-
Články
- Vzdělávání
- Časopisy
Top články
Nové číslo
- Témata
- Videa
- Podcasty
Nové podcasty
Reklama- Kariéra
Doporučené pozice
Reklama- Praxe
Antimicrobial-Induced DNA Damage and Genomic Instability in Microbial Pathogens
article has not abstract
Published in the journal: . PLoS Pathog 11(3): e32767. doi:10.1371/journal.ppat.1004678
Category: Pearls
doi: https://doi.org/10.1371/journal.ppat.1004678Summary
article has not abstract
Introduction
Combatting infectious disease is a critical global health concern and involves tackling both emerging infectious agents and newly–drug resistant strains of previously curable pathogens. The widespread and inappropriate use of antimicrobial agents has increased the frequency of resistance among human pathogens, including bacteria, fungi, and protozoan parasites, and threatened to undermine the efficacy of all existing antimicrobial drugs [1]. Whereas lethal doses of antimicrobials may select for preexisting resistant microbes, there is increasing interest in uncovering the cellular consequences of sublethal antimicrobial exposure on the development of antimicrobial resistance. There are numerous circumstances under which microbial organisms are exposed to low doses of antimicrobials, including in patients, in livestock animals, and in the environment [1–3]. Sublethal antimicrobial exposure can trigger DNA damage and genomic instability across the diversity of microbial pathogens, including bacterial and fungal species.
Here we investigate general mechanisms by which antimicrobials can damage microbial DNA. We also explore downstream cellular responses to DNA damage, including DNA repair. We will look at specific examples by which antimicrobial treatment, through DNA damage and cellular responses, can induce genetic perturbations ranging from small nucleotide mutationsto gross chromosomal rearrangements [1,4]. Overall, this review aims to explore genomic pressure exerted on bacterial and fungal pathogens by antimicrobial treatment, and implications for antimicrobial resistance.
Antimicrobial-Induced DNA Damage and Repair in Microbial Organisms
Microbial species contend with numerous environmental perturbations that can lead to DNA damage, including exposure to direct damage by ultraviolet (UV) light, or damage by chemical compounds. The ability to repair DNA damage and maintain genomic integrity is fundamental to survival of both bacterial and fungal pathogens. Even low doses of antimicrobials can directly or indirectly induce DNA damage and alterations (Fig. 1). In this section, we discuss general mechanisms by which antimicrobials can damage DNA, and strategies employed by microbial species to repair this damage.
Fig. 1. Antimicrobial-induced DNA damage in bacterial and fungal pathogens. 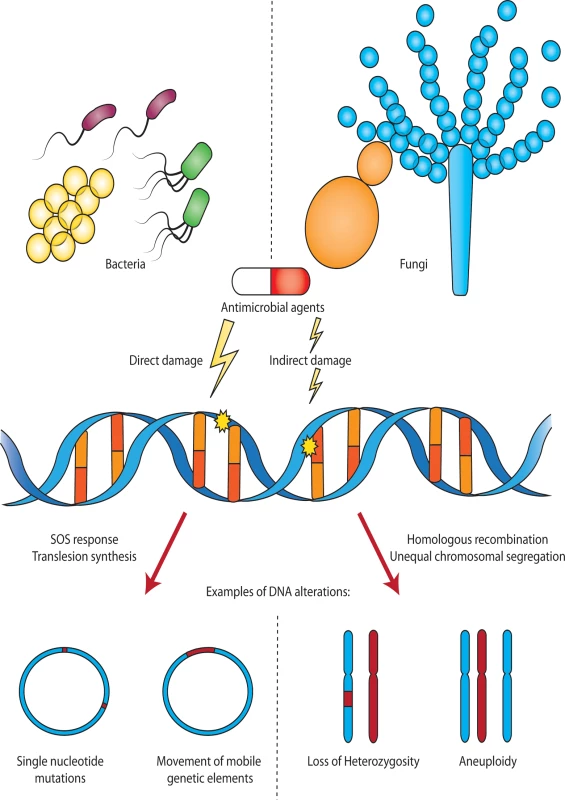
Sublethal doses of antimicrobial agents can directly or indirectly damage DNA in bacteria and fungi. In bacteria, DNA damage may lead to up-regulation of an SOS response, error-prone translesion DNA synthesis, or other stress responses that result in mutations including single nucleotide polymorphisms (SNPs) and the movement of mobile genetic elements. In fungi, treatment with antifungals can lead to DNA damage, resulting in homologous recombination and loss of heterozygosity (LOH), or other cellular stress responses, leading to unequal chromosomal segregation during mitosis and aneuploidy. Bacteria and fungi are not to scale. DNA damage by antimicrobial agents may occur by several distinct mechanisms. First, several antimicrobial agents cause direct chemical damage to DNA. An example of this is the antibiotic bleomycin, which binds DNA and directly induces double-strand breaks by a mechanism that is not fully understood [5]. Second, antimicrobials may interact with their target protein in a manner that directly induces DNA damage. For instance, the quinolone class of antibiotics specifically inhibits the ligase domain of topoisomerase enzymes, leaving the nuclease domains intact and thereby permitting the enzyme to cut DNA without re-ligation [6]. Finally, numerous antimicrobials result in metabolic perturbations, downstream of the interaction with their respective cellular targets. A commonly observed example of this is the production of reactive oxygen species (ROS) in response to antibiotics (including β-lactams, aminoglycosides, and quinolones) [7–9], antifungals (including polyenes and azoles) [10,11], and antiparasitics [12]. Antimicrobial-induced ROS, such as hydroxyl radicals, damage DNA through the formation of DNA strand breaks, and the incorporation of oxidized guanine residues into the genome [13,14].
