J, Luisi BF (2011) Hfq and its constellation of RNA. Nat Rev Microbiol 9 : 578–589. doi: 10.1038/nrmicro2615 21760622
54. Ali Azam T, Iwata A, Nishimura A, Ueda S, Ishihama A (1999) Growth phase-dependent variation in protein composition of the Escherichia coli nucleoid. J Bacteriol 181 : 6361–6370. 10515926
55. Garcia E, Nedialkov YA, Elliott J, Motin VL, Brubaker RR (1999) Molecular characterization of KatY (antigen 5), a thermoregulated chromosomally encoded catalase-peroxidase of Yersinia pestis. J Bacteriol 181 : 3114–3122. 10322012
56. Heroven A, Nagel G, Tran HJ, Parr S, Dersch P (2004) RovA is autoregulated and antagonizes H-NS-mediated silencing of invasin and rovA expression in Yersinia pseudotuberculosis. Mol Microbiol 53 : 871–888. 15255899
57. Lawrenz MB, Miller VL (2007) Comparative analysis of the regulation of rovA from the pathogenic yersiniae. J Bacteriol 189 : 5963–5975. 17573476
58. Tsui H, Leung H, Winkler ME (1994) Characterization of broadly pleiotropic phenotypes caused by an hfq insertion mutation in Escherichia coli K-12. Molecular Microbiology 13 : 35–49. 7984093
59. Bai G, Golubov A, Smith EA, McDonough KA (2010) The importance of the small RNA chaperone Hfq for growth of epidemic Yersinia pestis, but not Yersinia pseudotuberculosis, with implications for plague biology. J Bacteriol 192 : 4239–4245. doi: 10.1128/JB.00504-10 20543069
60. Crooks GE, Hon G, Chandonia JM, Brenner SE (2004) WebLogo: a sequence logo generator. Genome Res 14 : 1188–1190. 15173120
61. Kim D, Hong JS, Qiu Y, Nagarajan H, Seo JH, et al. (2012) Comparative analysis of regulatory elements between Escherichia coli and Klebsiella pneumoniae by genome-wide transcription start site profiling. PLoS Genet 8: e1002867. doi: 10.1371/journal.pgen.1002867 22912590
62. Kröger C, Dillon SC, Cameron AD, Papenfort K, Sivasankaran SK, et al. (2012) The transcriptional landscape and small RNAs of Salmonella enterica serovar Typhimurium. Proc Natl Acad Sci U S A 109: E1277–1286. doi: 10.1073/pnas.1201061109 22538806
63. Bailey TL, Elkan C (1994) Fitting a mixture model by expectation maximization to discover motifs in biopolymers. Proc Int Conf Intell Syst Mol Biol 2 : 28–36. 7584402
64. Kröger C, Dillon SC, Cameron AD, Papenfort K, Sivasankaran SK, et al. (2012) The transcriptional landscape and small RNAs of Salmonella enterica serovar Typhimurium. Proc Natl Acad Sci U S A 109: E1277–1286. doi: 10.1073/pnas.1201061109 22538806
65. Sharma CM, Hoffmann S, Darfeuille F, Reignier J, Findeiss S, et al. (2010) The primary transcriptome of the major human pathogen Helicobacter pylori. Nature 464 : 250–255. doi: 10.1038/nature08756 20164839
66. Wurtzel O, Yoder-Himes DR, Han K, Dandekar AA, Edelheit S, et al. (2012) The single-nucleotide resolution transcriptome of Pseudomonas aeruginosa grown in body temperature. PLoS Pathog 8: e1002945. doi: 10.1371/journal.ppat.1002945 23028334
67. Abreu-Goodger C, Merino E (2005) RibEx: a web server for locating riboswitches and other conserved bacterial regulatory elements. Nucleic Acids Res 33: W690–692. 15980564
68. Regulski EE, Moy RH, Weinberg Z, Barrick JE, Yao Z, et al. (2008) A widespread riboswitch candidate that controls bacterial genes involved in molybdenum cofactor and tungsten cofactor metabolism. Mol Microbiol 68 : 918–932. doi: 10.1111/j.1365-2958.2008.06208.x 18363797
