-
Články
- Vzdělávání
- Časopisy
Top články
Nové číslo
- Témata
- Videa
- Podcasty
Nové podcasty
Reklama- Kariéra
Doporučené pozice
Reklama- Praxe
Disruption of Lipid Metabolism Genes Causes Tissue Overgrowth Associated with Altered Developmental Signaling
Developmental patterning requires the precise interplay of numerous intercellular signaling pathways to ensure that cells are properly specified during tissue formation and organogenesis. The spatiotemporal function of many developmental pathways is strongly influenced by the biosynthesis and intracellular trafficking of signaling components. Receptors and ligands must be trafficked to the cell surface where they interact, and their subsequent endocytic internalization and endosomal trafficking is critical for both signal propagation and its down-modulation. In a forward genetic screen for mutations that alter intracellular Notch receptor trafficking in Drosophila melanogaster, we recovered mutants that disrupt genes encoding serine palmitoyltransferase and acetyl-CoA carboxylase. Both mutants cause Notch, Wingless, the Epidermal Growth Factor Receptor (EFGR), and Patched to accumulate abnormally in endosomal compartments. In mosaic animals, mutant tissues exhibit an unusual non-cell-autonomous effect whereby mutant cells are functionally rescued by secreted activities emanating from adjacent wildtype tissue. Strikingly, both mutants display prominent tissue overgrowth phenotypes that are partially attributable to altered Notch and Wnt signaling. Our analysis of the mutants demonstrates genetic links between abnormal lipid metabolism, perturbations in developmental signaling, and aberrant cell proliferation.
Published in the journal: . PLoS Genet 9(11): e32767. doi:10.1371/journal.pgen.1003917
Category: Research Article
doi: https://doi.org/10.1371/journal.pgen.1003917Summary
Developmental patterning requires the precise interplay of numerous intercellular signaling pathways to ensure that cells are properly specified during tissue formation and organogenesis. The spatiotemporal function of many developmental pathways is strongly influenced by the biosynthesis and intracellular trafficking of signaling components. Receptors and ligands must be trafficked to the cell surface where they interact, and their subsequent endocytic internalization and endosomal trafficking is critical for both signal propagation and its down-modulation. In a forward genetic screen for mutations that alter intracellular Notch receptor trafficking in Drosophila melanogaster, we recovered mutants that disrupt genes encoding serine palmitoyltransferase and acetyl-CoA carboxylase. Both mutants cause Notch, Wingless, the Epidermal Growth Factor Receptor (EFGR), and Patched to accumulate abnormally in endosomal compartments. In mosaic animals, mutant tissues exhibit an unusual non-cell-autonomous effect whereby mutant cells are functionally rescued by secreted activities emanating from adjacent wildtype tissue. Strikingly, both mutants display prominent tissue overgrowth phenotypes that are partially attributable to altered Notch and Wnt signaling. Our analysis of the mutants demonstrates genetic links between abnormal lipid metabolism, perturbations in developmental signaling, and aberrant cell proliferation.
Introduction
Developmental patterning in metazoans requires the coordinated activity of several intercellular signaling pathways. In D. melanogaster, Notch and Wnt signaling are critical for the formation of diverse organs and tissues, and both pathways also regulate cell proliferation and apoptosis during development [1]–[4]. Notch signaling is activated by binding of DSL ligands to the Notch receptor, which induces proteolytic cleavage of Notch by ADAM/TACE metalloproteases and subsequent cleavage by gamma-secretase to generate the Notch intracellular signaling fragment NICD [1], [2]. NICD translocates to the nucleus where it regulates target gene expression by displacing co-repressors from transcriptional complexes and converting them into active complexes [5], [6].
Notch signaling is strongly modulated by additional posttranslational mechanisms, including glycosylation, ubiquitylation, and endosomal trafficking [2], [7]. Endocytosis of Notch and its ligands in both signal-receiving and signal-sending cells is required for productive signaling [8], generating tensile forces needed to expose the ADAM/TACE cleavage site in Notch and facilitate receptor proteolysis [9]. Several mutants with impaired trafficking of ligand-activated Notch are associated with reduced signaling, while others that perturb trafficking of non-activated Notch exhibit Notch hyperactivation and tissue overgrowth [10]–[14]. Intracellular Notch trafficking thus regulates both signal activation and the degradation of inactive Notch receptors that might otherwise contribute to inappropriate signaling. Moreover, this endocytic regulation of Notch signaling is strongly influenced by its membrane lipid microenvironment; mutations in D. melanogaster phosphocholine cytidylyltransferase alter Notch endosomal routing and activation [15] and mutations in D. melanogaster alpha-1,4-N-acetylgalactosaminyltransferase-1 affect endocytosis and potency of the Notch ligands Delta and Serrate [16].
Canonical Wnt/Wingless signaling is similarly needed for a diverse array of tissue patterning processes and is also influenced by membrane trafficking [3], [4]. Wnt, a secreted ligand, binds to receptors of the Frizzled and LRP/Arrow families, leading to recruitment of Disheveled and stabilization of ß-catenin/Armadillo. Accumulation of ß-catenin/Armadillo in turn triggers downstream gene activation through TCF transcription factors [17], [18]. Wnt proteins are modified by lipid attachment [19], [20], and endocytosis limits secreted Wnt diffusion and modulates intracellular signaling [4], [21].