Repair of damaged DNA is critical for microbial survival, yet certain DNA damage repair pathways may introduce mutations into the genome. For bacteria, the SOS response is the global response to DNA damage. Triggered by intracellular uncoated single-stranded DNA (ssDNA), the SOS response can be induced upon DNA damage, via the activation of RecA [15]. RecA polymers bind ssDNA, and upon activation, stimulate cleavage of the LexA repressor, leading to derepression of SOS genes, including enzymes involved in DNA repair processes such as nucleotide excision repair or recombination [16]. As part of DNA repair, there can be a trade-off between survival and the fidelity of repair. Thus, bacteria may employ a DNA damage tolerance strategy, where low-fidelity DNA polymerases Pol IV and Pol V are induced and facilitate DNA replication across DNA damage lesions in a manner that introduces errors into the genome [17]. Eukaryotic microbes have homologous strategies to repair or tolerate DNA damage, with a global response involving the expression of genes involved in nucleotide excision repair, and error-prone translesion synthesis polymerases such as DNA polymerase zeta, and Rev1 [18]. In both bacteria and fungi, repair of DNA double-strand breaks may occur through non-homologous end joining, where cut ends are re-ligated in a manner that may be mutagenic, or through homologous recombination, using a homologous sequence as a template for repair [18,19].
Antimicrobial-Induced Single Nucleotide Mutagenesis
As a result of antimicrobial-induced DNA damage and repair discussed above, as well as additional stress-response pathways, microbial species may experience genomic instability. One example of this is an increase in the number of single nucleotide polymorphisms (SNPs) in response to antimicrobial treatment (Fig. 1). The mechanisms by which this occurs can broadly be categorized into DNA damage response pathways, and other stress response signaling pathways.
In bacterial species, one important pathway that mediates antimicrobial-induced mutagenesis is the DNA damage-induced SOS response. Sublethal doses of diverse classes of antibiotics, including aminoglycosides and quinolones, lead to a cellular SOS response in many bacterial species [20,21]. As described above, quinolones induce DNA damage through interaction with DNA topoisomerase, and thus elicit a bacterial SOS response. In Escherichia coli, quinolone-induced SOS leads to derepression of polymerases Pol II, Pol IV, and Pol V, which together introduce mutations into the genome [20,22]. Other classes of antibiotics, such as aminoglycosides, stimulate an SOS response as a result of drug-induced oxidative DNA damage [23]. In Vibrio cholerae and Klebsiella pneumoniae, SOS-mediated depletion of base excision repair factors such as the mismatch repair protein MutY likely leads to antibiotic-induced mutagenesis upon treatment with aminoglycosides [21,23].
Other general stress response pathways have also been implicated in antimicrobial-induced mutations. In bacteria, the RpoS sigma factor is a central regulator of the general stress response, which is activated in response to stress conditions. In E. coli, Pseudomonas aerigunosa, and V. cholerae, different classes of antimicrobial agents induce RpoS [23,24]. This leads to activation of the error-prone Pol IV polymerase and down-regulation of accurate DNA repair activity via the mismatch repair protein MutS, thus promoting mutations [24]. Up-regulation of general stress response pathways similarly mediate stress-induced mutations in fungal species. In the model yeast Saccharomyces cerevisiae, stress triggers an environmental stress response pathway, mediated through transcriptional regulators Msn2 and Msn4. Similar to what is observed in bacteria, these transcription factors activate downstream error-prone translesion synthesis via the Rev1 polymerase, thus increasing mutagenesis [25]. While antifungal-induced SNP mutagenesis has not been well documented in fungal pathogens, analysis of S. cerevisiae, with conserved regulatory machinery with pathogenic fungi [26], may provide novel mechanistic insight for fungal pathogens. For both bacterial and fungal pathogens, antimicrobial-induced mutagenesis has the capability to accelerate the acquisition of drug resistance and multi-drug resistance by increasing genetic and phenotypic diversity within the population [27], with important consequences for clinical use of antibiotics.
Large-Scale Genomic Alterations Induced by Antimicrobial Treatment
In addition to nucleotide mutagenesis, treatment with sublethal antimicrobial agents can also promote larger-scale genomic rearrangements in microbial pathogens. This includes movement of mobile genetic elements, chromosomal rearrangements, and whole chromosome aneuploidies. Such large-scale alterations highlight the difference between bacterial and fungal pathogens. Bacteria are able to exchange genetic information between individual cells via horizontal gene transfer, which occurs far more rarely amongst fungal pathogens [28]. Further, while the genetic material of bacterial pathogens is contained within a limited number of circular chromosomes and plasmids, fungal pathogens typically have several linear chromosomes, and may exist in haploid or diploid states [29]. Such differences in chromosomal number, ploidy, and replication are reflected in the forms of genomic alterations that occur in these pathogens upon antimicrobial treatment.