69. Cromie MJ, Shi Y, Latifi T, Groisman EA (2006) An RNA sensor for intracellular Mg(2+). Cell 125 : 71–84. 16615891
70. Groisman EA, Cromie MJ, Shi Y, Latifi T (2006) A Mg2+-responding RNA that controls the expression of a Mg2+ transporter. Cold Spring Harb Symp Quant Biol 71 : 251–258. 17381304
71. Brock JE, Pourshahian S, Giliberti J, Limbach PA, Janssen GR (2008) Ribosomes bind leaderless mRNA in Escherichia coli through recognition of their 5'-terminal AUG. RNA 14 : 2159–2169. doi: 10.1261/rna.1089208 18755843
72. Thomason MK, Fontaine F, De Lay N, Storz G (2012) A small RNA that regulates motility and biofilm formation in response to changes in nutrient availability in Escherichia coli. Mol Microbiol 84 : 17–35. doi: 10.1111/j.1365-2958.2012.07965.x 22289118
73. Anders S, Huber W (2010) Differential expression analysis for sequence count data. Genome Biol 11: R106. doi: 10.1186/gb-2010-11-10-r106 20979621
74. Isberg RR (1989) Determinants for thermoinducible cell binding and plasmid-encoded cellular penetration detected in the absence of the Yersinia pseudotuberculosis invasin protein. Infect Immun 57 : 1998–2005. 2543628
75. Isberg RR, Swain A, Falkow S (1988) Analysis of expression and thermoregulation of the Yersinia pseudotuberculosis inv gene with hybrid proteins. Infect Immun 56 : 2133–2138. 2840402
76. Kapatral V, Olson JW, Pepe JC, Miller VL, Minnich SA (1996) Temperature-dependent regulation of Yersinia enterocolitica Class III flagellar genes. Mol Microbiol 19 : 1061–1071. 8830263
77. Motin VL, Georgescu AM, Fitch JP, Gu PP, Nelson DO, et al. (2004) Temporal global changes in gene expression during temperature transition in Yersinia pestis. J Bacteriol 186 : 6298–6305. 15342600
78. Wolfe AJ (2005) The acetate switch. Microbiol Mol Biol Rev 69 : 12–50. 15755952
79. Cummings JH, Pomare EW, Branch WJ, Naylor CP, Macfarlane GT (1987) Short chain fatty acids in human large intestine, portal, hepatic and venous blood. Gut 28 : 1221–1227. 3678950
80. Schiano CA, Bellows LE, Lathem WW (2010) The small RNA chaperone Hfq is required for the virulence of Yersinia pseudotuberculosis. Infect Immun 78 : 2034–2044. doi: 10.1128/IAI.01046-09 20231416
81. Petersen S, Young GM (2002) Essential role for cyclic AMP and its receptor protein in Yersinia enterocolitica virulence. Infect Immun 70 : 3665–3672. 12065508
82. Herbst K, Bujara M, Heroven AK, Opitz W, Weichert M, et al. (2009) Intrinsic thermal sensing controls proteolysis of Yersinia virulence regulator RovA. PLoS Pathog 5: e1000435. doi: 10.1371/journal.ppat.1000435 19468295
83. Nagel G, Heroven AK, Eitel J, Dersch P (2003) Function and regulation of the transcriptional activator RovA of Yersinia pseudotuberculosis. Adv Exp Med Biol 529 : 285–287. 12756772
84. Gopel Y, Luttmann D, Heroven AK, Reichenbach B, Dersch P, et al. (2011) Common and divergent features in transcriptional control of the homologous small RNAs GlmY and GlmZ in Enterobacteriaceae. Nucleic Acids Res 39 : 1294–1309. doi: 10.1093/nar/gkq986 20965974