To identify new genes required for trafficking of developmental signaling molecules, we performed a forward genetic screen for mutations that alter the intracellular accumulation of Notch in developing D. melanogaster wing tissues. Among ∼40 new mutants recovered, two mutants displayed a strikingly similar phenotype of abnormal, non-cell-autonomous accumulation of Notch, Wingless, and other membrane proteins in endosomes and lysosomes. These two mutants disrupt essential enzymes of lipid metabolism, serine palmitoyltransferase (SPT) and acetyl-CoA carboxylase (ACC), which are encoded by the lace and ACC genes, respectively. SPT catalyzes the covalent attachment of serine to long chain fatty acyl-CoA to produce 3-ketodihydrosphingosine during sphingolipid biogenesis [22], [23]. Sphingolipids are major constituents of lipid rafts, a specialized membrane microdomain involved in endocytosis and signaling [24], [25]. In addition, bioactive sphingolipids participate in various signaling events, regulating cell proliferation, differentiation, apoptosis, and other cellular functions [26]. ACC catalyzes the carboxylation of acetyl-CoA to produce malonyl-CoA in the de novo synthesis of fatty acids [27], [28], which are needed for various cellular functions including energy storage, membrane biogenesis, and serving as precursors for phospholipid biosynthesis.
Notably, the D. melanogaster SPT and ACC mutants also exhibit tissue overgrowth phenotypes indicative of effects on cell proliferation. Analysis of downstream targets of Notch and Wingless reveals that this overgrowth is likely to involve modulatory effects on these pathways, reflecting both Notch hyperactivation and impaired Wingless signaling. Epistasis studies demonstrate that the overgrowth partially depends upon the Notch effector Su(H) as well as gamma-secretase function, and can also be partially suppressed by activated Armadillo, confirming that both Notch and Wingless dysregulation contributes to the lace and ACC mutant overgrowth phenotypes. Our findings emphasize the importance of lipid metabolism for establishing and maintaining the membrane compartments in which key developmental signaling pathways operate, and illustrate how general metabolic processes can exert complex, pleiotropic effects on multiple pathways needed for tissue growth and patterning.
Results
Abnormal Notch trafficking in D. melanogaster lace and ACC mutants
To identify new genes required for proper Notch trafficking, we designed a forward genetic screen in which homozygous mutant tissue is directly examined for aberrant Notch accumulation using antibody immunoanalysis. Because important trafficking genes would likely encode products essential for organismal viability, we created clones of homozygous mutant tissue in developing imaginal wing discs of otherwise heterozygous D. melanogaster using the FLP-FRT mosaic method [29] (see Materials and Methods). This approach also allows mutant tissues to be compared directly to adjacent heterozygous tissue in each sample, eliminating variability in fixation time, antibody penetration, and other parameters. Following screening of 3335 mutagenized second chromosome arms, we identified over 40 genes which, when mutated, alter the pattern of Notch trafficking as visualized using an antibody directed against the Notch intracellular domain.
Two lethal mutants, subsequently identified as lace and acetyl-CoA carboxylase (ACC) mutants, exhibited similar effects on Notch trafficking in homozygous mutant clones. In both cases, mutant cells display large, abnormal intracellular Notch-positive vesicles confined to internal clone regions approximately 3–4 cell diameters away from clone boundaries (Figures 1A and 1D). In homozygous mutant cells near clone boundaries, mutant effects on Notch trafficking are evidently rescued by non-cell-autonomous activity provided by nearby wildtype cells. The D. melanogaster wing disc is an oriented columnar epithelial monolayer, and the aberrant Notch-containing vesicles are observed throughout mutant cells from basement membrane to apical surface, in contrast to the predominantly apical accumulation of Notch in wildtype cells (Figures 1A and 1D). This non-autonomous Notch trafficking defect is consistently observed in both newly isolated alleles of ACC (ACC1 and ACC2), both newly isolated alleles of lace (lace18 and lace19), and a previously isolated amorphic allele of lace (lace2 [30]) (Figures 1A and 1D; Supplemental Figure S1A–E). In the lace and ACC mutants, the Notch ligand Delta is similarly mislocalized in a non-cell-autonomous manner (Figures 1C and 1F).