In bacteria, antimicrobial treatment can trigger the movement of mobile genetic elements [1]. For Staphylococcus aureus bacteria, treatment with subinhibitory concentrations of quinolone antibiotics leads to up-regulation of the LexA-dependent SOS response, resulting in increased transposition of the IS256 transposable insertion element [30]. Similarly, antibiotic-induced SOS mediates the movement of integrating conjugative elements (ICEs), a group of bacterial mobile genetic elements that integrate into the chromosome and transfer between cells during conjugation [31]. In V. cholerae bacteria, sublethal doses of quinolone antibiotic induces an SOS response, which increases the expression of genes necessary for ICE transfer, and thus the frequency of conjugative transfers of this mobile element [31]. As many ICEs encode antibiotic resistance determinants, antibiotic-induced transposition between cells may promote the spread of antibiotic resistance genes [31]. Antibiotics can also stimulate the movement of mobile elements indirectly, by increasing cellular competence [32,33]. For Streptococcus pneumoniae, antibiotic-induced genomic replication stress results in stalled replication forks, while DNA replication initiation proceeds [32]. This results in an amplification and overexpression of genes in proximity to the origin of replication, including factors involved in natural cellular competence [32]. This increase in competence and genetic transformability facilitates the acquisition of antibiotic resistance by allowing these pathogens to more readily uptake DNA, including antibiotic resistance determinants, from their environment [32]. Thus antibiotic treatment can both induce movement of antibiotic-encoding mobile elements, and stimulate cellular competence, which together can strongly promote the acquisition and spread of genetic resistance determinants within populations.
Although horizontal gene transfer is rarely observed amongst human fungal pathogens, treatment with antifungals can promote alternative forms of genomic instability, via gross chromosomal rearrangements. The antifungal fluconazole, which targets fungal membrane integrity, also leads to the up-regulation of cellular stress response pathways [29], and promotes genomic rearrangements [4]. For the diploid fungal pathogen Candida albicans, sublethal doses of fluconazole promote increased rates of loss of heterozygosity (LOH) [34], a form of gross chromosomal rearrangement in diploid organisms that results in the loss of genetic heterozygosity at a particular locus or throughout an entire chromosome (Fig. 1). Furthermore, C. albicans exposed to antifungal stress promotes the formation of isochromosomes, in which entire chromosome arms are exchanged, creating a chromosome comprised of two identical chromosome arms flanking a centromere [35]. Although the mechanism of antifungal-mediated chromosomal alterations is unknown, it has been suggested that DNA double-strand breaks induced by antifungal agents [34,36] and repaired via recombination between chromosomes, may contribute to such genomic rearrangements. Both LOH and isochromosomes play an important role in acquired resistance to antifungals in C. albicans, through homozygosis and duplication of genes encoding both the drug target of the azoles (ergosterol biosynthesis enzyme Erg11), and regulators of drug efflux [4]. Duplication and thus overexpression of Erg11 reduces the efficacy of the azole drugs and promotes resistance, while duplication of transcriptional regulators of drug efflux pumps (such as Tac1 and Mrr1), may promote multi-drug resistance by increasing the efficacy by which antifungals are exported from the cell [4].
Finally, several eukaryotic pathogens have especially plastic genomes, and readily become aneuploid via entire chromosome gains or losses under antimicrobial stress conditions. As aneuploidy results from errors in mitotic cell division, differences between eukaryotic mitosis and binary fission in prokaryotic bacteria, likely accounts for this phenomenon in eukaryotic pathogens. Fungal pathogens, including C. albicans and Cryptococcus neoformans, have particularly flexible genomes [4,37], and aneuploid lineages of these pathogens are frequently identified both in the laboratory, and amongst clinical isolates [38]. The antifungal agent fluconazole induces the formation of aneuploidies in C. albicans [35,39], and chromosome disomies in C. neoformans [40,41] (Fig. 1), both of which are linked with the development of antifungal drug resistance from increased copy numbers of key antifungal resistance determinants, including antifungal target proteins and drug transporters [40]. In Candida species, this stress-induced aneuploidy occurs from aberrant mitosis due to antifungal stress, resulting in the formation of tetraploid cells, and unequal chromosomal segregation [42]. In S. cerevisiae, stress-induced aneuploidies occur under diverse stress conditions, including low-dose antifungal treatment, and are linked to protein chaperone Hsp90-mediated disruption of the kinetochore complex, leading to chromosomal instability [43]. Antimicrobial-induced genomic instability leading the chromosomal aneuploidies, including those associated with drug resistance, is a unique way in which fungal pathogens adapt to antimicrobial stress conditions.
Conclusions
Antimicrobial-induced DNA damage and genomic instability occurs across the diversity of bacterial and fungal pathogens. However, the types of genetic alterations vary between these prokaryotic and eukaryotic pathogens, which differ in their genomic composition, as well as mechanisms of genetic replication and cell division. For instance, although diploid fungal pathogens such as C. albicans may be buffered against the effects of certain mutations, they are more likely to undergo aneuploidy or LOH events between homologous chromosome pairs. Despite these differences, genetic alterations that are advantageous, including direct genetic alterations that confer antimicrobial resistance, as well as indirect alterations such as increased cellular competence, may facilitate pathogen survival in the face of antimicrobial stress.