85. Udekwu KI, Wagner EG (2007) Sigma E controls biogenesis of the antisense RNA MicA. Nucleic Acids Res 35 : 1279–1288. 17267407
86. Johansen J, Rasmussen AA, Overgaard M, Valentin-Hansen P (2006) Conserved small non-coding RNAs that belong to the sigma E regulon: role in down-regulation of outer membrane proteins. J Mol Biol 364 : 1–8. 17007876
87. Lin HH, Hsu CC, Yang CD, Ju YW, Chen YP, et al. (2011) Negative effect of glucose on ompA mRNA stability: a potential role of cyclic AMP in the repression of hfq in Escherichia coli. J Bacteriol 193 : 5833–5840. doi: 10.1128/JB.05359-11 21840983
88. Bücker R, Heroven AK, Becker J, Dersch P, Wittmann C (2014) The pyruvate—tricarboxylic acid cycle node: a focal point of virulence control in the enteric pathogen Yersinia pseudotuberculosis. J Biol Chem. 289 : 30114–32. doi: 10.1074/jbc.M114.581348 25164818
89. Sambrook J (2001) Molecular Cloning: A Laboratory Manual,: Cold Spring Harbor Laboratories, Cold Spring Harbor, NY.
90. Dötsch A, Eckweiler D, Schniederjans M, Zimmermann A, Jensen V, et al. (2012) The Pseudomonas aeruginosa transcriptome in planktonic cultures and static biofilms using RNA sequencing. PLoS One 7: e31092. doi: 10.1371/journal.pone.0031092 22319605
91. Aronesty E (2011) ea-utils: Command-line tools for processing biological data. http://code.google.com/p/ea-utils.
92. Langmead B, Salzberg SL (2012) Fast gapped-read alignment with Bowtie 2. Nat Methods 9 : 357–359. doi: 10.1038/nmeth.1923 22388286
93. Li H, Handsaker B, Wysoker A, Fennell T, Ruan J, et al. (2009) The sequence alignment/map format and SAMtools. Bioinformatics 25 : 2078–2079. doi: 10.1093/bioinformatics/btp352 19505943
94. Schluter JP, Reinkensmeier J, Barnett MJ, Lang C, Krol E, et al. (2013) Global mapping of transcription start sites and promoter motifs in the symbiotic alpha-proteobacterium Sinorhizobium meliloti 1021. BMC Genomics 14 : 156. doi: 10.1186/1471-2164-14-156 23497287
95. Schmidtke C, Findeiss S, Sharma CM, Kuhfuss J, Hoffmann S, et al. (2012) Genome-wide transcriptome analysis of the plant pathogen Xanthomonas identifies sRNAs with putative virulence functions. Nucleic Acids Res 40 : 2020–2031. doi: 10.1093/nar/gkr904 22080557
96. Amman F, Wolfinger MT, Lorenz R, Hofacker IL, Stadler PF, et al. (2014) TSSAR: TSS annotation regime for dRNA-seq data. BMC Bioinformatics 15 : 89. doi: 10.1186/1471-2105-15-89 24674136
97. Pfaffl MW (2001) A new mathematical model for relative quantification in real-time RT-PCR. Nucleic Acids Res 29: e45. 11328886
98. Heroven AK, Dersch P (2006) RovM, a novel LysR-type regulator of the virulence activator gene rovA, controls cell invasion, virulence and motility of Yersinia pseudotuberculosis. Mol Microbiol 62 : 1469–1483. 17074075
Štítky
Genetika Reprodukční medicína
Článek NLRC5 Exclusively Transactivates MHC Class I and Related Genes through a Distinctive SXY ModuleČlánek Inhibition of Telomere Recombination by Inactivation of KEOPS Subunit Cgi121 Promotes Cell LongevityČlánek HOMER2, a Stereociliary Scaffolding Protein, Is Essential for Normal Hearing in Humans and MiceČlánek LRGUK-1 Is Required for Basal Body and Manchette Function during Spermatogenesis and Male FertilityČlánek The GATA Factor Regulates . Developmental Timing by Promoting Expression