Fig. 1. Notch and Delta accumulate abnormally in lace and ACC mutant tissues. 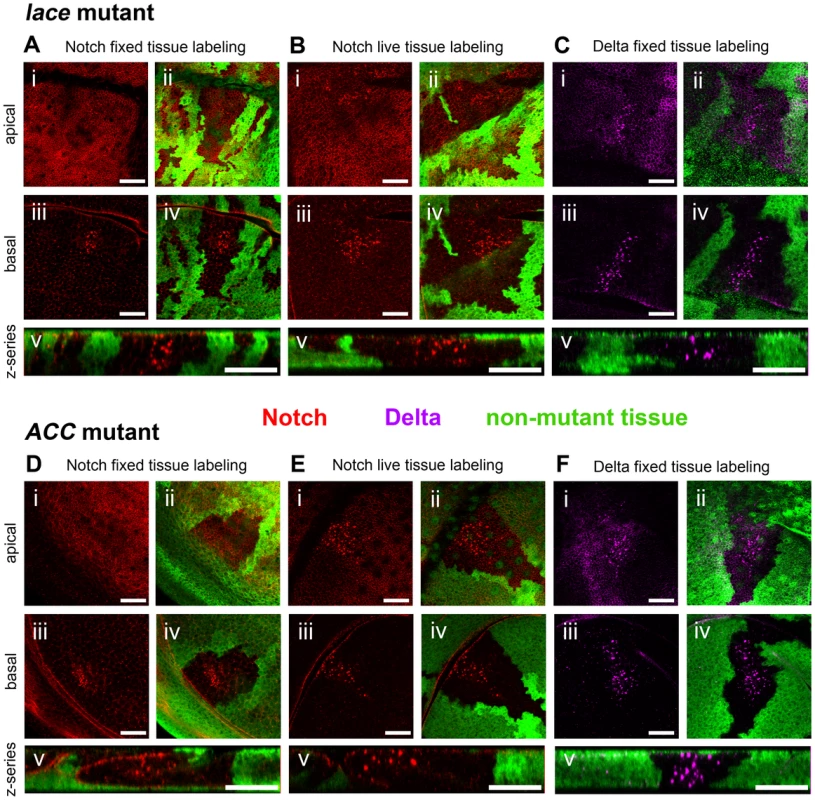
Confocal optical sections through D. melanogaster wing imaginal discs bearing homozygous mutant clones of lace18 (A–C) or ACC1 (D–F) showing accumulation of Notch (A, D) and Delta (C, F) in fixed tissue samples, and endocytic internalization of Notch in live tissue samples (B, E). Areas devoid of GFP marker gene expression (green) correspond to mutant cell regions. Each set of five images (i–v) depict an apical (i, ii) and basal (iii–iv) horizontal section showing Notch (red in A, B, D and E) or Delta (magenta in C and F) accumulation, the same images overlaid with corresponding GFP expression to indicate clone locations (ii and iv), and a representative z-series showing the distribution of Notch or Delta along the apicobasal axis of the disc tissue (v). Scale bars, 20 µm. We examined the localization of additional cell-surface molecules to determine whether the requirement for lace and ACC is specific to Notch signaling or also pertains to other developmental pathways. Wingless, the secreted ligand of the Wnt pathway, the Epidermal Growth Factor Receptor (EGFR), a receptor tyrosine kinase that activates Ras/MAPK signaling, and Patched, the receptor for the Hedgehog signal, also accumulate non-cell-autonomously in large intracellular vesicles in lace or ACC homozygous mutant cells (Figure 2A–L). Moreover, expression of an exogenous mCD8::GFP fusion protein also leads to its weak overaccumulation in the enlarged Notch-associated vesicles in lace or ACC mutant cells, although this co-accumulation of Notch and mCD8:GFP is primarily observed in apical but not basal cell regions (Figure 2M–P). Thus lace and ACC are likely to play a general role in intracellular protein trafficking and may influence the proper routing of numerous membrane proteins.
Fig. 2. Abnormal trafficking of Wingless, EGFR, Patched, and mCD8::GFP in lace and ACC mutant tissues. 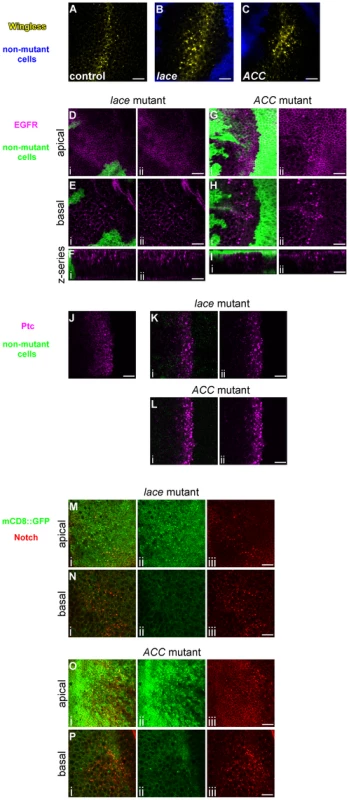
Confocal optical sections through D. melanogaster wing imaginal discs bearing homozygous mutant clones of lace2 (B, D–F, K, M, N) or ACC1 (C, G–I, L, O, P) showing accumulation of Wingless (A–C), EGFR (D–I), Patched (J–L), and mCD8::GFP (M–P). For A–C, Wingless expression is shown in yellow, and homozygous mutant clone regions are indicated by the absence of blue marker signal. For D–I, K, and L, each image pair depicts (i) the merged signals showing protein accumulation (magenta) and clone locations (areas devoid of green GFP signal) and (ii) the protein accumulation signal alone. For M–P, image triplets depict (i) mCD8::GFP (green) and Notch (red) accumulation, (ii) mCD8::GFP alone, and (iii) Notch alone. Apical, basal, and vertical z-series orientations are indicated at left in D–I, M–P. Wildtype control images are shown for the endogenous Wingless (A) and Patched (J) non-uniform expression domains. Scale bars, 10 µm. Molecular lesions in lace and ACC
Genetic mapping and complementation established that the newly recovered mutations are alleles of lace, which encodes serine palmitoyltransferase (SPT), and ACC encoding acetyl-CoA carboxylase. We determined the genomic sequence of the two lace alleles isolated from our screen as well as that of the previously isolated amorphic allele lace2 [30], all of which bear point mutations altering a single amino acid (Figure 3A). As noted above, we confirmed that the null allele lace2 shows the same non-cell-autonomous effect on Notch accumulation (see Supplemental Figure S1A–C), and thus lace2 was utilized for all subsequent analyses.