The scale of stress-induced genomic alterations, from SNPs to whole chromosome aneuploidy, likely has varying degrees of phenotypic consequences for microbial pathogens. For many fungal pathogens, which unlike bacteria, cannot increase their genetic diversity through horizontal gene transfer, and which rarely undergo sexual reproduction for genetic recombination [44], large-scale chromosomal rearrangements and aneuploidies may provide a unique mechanism to rapidly generate genetic diversity and adapt to their environments under conditions of stress. This mechanism may further extend to other eukaryotic pathogens, such as the trypanosomal parasite Leishmania. Like fungal pathogens, stress-induced aneuploidies occur in Leishmania in the presence of antiparasitic drugs, potentially as a result of known roles for certain anti-trypanosomal drugs in spindle apparatus formation and chromosome segregation [45]. This suggests that genome plasticity may be conserved across diverse eukaryotic pathogens, and may provide a distinctive mechanism for stress adaptation.
As stress-induced mutation provides a mechanism for microbial pathogens to develop resistance, it is critical to understand how antimicrobial therapeutics may enhance or limit pathogen evolvability. One therapeutic strategy to limit acquired drug resistance is to target the pathogen response to antimicrobials [19]. For instance, preventing SOS induction by targeting central SOS regulators such as the protease LexA can prevent mutations and the evolution of antibiotic drug resistance in E. coli [22,46]. Similarly, quinolone antibiotics do not induce mutations in Salmonella typhimurium strains lacking the Pol V homolog [47]. Additionally, new research has identified certain antimicrobial peptides that, unlike antibiotics, do not elicit an SOS response or increase bacterial mutation rate [48]. This finding suggests promising avenues for identifying novel antimicrobial agents that do not expedite the evolution of antimicrobial resistance.
Zdroje
1. Andersson DI, Hughes D (2014) Microbiological effects of sublethal levels of antibiotics. Nature reviews Microbiology 12 : 465–478. doi: 10.1038/nrmicro3270 24861036
2. Müller M, Peña dela A, Derendorf H (2004) Issues in pharmacokinetics and pharmacodynamics of anti-infective agents: distribution in tissue. Antimicrobial agents and chemotherapy 48 : 1441–1453. 15105091
3. Looft T, Johnson TA, Allen HK, Bayles DO, Alt DP, et al. (2012) In-feed antibiotic effects on the swine intestinal microbiome. Proc Natl Acad Sci U S A 109 : 1691–1696. doi: 10.1073/pnas.1120238109 22307632
4. Selmecki A, Forche A, Berman J (2010) Genomic plasticity of the human fungal pathogen Candida albicans. Eukaryotic cell 9 : 991–1008. doi: 10.1128/EC.00060-10 20495058
5. Hecht SM (2000) Bleomycin: new perspectives on the mechanism of action. J Nat Prod 63 : 158–168. 10650103
6. Hawkey PM (2003) Mechanisms of quinolone action and microbial response. The Journal of antimicrobial chemotherapy 51 Suppl 1 : 29–35. 12702701
7. Dwyer DJ, Belenky PA, Yang JH, Macdonald IC, Martell JD, et al. (2014) Antibiotics induce redox-related physiological alterations as part of their lethality. Proc Natl Acad Sci U S A 111: E2100–E2109. doi: 10.1073/pnas.1401876111 24803433
8. Dwyer DJ, Collins JJ, Walker GC (2014) Unraveling the Physiological Complexities of Antibiotic Lethality. Annu Rev Pharmacol Toxicol 55 : 313–32. doi: 10.1146/annurev-pharmtox-010814-124712 25251995
9. Zhao X, Drlica K (2014) Reactive oxygen species and the bacterial response to lethal stress. Current opinion in microbiology 21C: 1–6.
10. Belenky P, Camacho D, Collins JJ (2013) Fungicidal drugs induce a common oxidative-damage cellular death pathway. Cell Rep 3 : 350–358. doi: 10.1016/j.celrep.2012.12.021 23416050
11. Mesa-Arango AC, Trevijano-Contador N, Román E, Sánchez-Fresneda R, Casas C, et al. (2014) The production of reactive oxygen species is an universal action mechanism of Amphotericin B against pathogenic yeasts and contributes to the fungicidal effect of this drug. Antimicrobial agents and chemotherapy 58 : 6627–38. doi: 10.1128/AAC.03570-14 25155595