of the Family MicroRNAsČlánek Systems Biology of Tissue-Specific Response to Reveals Differentiated Apoptosis in the Tick VectorČlánek Phenotype Specific Analyses Reveal Distinct Regulatory Mechanism for Chronically Activated p53Článek The Nuclear Receptor DAF-12 Regulates Nutrient Metabolism and Reproductive Growth in NematodesČlánek The ATM Signaling Cascade Promotes Recombination-Dependent Pachytene Arrest in Mouse SpermatocytesČlánek The Small Protein MntS and Exporter MntP Optimize the Intracellular Concentration of Manganese
Článek vyšel v časopisePLOS Genetics
Nejčtenější tento týden
2015 Číslo 3
-
Všechny články tohoto čísla
- NLRC5 Exclusively Transactivates MHC Class I and Related Genes through a Distinctive SXY Module
- Licensing of Primordial Germ Cells for Gametogenesis Depends on Genital Ridge Signaling
- A Genomic Duplication is Associated with Ectopic Eomesodermin Expression in the Embryonic Chicken Comb and Two Duplex-comb Phenotypes
- Genome-wide Association Study and Meta-Analysis Identify as Genome-wide Significant Susceptibility Gene for Bladder Exstrophy
- Mutations of Human , Encoding the Mitochondrial Asparaginyl-tRNA Synthetase, Cause Nonsyndromic Deafness and Leigh Syndrome
- Exome Sequencing in an Admixed Isolated Population Indicates Variants Confer a Risk for Specific Language Impairment
- Genome-Wide Association Studies in Dogs and Humans Identify as a Risk Variant for Cleft Lip and Palate
- Rapid Evolution of Recombinant for Xylose Fermentation through Formation of Extra-chromosomal Circular DNA
- The Ribosome Biogenesis Factor Nol11 Is Required for Optimal rDNA Transcription and Craniofacial Development in
- Methyl Farnesoate Plays a Dual Role in Regulating Metamorphosis
- Maternal Co-ordinate Gene Regulation and Axis Polarity in the Scuttle Fly
- Maternal Filaggrin Mutations Increase the Risk of Atopic Dermatitis in Children: An Effect Independent of Mutation Inheritance
- Inhibition of Telomere Recombination by Inactivation of KEOPS Subunit Cgi121 Promotes Cell Longevity
- Clonality and Evolutionary History of Rhabdomyosarcoma
- HOMER2, a Stereociliary Scaffolding Protein, Is Essential for Normal Hearing in Humans and Mice
- Methylation-Sensitive Expression of a DNA Demethylase Gene Serves As an Epigenetic Rheostat
- BREVIPEDICELLUS Interacts with the SWI2/SNF2 Chromatin Remodeling ATPase BRAHMA to Regulate and Expression in Control of Inflorescence Architecture
- Seizures Are Regulated by Ubiquitin-specific Peptidase 9 X-linked (USP9X), a De-Ubiquitinase
- The Fun30 Chromatin Remodeler Fft3 Controls Nuclear Organization and Chromatin Structure of Insulators and Subtelomeres in Fission Yeast
- A Cascade of Iron-Containing Proteins Governs the Genetic Iron Starvation Response to Promote Iron Uptake and Inhibit Iron Storage in Fission Yeast
- Mutation in MRPS34 Compromises Protein Synthesis and Causes Mitochondrial Dysfunction
- LRGUK-1 Is Required for Basal Body and Manchette Function during Spermatogenesis and Male Fertility
- Cis-Regulatory Mechanisms for Robust Olfactory Sensory Neuron Class-restricted Odorant Receptor Gene Expression in
- Effects on Murine Behavior and Lifespan of Selectively Decreasing Expression of Mutant Huntingtin Allele by Supt4h Knockdown