Fig. 3. Analysis of molecular lesions associated with lace and ACC mutants and alignment of human and D. melanogaster ACC protein domains. 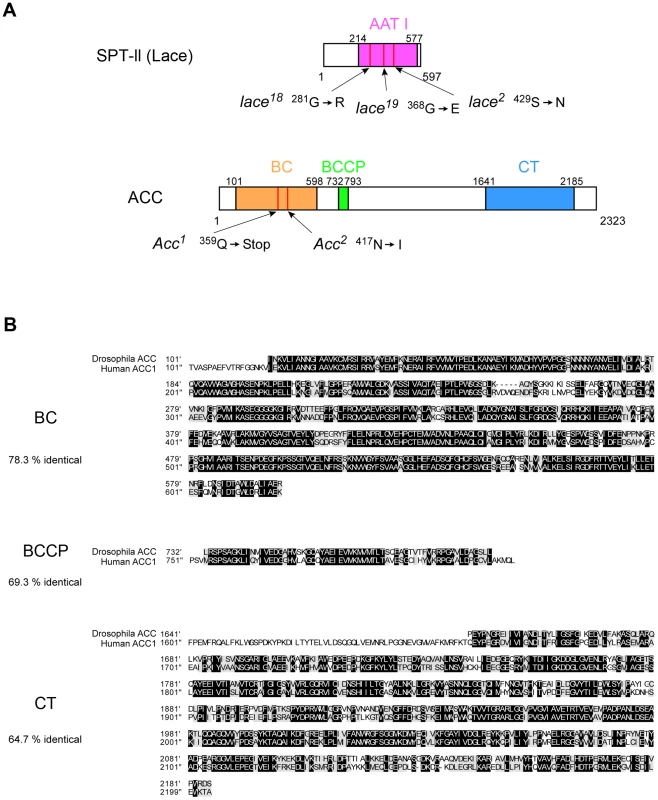
(A) Diagram of the Lace protein (SPT-II, serine palmitoyltransferase II) showing amino acid substitutions in the AAT I (amino-acid acetyltransferase I) domain in lace18, lace19, and lace2 mutants, and diagram of the ACC protein showing mutant lesions associated with ACC1 and ACC2 mutants. (B) Alignment of human and D. melanogaster ACC activity domains BC (biotin carboxylase), BCCP (biotin carboxyl carrier protein), and CT (carboxyltransferase) as depicted in panel A. Black boxes indicate identical residues; shaded boxes indicate conservative substitutions; percent identity is denoted at left for each domain. The newly recovered ACC mutants fail to complement P-element insertion line B131, in which the transposon is inserted into the intron of ACC [31]. ACC encodes D. melanogaster acetyl-CoA carboxylase (ACC), an enzyme that is highly conserved from bacteria to humans [27]. Over three separate domains, >60% of the amino acids are identical between D. melanogaster ACC and human ACC1 (Figure 3B). We sequenced ACC mutant alleles ACC1 and ACC2, and found that ACC1 contains a premature stop codon, indicating that ACC1 likely represents a null allele, while ACC2 encodes an amino acid substitution within the N-terminal conserved domain (Figure 3A). For subsequent studies, we used the presumptive null allele ACC1.
To confirm that the Notch trafficking defects observed in these mutants are specifically attributable to loss of lace and ACC activity, we expressed wildtype UAS-laceHA [32] (in which Lace contains an HA-epitope tag) and UAS-ACC cDNA constructs in posterior compartment clones of lace and ACC mutant cells, respectively, utilizing a hh-GAL4 driver line with UAS-FLP (see Materials and Methods). Expression of UAS-laceHA almost completely suppresses the Notch accumulation phenotype of lace2 clones, and expression of UAS-ACC fully suppresses this phenotype in ACC1 clones (Supplemental Figure S1F and S1G). To test whether non-specific hh-GAL4-mediated expression of either transgene can rescue the Notch accumulation defect, we also expressed UAS-laceHA in ACC1 clones, and conversely UAS-ACC in lace2 clones using the same approach. No significant rescue was observed in either case (Supplemental Figure S1H and S1I), indicating that the non-autonomous Notch trafficking defects seen in both mutants reflect specific requirements for lace and ACC gene activities rather than a general reduction in lipid homeostasis.
Notch accumulates abnormally in endocytic compartments of mutant cells
To identify the cellular compartment in which Notch accumulates in the lace and ACC mutants, we performed antibody uptake studies on clone-bearing wing discs. When live, unpermeabilized discs are incubated with antibodies that bind to the extracellularly exposed domain of Notch, the antibodies specifically detect the subpool of Notch at the cell surface and in surface-derived endocytic compartments [33]. Incubating lace and ACC mosaic mutant discs with anti-Notch extracellular antibody C458.2H, followed by a brief incubation period to allow antibody uptake by the live cells, subsequent tissue fixation and imaging revealed that for both mutants, the abnormal Notch vesicles reside in the endocytic trafficking pathway (Figures 1B and 1E).
To determine the specific endocytic compartment in which Notch accumulates, we performed double-labeling studies with Notch and various organelle markers, including Rab5-YFP, Rab7-YFP, Rab11-YFP, and LAMP-HRP, which label early endosomes, late endosomes, recycling endosomes, and late endosomes/lysosomes, respectively [34], [35]. In lace mutant cells, 38% of abnormal Notch vesicles colocalize with Rab5-YFP, 16% with LAMP-HRP, and 11% with Rab7-YFP (Figure 4E–4H; Supplemental Table S1). In ACC mutant cells, 55% of Notch-positive vesicles colocalize with LAMP-HRP, 15% with Rab5-YFP, and 12% with Rab7-YFP (Figure 4J–4M; Supplemental Table S1). In both mutants, the Notch-positive vesicles exhibit a much lower degree of colocalization with other organelle markers, including Rab11-YFP, Clathrin light chain-GFP (Clc-GFP), PDI-GFP (an ER marker), Golgi-YFP, and Sara (Figure 4G and 4L; Supplemental Figure S2A–H; Supplemental Table S1). Interestingly, LAMP-positive late endosomes/lysosomes are dramatically enlarged in lace and ACC mutant cells (Figures 4I and 4N, Supplemental Table S2), while other compartments are only slightly affected (Supplemental Table S2). Collectively, these results indicate that Notch accumulates primarily in early endosomes, late endosomes and lysosomes in lace mutant cells, but predominantly in lysosomes, and to a lesser extent in early and late endosomes in ACC mutant cells. The distinct patterns of Notch mislocalization in the two mutants might reflect different alterations in these endocytic compartments, and might also contribute to the differential effects of these mutants on Notch and Wingless signaling (see below). However, it should be noted that these organelle marker studies involve overexpression of endosomal machinery components under UAS control, which although widely used to label different endocytic compartments, might lead to abnormal endosomal compartment morphogenesis and/or function.