12. Moreira W, Leprohon P, Ouellette M (2011) Tolerance to drug-induced cell death favours the acquisition of multidrug resistance in Leishmania. Cell Death Dis 2: e201. doi: 10.1038/cddis.2011.83 21881603
13. Blázquez J, Couce A, Rodríguez-Beltrán J, Rodríguez-Rojas A (2012) Antimicrobials as promoters of genetic variation. Current opinion in microbiology 15 : 561–569. doi: 10.1016/j.mib.2012.07.007 22890188
14. Foti JJ, Devadoss B, Winkler JA, Collins JJ, Walker GC (2012) Oxidation of the Guanine Nucleotide Pool Underlies Cell Death by Bactericidal Antibiotics. Science 336 : 315–319. doi: 10.1126/science.1219192 22517853
15. Rajagopalan M, Lu C, Woodgate R, O'Donnell M, Goodman MF, et al. (1992) Activity of the purified mutagenesis proteins UmuC, UmuD', and RecA in replicative bypass of an abasic DNA lesion by DNA polymerase III. Proceedings of the National Academy of Sciences of the United States of America 89 : 10777–10781. 1438275
16. Brent R, Ptashne M (1981) Mechanism of action of the lexA gene product. Proceedings of the National Academy of Sciences of the United States of America 78 : 4204–4208. 7027256
17. Wagner J, Gruz P, Kim SR, Yamada M, Matsui K, et al. (1999) The dinB gene encodes a novel E. coli DNA polymerase, DNA pol IV, involved in mutagenesis. Mol Cell 4 : 281–286. 10488344
18. Boiteux S, Jinks-Robertson S (2013) DNA repair mechanisms and the bypass of DNA damage in Saccharomyces cerevisiae. Genetics 193 : 1025–1064. doi: 10.1534/genetics.112.145219 23547164
19. Rosenberg SM, Shee C, Frisch RL, Hastings PJ (2012) Stress-induced mutation via DNA breaks in Escherichia coli: a molecular mechanism with implications for evolution and medicine. Bioessays 34 : 885–892. doi: 10.1002/bies.201200050 22911060
20. Thi TD Lopez E, Rodriguez-Rojas A, Rodriguez-Beltran JON, Couce A, et al. (2011) Effect of recA inactivation on mutagenesis of Escherichia coli exposed to sublethal concentrations of antimicrobials. The Journal of antimicrobial chemotherapy 66 : 531–538. doi: 10.1093/jac/dkq496 21212055
21. Baharoglu Z, Mazel D (2011) Vibrio cholerae triggers SOS and mutagenesis in response to a wide range of antibiotics: a route towards multiresistance. Antimicrobial agents and chemotherapy 55 : 2438–2441. doi: 10.1128/AAC.01549-10 21300836
22. Cirz RT, Chin JK, Andes DR, de Crécy-Lagard V, Craig WA, et al. (2005) Inhibition of mutation and combating the evolution of antibiotic resistance. PLoS Biol 3: e176. 15869329
23. Baharoglu Z, Krin E, Mazel D (2013) RpoS plays a central role in the SOS induction by sub-lethal aminoglycoside concentrations in Vibrio cholerae. PLoS Genet 9: e1003421. doi: 10.1371/journal.pgen.1003421 23613664
24. Gutierrez A, Laureti L, Crussard S, Abida H, Rodr i guez-Rojas A, et al. (2013) β-lactam antibiotics promote bacterial mutagenesis via an RpoS-mediated reduction in replication fidelity. Nature communications 4 : 1610. doi: 10.1038/ncomms2607 23511474
25. Shor E, Fox CA, Broach JR (2013) The yeast environmental stress response regulates mutagenesis induced by proteotoxic stress. PLoS Genet 9: e1003680. doi: 10.1371/journal.pgen.1003680 23935537
26. Ryan O, Shapiro RS, Kurat CF, Mayhew D, Baryshnikova A, et al. (2012) Global gene deletion analysis exploring yeast filamentous growth. Science (New York, NY) 337 : 1353–1356. doi: 10.1126/science.1224339 22984072
27. Kohanski MA, Depristo MA, Collins JJ (2010) Sublethal Antibiotic Treatment Leads to Multidrug Resistance via Radical-Induced Mutagenesis. Molecular Cell 37 : 311–320. doi: 10.1016/j.molcel.2010.01.003 20159551
28. Fitzpatrick DA (2012) Horizontal gene transfer in fungi. FEMS Microbiol Lett 329 : 1–8. doi: 10.1111/j.1574-6968.2011.02465.x 22112233
29. Shapiro RS, Robbins N, Cowen LE (2011) Regulatory circuitry governing fungal development, drug resistance, and disease. Microbiol Mol Biol Rev 75 : 213–267. doi: 10.1128/MMBR.00045-10 21646428
30. Nagel M, Reuter T, Jansen A, Szekat C, Bierbaum G (2011) Influence of ciprofloxacin and vancomycin on mutation rate and transposition of IS256 in Staphylococcus aureus. Int J Med Microbiol 301 : 229–236. doi: 10.1016/j.ijmm.2010.08.021 21115395
31. Beaber JW, Hochhut B, Waldor MK (2004) SOS response promotes horizontal dissemination of antibiotic resistance genes. Nature 427 : 72–74. 14688795