- HDAC4-Myogenin Axis As an Important Marker of HD-Related Skeletal Muscle Atrophy
- A Conserved Domain in the Scc3 Subunit of Cohesin Mediates the Interaction with Both Mcd1 and the Cohesin Loader Complex
- Selective and Genetic Constraints on Pneumococcal Serotype Switching
- Bacterial Infection Drives the Expression Dynamics of microRNAs and Their isomiRs
- The GATA Factor Regulates . Developmental Timing by Promoting Expression of the Family MicroRNAs
- Accumulation of Glucosylceramide in the Absence of the Beta-Glucosidase GBA2 Alters Cytoskeletal Dynamics
- Reproductive Isolation of Hybrid Populations Driven by Genetic Incompatibilities
- The Contribution of Alu Elements to Mutagenic DNA Double-Strand Break Repair
- Systems Biology of Tissue-Specific Response to Reveals Differentiated Apoptosis in the Tick Vector
- Tfap2a Promotes Specification and Maturation of Neurons in the Inner Ear through Modulation of Bmp, Fgf and Notch Signaling
- The Lysine Acetyltransferase Activator Brpf1 Governs Dentate Gyrus Development through Neural Stem Cells and Progenitors
- PHABULOSA Controls the Quiescent Center-Independent Root Meristem Activities in
- DNA Polymerase ζ-Dependent Lesion Bypass in Is Accompanied by Error-Prone Copying of Long Stretches of Adjacent DNA
- Examining the Evolution of the Regulatory Circuit Controlling Secondary Metabolism and Development in the Fungal Genus
- Zinc Finger Independent Genome-Wide Binding of Sp2 Potentiates Recruitment of Histone-Fold Protein Nf-y Distinguishing It from Sp1 and Sp3
- GAGA Factor Maintains Nucleosome-Free Regions and Has a Role in RNA Polymerase II Recruitment to Promoters
- Neurospora Importin α Is Required for Normal Heterochromatic Formation and DNA Methylation
- Ccr4-Not Regulates RNA Polymerase I Transcription and Couples Nutrient Signaling to the Control of Ribosomal RNA Biogenesis
- Phenotype Specific Analyses Reveal Distinct Regulatory Mechanism for Chronically Activated p53
- A Systems-Level Interrogation Identifies Regulators of Blood Cell Number and Survival
- Morphological Mutations: Lessons from the Cockscomb
- Genetic Interaction Mapping Reveals a Role for the SWI/SNF Nucleosome Remodeler in Spliceosome Activation in Fission Yeast
- The Role of China in the Global Spread of the Current Cholera Pandemic
- The Nuclear Receptor DAF-12 Regulates Nutrient Metabolism and Reproductive Growth in Nematodes
- A Zinc Finger Motif-Containing Protein Is Essential for Chloroplast RNA Editing
- Resistance to Gray Leaf Spot of Maize: Genetic Architecture and Mechanisms Elucidated through Nested Association Mapping and Near-Isogenic Line Analysis
- Small Regulatory RNA-Induced Growth Rate Heterogeneity of
- Mitochondrial Dysfunction Reveals the Role of mRNA Poly(A) Tail Regulation in Oculopharyngeal Muscular Dystrophy Pathogenesis
- Complex Genomic Rearrangements at the Locus Include Triplication and Quadruplication
- Male-Biased Aganglionic Megacolon in the TashT Mouse Line Due to Perturbation of Silencer Elements in a Large Gene Desert of Chromosome 10
- Sex Ratio Meiotic Drive as a Plausible Evolutionary Mechanism for Hybrid Male Sterility
- Tertiary siRNAs Mediate Paramutation in .