Fig. 4. Colocalization of Notch with endosomal and lysosomal markers in lace and ACC mutant tissue clones. 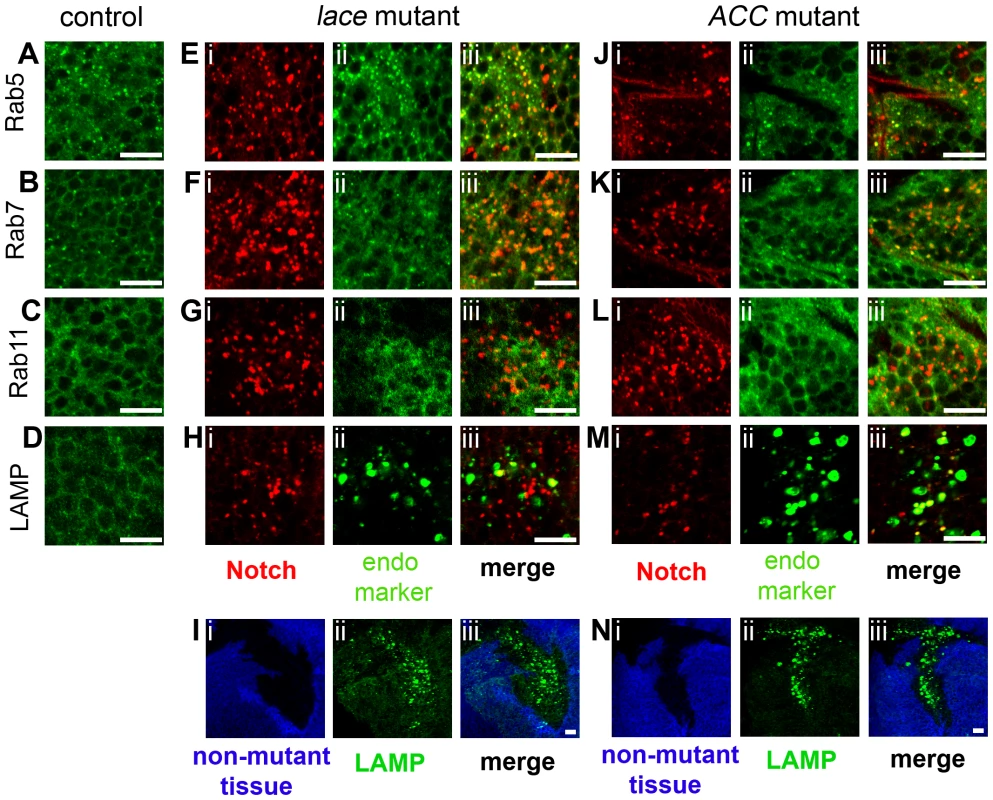
Each confocal image triplet (i–iii) depicts lace2 (E–I) or ACC1 (J–N) mutant wing disc clones, showing Notch overaccumulation (red in i for E–H, J–M) or mutant clone locations (absence of blue signal in i for I and N), the subcellular localization of the indicated organelle marker (green in ii), and the corresponding merged images at right (iii). Corresponding wildtype control images for each marker are shown in A–D; da-GAL4; UAS-Rab5-YFP, Rab7-YFP, or Rab11-YFP and UAS-LAMP-HRP were utilized for controls. Organelle markers in each panel are as follows: Rab5-YFP (Rab5; A, E, J), Rab7-YFP (Rab7; B, F, K), Rab11-YFP (Rab11; C, G, L), and LAMP-HRP (LAMP; D, H, I, M, N). Note elevated LAMP-HRP expression in lace2 and ACC1 mutant clones in I and N. Scale bars, 10 µm. Loss of lace and ACC activity causes tissue overgrowth
The lace and ACC mutants were examined for whether they might also show a cell proliferation phenotype, since several D. melanogaster endocytic trafficking mutants cause cell overproliferation, especially in large clones produced using the Minute system [36] or through ectopic expression of the Caspase inhibitor p35 [10]–[14]. Using the FLP-FRT system with Minute chromosomes to generate large clones of either lace or ACC mutant cells, we found that these clones showed significant tissue overgrowth (Figure 5A–5C). To control for clone size and location, we next produced lace and ACC mutant clones in specific disc regions by expressing UAS-FLP under the control of hh-GAL4. Testing multiple alleles of lace and ACC using this approach revealed that the overgrowth phenotypes are variable, ranging from ∼2% up to ∼95% for different lace alleles, and from ∼10% to ∼28% for the two available ACC alleles (Figure 5G–L; Supplemental Table S3). The lace and ACC overproliferation phenotypes are almost completely suppressed by overexpression of Lace and ACC, respectively (Figure 5N and 5P; Supplemental Table S3). Consistent with our findings above for the Notch trafficking phenotype, the overproliferation phenotypes of lace and ACC mutant cells could not be rescued by the converse overexpression of ACC and Lace, respectively (Figure 5O and 5Q; Supplemental Table S3).