32. Slager J, Kjos M, Attaiech L, Veening J - W (2014) Antibiotic-Induced Replication Stress Triggers Bacterial Competence by Increasing Gene Dosage near the Origin. Cell 157 : 395–406. doi: 10.1016/j.cell.2014.01.068 24725406
33. Prudhomme M, Attaiech L, Sanchez G, Martin B, Claverys J - P (2006) Antibiotic stress induces genetic transformability in the human pathogen Streptococcus pneumoniae. Science (New York, NY) 313 : 89–92. 16825569
34. Forche A, Abbey D, Pisithkul T (2011) Stress Alters Rates and Types of Loss of Heterozygosity in Candida albicans 2: pii: e00129–11. doi: 10.1128/mBio.00129-11 21791579
35. Selmecki AM, Dulmage K, Cowen LE, Anderson JB, Berman J (2009) Acquisition of aneuploidy provides increased fitness during the evolution of antifungal drug resistance. PLoS Genet 5: e1000705. doi: 10.1371/journal.pgen.1000705 19876375
36. Legrand M, Chan CL, Jauert PA, Kirkpatrick DT (2007) Role of DNA mismatch repair and double-strand break repair in genome stability and antifungal drug resistance in Candida albicans. Eukaryotic cell 6 : 2194–2205. 17965250
37. Ormerod KL, Fraser JA (2013) Balancing stability and flexibility within the genome of the pathogen Cryptococcus neoformans. PLoS pathogens 9: e1003764. doi: 10.1371/journal.ppat.1003764 24348244
38. Kwon-Chung KJ, Chang YC (2012) Aneuploidy and drug resistance in pathogenic fungi. PLoS pathogens 8: e1003022. doi: 10.1371/journal.ppat.1003022 23166494
39. Chang F - M, Ou T - Y, Cheng W - N, Chou M - L, Lee K - C, et al. (2014) Short-term exposure to fluconazole induces chromosome loss in Candida albicans: An approach to produce haploid cells. Fungal Genet Biol 70 : 68–76. doi: 10.1016/j.fgb.2014.06.009 25038494
40. Sionov E, Lee H, Chang YC, Kwon-Chung KJ (2010) Cryptococcus neoformans overcomes stress of azole drugs by formation of disomy in specific multiple chromosomes. PLoS pathogens 6: e1000848. doi: 10.1371/journal.ppat.1000848 20368972
41. Semighini CP, Averette AF, Perfect JR, Heitman J (2011) Deletion of Cryptococcus neoformans AIF ortholog promotes chromosome aneuploidy and fluconazole-resistance in a metacaspase-independent manner. PLoS pathogens 7: e1002364. doi: 10.1371/journal.ppat.1002364 22114551
42. Harrison BD, Hashemi J, Bibi M, Pulver R, Bavli D, et al. (2014) A tetraploid intermediate precedes aneuploid formation in yeasts exposed to fluconazole. PLoS Biol 12: e1001815. doi: 10.1371/journal.pbio.1001815 24642609
43. Chen G, Bradford WD, Seidel CW, Li R (2012) Hsp90 stress potentiates rapid cellular adaptation through induction of aneuploidy. Nature 482 : 246–50. doi: 10.1038/nature10795 22286062
44. Ene IV, Bennett RJ (2014) The cryptic sexual strategies of human fungal pathogens. Nature reviews Microbiology 12 : 239–251. doi: 10.1038/nrmicro3236 24625892
45. Mannaert A, Downing T, Imamura H, Dujardin J - C (2012) Adaptive mechanisms in pathogens: universal aneuploidy in Leishmania. Trends Parasitol 28 : 370–376. doi: 10.1016/j.pt.2012.06.003 22789456
46. Lu TK, Collins JJ (2009) Engineered bacteriophage targeting gene networks as adjuvants for antibiotic therapy. Proc Natl Acad Sci U S A 106 : 4629–4634. doi: 10.1073/pnas.0800442106 19255432
47. Ysern P, Clerch B, Castańo M, Gibert I, Barbé J, et al. (1990) Induction of SOS genes in Escherichia coli and mutagenesis in Salmonella typhimurium by fluoroquinolones. Mutagenesis 5 : 63–66. 2158613
48. Rodríguez-Rojas A, Makarova O, Rolff J (2014) Antimicrobials, Stress and Mutagenesis. PLoS pathogens 10: e1004445. doi: 10.1371/journal.ppat.1004445 25299705
Štítky
Hygiena a epidemiologie Infekční lékařství Laboratoř
Článek A Phospholipase Is Involved in Disruption of the Liver Stage Parasitophorous Vacuole MembraneČlánek Host ESCRT Proteins Are Required for Bromovirus RNA Replication Compartment Assembly and FunctionČlánek Enhanced CD8 T Cell Responses through GITR-Mediated Costimulation Resolve Chronic Viral Infection
Článek vyšel v časopisePLOS Pathogens
Nejčtenější tento týden
2015 Číslo 3- Měli bychom postcovidový syndrom léčit antidepresivy?
- Farmakovigilanční studie perorálních antivirotik indikovaných v léčbě COVID-19
- Jak souvisí postcovidový syndrom s poškozením mozku?