- RECG Maintains Plastid and Mitochondrial Genome Stability by Suppressing Extensive Recombination between Short Dispersed Repeats
- Escape from X Inactivation Varies in Mouse Tissues
- Opposite Phenotypes of Muscle Strength and Locomotor Function in Mouse Models of Partial Trisomy and Monosomy 21 for the Proximal Region
- Glycosyl Phosphatidylinositol Anchor Biosynthesis Is Essential for Maintaining Epithelial Integrity during Embryogenesis
- Hyperdiverse Gene Cluster in Snail Host Conveys Resistance to Human Schistosome Parasites
- The Class Homeodomain Factors and Cooperate in . Embryonic Progenitor Cells to Regulate Robust Development
- Recombination between Homologous Chromosomes Induced by Unrepaired UV-Generated DNA Damage Requires Mus81p and Is Suppressed by Mms2p
- Synergistic Interactions between Orthologues of Genes Spanned by Human CNVs Support Multiple-Hit Models of Autism
- Gene Networks Underlying Convergent and Pleiotropic Phenotypes in a Large and Systematically-Phenotyped Cohort with Heterogeneous Developmental Disorders
- The ATM Signaling Cascade Promotes Recombination-Dependent Pachytene Arrest in Mouse Spermatocytes
- Combinatorial Control of Light Induced Chromatin Remodeling and Gene Activation in
- Linking Aβ42-Induced Hyperexcitability to Neurodegeneration, Learning and Motor Deficits, and a Shorter Lifespan in an Alzheimer’s Model
- The Complex Contributions of Genetics and Nutrition to Immunity in
- NatB Domain-Containing CRA-1 Antagonizes Hydrolase ACER-1 Linking Acetyl-CoA Metabolism to the Initiation of Recombination during . Meiosis
- Transcriptomic Profiling of Reveals Reprogramming of the Crp Regulon by Temperature and Uncovers Crp as a Master Regulator of Small RNAs
- Osteopetrorickets due to Snx10 Deficiency in Mice Results from Both Failed Osteoclast Activity and Loss of Gastric Acid-Dependent Calcium Absorption
- A Genomic Portrait of Haplotype Diversity and Signatures of Selection in Indigenous Southern African Populations
- Sequence Features and Transcriptional Stalling within Centromere DNA Promote Establishment of CENP-A Chromatin
- Inhibits Neuromuscular Junction Growth by Downregulating the BMP Receptor Thickveins
- Replicative DNA Polymerase δ but Not ε Proofreads Errors in and in
- Unsaturation of Very-Long-Chain Ceramides Protects Plant from Hypoxia-Induced Damages by Modulating Ethylene Signaling in
- The Small Protein MntS and Exporter MntP Optimize the Intracellular Concentration of Manganese
- A Meta-analysis of Gene Expression Signatures of Blood Pressure and Hypertension
- Pervasive Variation of Transcription Factor Orthologs Contributes to Regulatory Network Evolution
- Network Analyses Reveal Novel Aspects of ALS Pathogenesis
- A Role for the Budding Yeast Separase, Esp1, in Ty1 Element Retrotransposition
- Nab3 Facilitates the Function of the TRAMP Complex in RNA Processing via Recruitment of Rrp6 Independent of Nrd1
- A RecA Protein Surface Required for Activation of DNA Polymerase V
- PLOS Genetics
- Archiv čísel
- Aktuální číslo
- Informace o časopisu
Nejčtenější v tomto čísle- Clonality and Evolutionary History of Rhabdomyosarcoma
- Morphological Mutations: Lessons from the Cockscomb
- Maternal Filaggrin Mutations Increase the Risk of Atopic Dermatitis in Children: An Effect Independent of Mutation Inheritance
- Transcriptomic Profiling of Reveals Reprogramming of the Crp Regulon by Temperature and Uncovers Crp as a Master Regulator of Small RNAs
Kurzy
Zvyšte si kvalifikaci online z pohodlí domova
Současné možnosti léčby obezity
nový kurzAutoři: MUDr. Martin Hrubý
Všechny kurzyPřihlášení#ADS_BOTTOM_SCRIPTS#Zapomenuté hesloZadejte e-mailovou adresu, se kterou jste vytvářel(a) účet, budou Vám na ni zaslány informace k nastavení nového hesla.
- Vzdělávání