Fig. 5. Analysis of tissue overgrowth and cell proliferation in lace and ACC mutant cells. 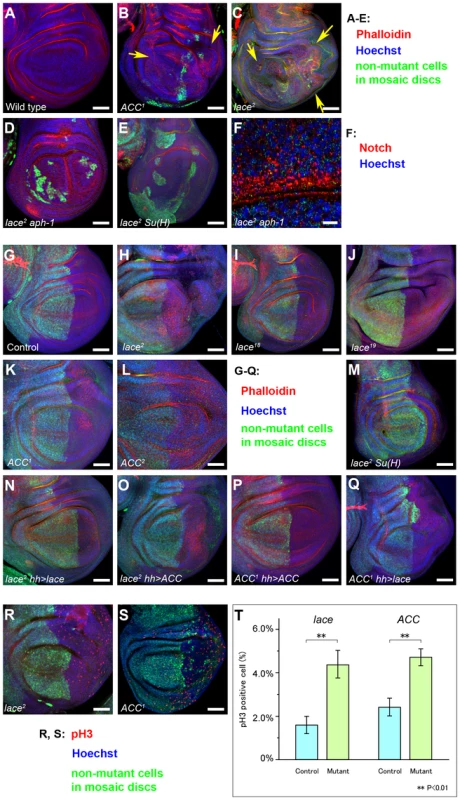
Confocal images of wing disc pouches corresponding to (A) wildtype, (B) ACC1 mutant clones, (C) lace2 mutant clones (arrows indicate tissue overgrowth regions in B and C), (D) lace2 aph-1D35 double mutant clones, and (E) lace2 Su(H)k07904 double mutant clones, with confocal signals for Hoechst (blue), Phalloidin (red) and Myc (green) staining used to reveal tissue architecture and mutant vs. wildtype cell territories in clone-bearing discs. (F) Confocal section of a lace2 aph-1D35 clone, showing high vesicular Notch accumulation (red). Hoechst-stained nuclei are shown in blue; faint green signal represents Myc antibody background staining that was used to identify the Myc-negative clones. (G–S) Mutant clones encompassing the wing posterior compartment were induced using the hh-GAL4; UAS-FLP system for (G) the control wildtype genotype (FRT40A FRTG13), (H, N, O, R) lace2, (I) lace18, (J) lace19, (K, P, Q, S) ACC1, (L) ACC2, and (M) lace2 Su(H)k07904. (N–Q) UAS-cDNA constructs encoding wildtype LaceHA (N, Q) or ACC (O, P) were expressed in either lace2 (N, O) or ACC1 (P, Q) mutant clones as indicated. Discs in G–Q were examined for Myc expression to identify Myc-negative clone regions (lack of green signal), Phalloidin (red), and Hoechst (blue). Genotypes of each panel correspond to those listed in Supplemental Table S3. (R, S) Wing imaginal discs with lace2 (R) or ACC1 (S) posterior compartment clones were analyzed with anti-phosphohistone H3 antibody (pH 3; red), anti-Myc (green; clone marker as above), and Hoechst (blue). Genotypes of (R) and (S) are the same as (H) and (K), respectively. (T) The percentages of pH 3-positive nuclei in lace2 or ACC1 homozygous mutant cells compared to control heterozygous cells were determined by analyzing Myc-negative and Myc-positive 127 µm×127 µm sectors, respectively, for ten discs of each genotype (**; P<0.01 by t-test). Scale bars, 50 µm in A–E, G–S; 10 µm in F. To confirm overproliferation at the cellular level in lace and ACC mutant cells, we examined phosphohistone H3 (pH 3) signals in clones of both mutants. The percentage of pH 3-positive cells is significantly increased in lace and ACC mutant cells compared to neighboring heterozygous cells (Figure 5R–T). We also examined apical-basal cell polarity in these overgrown mutant discs, since it is disrupted in other D. melanogaster endocytic mutants [10]–[14], in the glycosphingolipid metabolism mutants egghead and brainiac that disrupt Notch signaling during D. melanogaster oogenesis [37], and in several C. elegans mutants that likewise affect glycosphingolipid biosynthesis [38]. Unexpectedly, despite using four different markers to examine the apicobasal structure of lace and ACC mutant cells, we found that cell polarity is apparently unaffected in these mutant cells (Supplemental Figure S3A–T).
Notch signaling is altered in lace and ACC mutant cells
To determine which signaling pathways might be responsible for these overproliferation effects, we examined developmental gene expression patterns. Expression of Cut, which marks the presumptive wing margin during late larval development, was strongly reduced in regions where the margin extended deeply into lace or ACC mutant clones (Figure 6A–C). Cut expression depends on both Notch and Wingless activity [39]–[41], so we independently assessed Notch signaling in mutant clones using two additional Notch-responsive reporters. Expression of the vestigial boundary enhancer-lacZ (vgBE-lacZ) reporter [42] was diminished in lace and ACC mutant cells (Figure 6D–F). A second reporter, Gbe+Su(H)m8 [43], also showed reduced expression along the dorsal-ventral (D/V) boundary in lace and ACC mutant clones; however, ectopic weak signal induction was also observed (Figure 6G–I). This result indicates that loss of either lace or ACC has complex, differential effects on Notch signal activation depending upon its cellular context.