- 10 bodů k očkování proti COVID-19: stanovisko České společnosti alergologie a klinické imunologie ČLS JEP
-
Všechny články tohoto čísla
- To Be or Not IIb: A Multi-Step Process for Epstein-Barr Virus Latency Establishment and Consequences for B Cell Tumorigenesis
- Is Antigenic Sin Always “Original?” Re-examining the Evidence Regarding Circulation of a Human H1 Influenza Virus Immediately Prior to the 1918 Spanish Flu
- The Great Escape: Pathogen Versus Host
- Coping with Stress and the Emergence of Multidrug Resistance in Fungi
- Catch Me If You Can: The Link between Autophagy and Viruses
- Bacterial Immune Evasion through Manipulation of Host Inhibitory Immune Signaling
- Evidence for Ubiquitin-Regulated Nuclear and Subnuclear Trafficking among Matrix Proteins
- BILBO1 Is a Scaffold Protein of the Flagellar Pocket Collar in the Pathogen
- Production of Anti-LPS IgM by B1a B Cells Depends on IL-1β and Is Protective against Lung Infection with LVS
- Virulence Regulation with Venus Flytrap Domains: Structure and Function of the Periplasmic Moiety of the Sensor-Kinase BvgS
- α-Hemolysin Counteracts the Anti-Virulence Innate Immune Response Triggered by the Rho GTPase Activating Toxin CNF1 during Bacteremia
- Induction of Interferon-Stimulated Genes by IRF3 Promotes Replication of
- Intracellular Growth Is Dependent on Tyrosine Catabolism in the Dimorphic Fungal Pathogen
- HCV Induces the Expression of Rubicon and UVRAG to Temporally Regulate the Maturation of Autophagosomes and Viral Replication
- Spatiotemporal Analysis of Hepatitis C Virus Infection
- Subgingival Microbial Communities in Leukocyte Adhesion Deficiency and Their Relationship with Local Immunopathology
- Interaction between the Type III Effector VopO and GEF-H1 Activates the RhoA-ROCK Pathway
- Attenuation of Tick-Borne Encephalitis Virus Using Large-Scale Random Codon Re-encoding
- Establishment of HSV1 Latency in Immunodeficient Mice Facilitates Efficient Reactivation
- XRN1 Stalling in the 5’ UTR of Hepatitis C Virus and Bovine Viral Diarrhea Virus Is Associated with Dysregulated Host mRNA Stability
- γδ T Cells Confer Protection against Murine Cytomegalovirus (MCMV)
- Rhadinovirus Host Entry by Co-operative Infection
- A Phospholipase Is Involved in Disruption of the Liver Stage Parasitophorous Vacuole Membrane
- Dermal Neutrophil, Macrophage and Dendritic Cell Responses to Transmitted by Fleas
- Elucidation of Sigma Factor-Associated Networks in Reveals a Modular Architecture with Limited and Function-Specific Crosstalk
- A Conserved NS3 Surface Patch Orchestrates NS2 Protease Stimulation, NS5A Hyperphosphorylation and HCV Genome Replication
- Host ESCRT Proteins Are Required for Bromovirus RNA Replication Compartment Assembly and Function
- Disruption of IL-21 Signaling Affects T Cell-B Cell Interactions and Abrogates Protective Humoral Immunity to Malaria
- Compartmentalized Replication of R5 T Cell-Tropic HIV-1 in the Central Nervous System Early in the Course of Infection
- Diminished Reovirus Capsid Stability Alters Disease Pathogenesis and Littermate Transmission
- Characterization of CD8 T Cell Differentiation following SIVΔnef Vaccination by Transcription Factor Expression Profiling
- Visualization of HIV-1 Interactions with Penile and Foreskin Epithelia: Clues for Female-to-Male HIV Transmission
- Sensing Cytosolic RpsL by Macrophages Induces Lysosomal Cell Death and Termination of Bacterial Infection
- PKCη/Rdx-driven Phosphorylation of PDK1: A Novel Mechanism Promoting Cancer Cell Survival and Permissiveness for Parvovirus-induced Lysis
- Metalloprotease NleC Suppresses Host NF-κB/Inflammatory Responses by Cleaving p65 and Interfering with the p65/RPS3 Interaction
- Immune Antibodies and Helminth Products Drive CXCR2-Dependent Macrophage-Myofibroblast Crosstalk to Promote Intestinal Repair
- Adenovirus Entry From the Apical Surface of Polarized Epithelia Is Facilitated by the Host Innate Immune Response
- The RNA Template Channel of the RNA-Dependent RNA Polymerase as a Target for Development of Antiviral Therapy of Multiple Genera within a Virus Family
- Neutrophils: Between Host Defence, Immune Modulation, and Tissue Injury
- CD169-Mediated Trafficking of HIV to Plasma Membrane Invaginations in Dendritic Cells Attenuates Efficacy of Anti-gp120 Broadly Neutralizing Antibodies
- Japanese Encephalitis Virus Nonstructural Protein NS5 Interacts with Mitochondrial Trifunctional Protein and Impairs Fatty Acid β-Oxidation
- Yip1A, a Novel Host Factor for the Activation of the IRE1 Pathway of the Unfolded Protein Response during Infection
- TRIM26 Negatively Regulates Interferon-β Production and Antiviral Response through Polyubiquitination and Degradation of Nuclear IRF3
- Parallel Epigenomic and Transcriptomic Responses to Viral Infection in Honey Bees ()
- A Crystal Structure of the Dengue Virus NS5 Protein Reveals a Novel Inter-domain Interface Essential for Protein Flexibility and Virus Replication
- Enhanced CD8 T Cell Responses through GITR-Mediated Costimulation Resolve Chronic Viral Infection
- Exome and Transcriptome Sequencing of Identifies a Locus That Confers Resistance to and Alters the Immune Response
- The Role of Misshapen NCK-related kinase (MINK), a Novel Ste20 Family Kinase, in the IRES-Mediated Protein Translation of Human Enterovirus 71
- Chitin Recognition via Chitotriosidase Promotes Pathologic Type-2 Helper T Cell Responses to Cryptococcal Infection
- Activates Both IL-1β and IL-1 Receptor Antagonist to Modulate Lung Inflammation during Pneumonic Plague
- Persistence of Transmitted HIV-1 Drug Resistance Mutations Associated with Fitness Costs and Viral Genetic Backgrounds
- An 18 kDa Scaffold Protein Is Critical for Biofilm Formation
- Early Virological and Immunological Events in Asymptomatic Epstein-Barr Virus Infection in African Children
- Human CD8 T-cells Recognizing Peptides from () Presented by HLA-E Have an Unorthodox Th2-like, Multifunctional, Inhibitory Phenotype and Represent a Novel Human T-cell Subset
- Decreased HIV-Specific T-Regulatory Responses Are Associated with Effective DC-Vaccine Induced Immunity
- RSV Vaccine-Enhanced Disease Is Orchestrated by the Combined Actions of Distinct CD4 T Cell Subsets
- Concerted Activity of IgG1 Antibodies and IL-4/IL-25-Dependent Effector Cells Trap Helminth Larvae in the Tissues following Vaccination with Defined Secreted Antigens, Providing Sterile Immunity to Challenge Infection
- Structure of the Low pH Conformation of Chandipura Virus G Reveals Important Features in the Evolution of the Vesiculovirus Glycoprotein
- PPM1A Regulates Antiviral Signaling by Antagonizing TBK1-Mediated STING Phosphorylation and Aggregation
- Lipidomic Analysis Links Mycobactin Synthase K to Iron Uptake and Virulence in .