Fig. 6. Notch and Wingless signaling abnormalities in lace and ACC mutants. 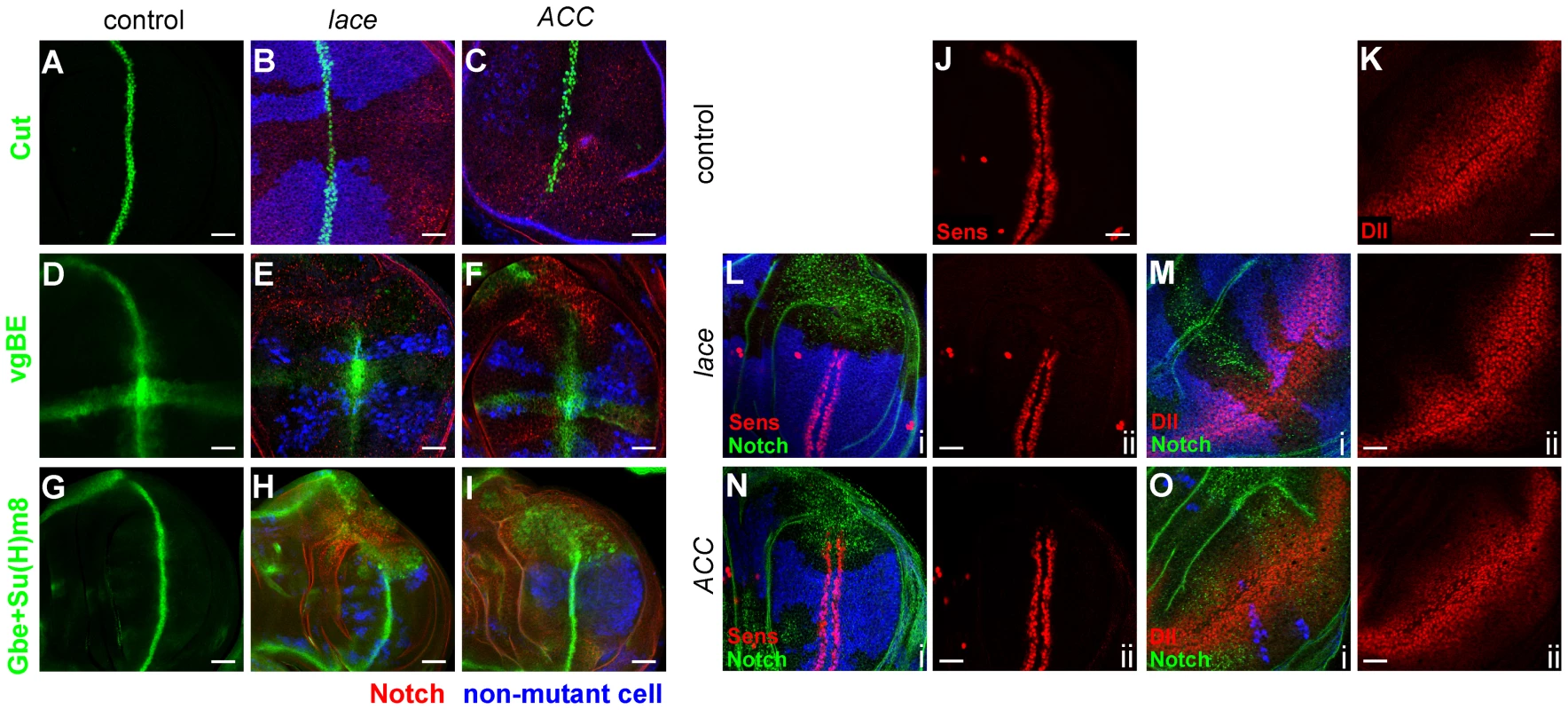
(A–I) Wing disc mutant clones of lace2 (B, E, H) and ACC1 (C, F, I) analyzed for expression of the Notch pathway reporters Cut (B, C; green), vestigial boundary enhancer (E, F; vgBE; green), and Gbe+Su(H)m8 (H, I; green). Mutant cell territories are indicated by absence of blue Myc or lacZ signal, and Notch accumulation is shown in red in B, C, E, F, H, and I. Wildtype expression patterns of the indicated Notch reporters are shown in A, D, and G (green). (J–O) Wing disc clones for lace2 (L, M) and ACC1 (N, O) were examined for activity of Wingless pathway reporters Senseless (Sens; J, L, N) and Distalless (Dll; K, M, O). For each panel i, mutant clone locations are indicated by absence of blue Myc or lacZ expression, Notch accumulation is shown in green, and Sens or Dll expression is in red; panel ii depicts the corresponding red channel only. Scale bars, 20 µm. These findings suggest that loss of lace or ACC has variable effects on Notch signaling in different tissues and even different wing disc regions. The Gbe+Su(H)m8 expression data suggest that Notch signaling might be modestly upregulated in non-margin disc regions, potentially contributing to the overproliferation phenotype. To test this idea, we asked whether the overproliferation requires functional gamma-secretase, the proteolytic enzyme complex that cleaves Notch to produce NICD. Wing disc clones mutant for both lace and aph-1 were generated, in which the aph-1 mutation inactivates an essential subunit of gamma-secretase [44]. In these clones, the aph-1 mutation strongly suppressed the overproliferation normally caused by the lace mutation but did not prevent elevated endosomal accumulation of Notch (Figure 5D and 5F). Taken together, these findings indicate that gamma-secretase-mediated Notch signaling activity is likely to be elevated in proliferating zones of the wing disc, leading directly or indirectly to the observed overgrowth. Confirming this interpretation, double mutant clones of lace and Su(H), which encodes a dedicated effector for Notch signaling, also exhibited substantial suppression of the lace wing disc overgrowth phenotype (Figure 5E and 5M; Supplemental Figure S3).
Wingless signaling is perturbed in lace and ACC mutants
Wingless signaling is also altered in lace and ACC mutant cells based on expression of two downstream targets, Senseless (Sens) and Distalless (Dll), which respond to strong and weak Wingless signaling, respectively [41], [45], [46]. In lace mutant cells, expression of both Wingless targets is decreased (Figure 6L and 6M), while Sens expression is only weakly decreased and Dll expression is apparently unaffected in ACC mutant cells (Figure 6N and 6O). To determine whether altered Wnt signaling might contribute to the lace and ACC mutant overproliferation phenotypes, an activated form of armadillo (encoding β-Catenin, an effector of Wnt signal), termed armS10, was expressed in lace and ACC mutant clones. The overproliferation phenotype was significantly suppressed in lace mutant clones, in terms of both frequency and severity of the phenotype (Figure 7A and 7C; Supplemental Table S4). For ACC clones, the severity of the overgrowth phenotype was reduced by armS10 expression (Figure 7B and 7D), although the percentage of discs exhibiting detectable overgrowth was not significantly different between control and armS10-expressing discs (Supplemental Table S4). These results are consistent with the idea that altered Wnt signaling partially contributes to the overproliferation phenotype in lace mutant cells, but seems to have a more modest role in the overproliferation observed for ACC-deficient cells.