- Roles and Programming of Arabidopsis ARGONAUTE Proteins during Infection
- Impact of Infection on Host Macrophage Nuclear Physiology and Nucleopore Complex Integrity
- The Impact of Host Diet on Titer in
- Antimicrobial-Induced DNA Damage and Genomic Instability in Microbial Pathogens
- Herpesviral G Protein-Coupled Receptors Activate NFAT to Induce Tumor Formation via Inhibiting the SERCA Calcium ATPase
- The Causes and Consequences of Changes in Virulence following Pathogen Host Shifts
- Small GTPase Rab21 Mediates Fibronectin Induced Actin Reorganization in : Implications in Pathogen Invasion
- Positive Role of Promyelocytic Leukemia Protein in Type I Interferon Response and Its Regulation by Human Cytomegalovirus
- NEDDylation Is Essential for Kaposi’s Sarcoma-Associated Herpesvirus Latency and Lytic Reactivation and Represents a Novel Anti-KSHV Target
- β-HPV 5 and 8 E6 Disrupt Homology Dependent Double Strand Break Repair by Attenuating BRCA1 and BRCA2 Expression and Foci Formation
- An O Antigen Capsule Modulates Bacterial Pathogenesis in
- Variable Processing and Cross-presentation of HIV by Dendritic Cells and Macrophages Shapes CTL Immunodominance and Immune Escape
- Probing the Metabolic Network in Bloodstream-Form Using Untargeted Metabolomics with Stable Isotope Labelled Glucose
- Adhesive Fiber Stratification in Uropathogenic Biofilms Unveils Oxygen-Mediated Control of Type 1 Pili
- Vaccinia Virus Protein Complex F12/E2 Interacts with Kinesin Light Chain Isoform 2 to Engage the Kinesin-1 Motor Complex
- Modulates Host Macrophage Mitochondrial Metabolism by Hijacking the SIRT1-AMPK Axis
- Human T-Cell Leukemia Virus Type 1 (HTLV-1) Tax Requires CADM1/TSLC1 for Inactivation of the NF-κB Inhibitor A20 and Constitutive NF-κB Signaling
- Suppression of RNAi by dsRNA-Degrading RNaseIII Enzymes of Viruses in Animals and Plants
- Spatiotemporal Regulation of a T4SS Substrate by the Metaeffector SidJ
- Antigenic Properties of the Human Immunodeficiency Virus Envelope Glycoprotein Gp120 on Virions Bound to Target Cells
- Dependence of Intracellular and Exosomal microRNAs on Viral Oncogene Expression in HPV-positive Tumor Cells
- Identification of a Peptide-Pheromone that Enhances Escape from Host Cell Vacuoles
- Impaired Systemic Tetrahydrobiopterin Bioavailability and Increased Dihydrobiopterin in Adult Falciparum Malaria: Association with Disease Severity, Impaired Microvascular Function and Increased Endothelial Activation
- Transgenic Expression of the Dicotyledonous Pattern Recognition Receptor EFR in Rice Leads to Ligand-Dependent Activation of Defense Responses
- Comprehensive Antigenic Map of a Cleaved Soluble HIV-1 Envelope Trimer
- Low Doses of Imatinib Induce Myelopoiesis and Enhance Host Anti-microbial Immunity
- Impaired Systemic Tetrahydrobiopterin Bioavailability and Increased Oxidized Biopterins in Pediatric Falciparum Malaria: Association with Disease Severity
- PLOS Pathogens
- Archiv čísel
- Aktuální číslo
- Informace o časopisu
Nejčtenější v tomto čísle- Bacterial Immune Evasion through Manipulation of Host Inhibitory Immune Signaling
- BILBO1 Is a Scaffold Protein of the Flagellar Pocket Collar in the Pathogen
- Antimicrobial-Induced DNA Damage and Genomic Instability in Microbial Pathogens
- Attenuation of Tick-Borne Encephalitis Virus Using Large-Scale Random Codon Re-encoding
Kurzy
Zvyšte si kvalifikaci online z pohodlí domova
Současné možnosti léčby obezity
nový kurzAutoři: MUDr. Martin Hrubý
Všechny kurzyPřihlášení#ADS_BOTTOM_SCRIPTS#Zapomenuté hesloZadejte e-mailovou adresu, se kterou jste vytvářel(a) účet, budou Vám na ni zaslány informace k nastavení nového hesla.
- Vzdělávání