Fig. 7. The lace overproliferation phenotype is partially rescued by activated Armadillo. 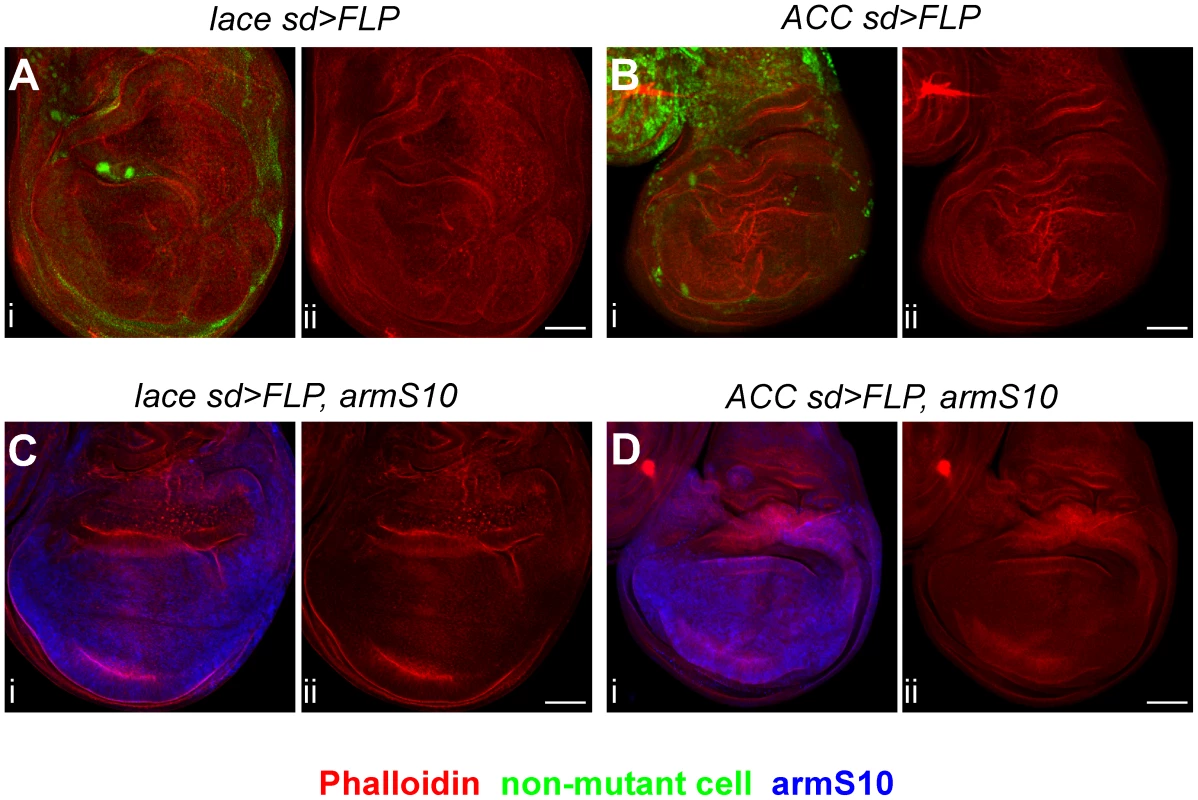
Mutant clones of lace2 (A, C) or ACC1 (B, D) were induced using sd-GAL4; UAS-FLP, and examined for tissue overgrowth in the absence (A, B) or presence (C, D) of a constitutively activated form of Armadillo (armS10), expressed using a UAS-armS10 transgene. For each image pair, panel i shows tissue growth patterns as revealed by Phalloidin (red in A–D), the mutant clone marker (green indicates non-mutant cells in A, B), or armS10 expression (blue in C, D), with the Phalloidin signal alone shown in panel ii. Complete genotypes are listed in Supplemental Table S4. Note that tissue overgrowth in the wing hinge region was not suppressed (C, D) because armS10 is not expressed in the hinge at this stage. Scale bars, 50 µm. In the above studies, we consistently observed that overgrowth phenotypes in lace and ACC clones were confined to the pouch and hinge region of the wing and not seen in the notum region, so we examined expression of a third Wnt downstream reporter, fz3-lacZ, in notum cells of mutant clone-bearing wing discs. Expression of fz3-lacZ was not altered in either lace or ACC mutant clones (data not shown), suggesting that the effects of the mutants on Wnt signaling, as with Notch signaling, are tissue-dependent.
Altered Dpp, Hippo, MAPK, Akt and JAK-STAT signaling do not contribute to the overproliferation observed for lace and ACC mutant cells
We also examined two pathways that mediate cell proliferation in D. melanogaster, namely Dpp and Hippo signaling, to determine if their activity was also perturbed by loss of lace or ACC activity. In lace and ACC mutant clones, we failed to detect elevated or ectopic expression of phosphorylated Mad, which transduces the active Dpp signal [47], or Spalt, a transcriptionally-induced target of Dpp and Mad in the wing imaginal disc (Supplemental Figure S4C, S4D, S4G and S4H). In some large lace clones, expression of these markers was reduced or absent, which might be an indirect consequence of globally disrupted developmental patterning in large mutant clones (cf. Supplemental Figure S4C).
Two downstream markers of Hippo signaling, Cyclin E and DIAP1-lacZ, are upregulated when Hippo signaliing is inactivated and are also associated with overgrowth phenotypes [48]. We observed a slight increase in DIAP1-lacZ expression in lace and ACC clones (Supplemental Figure S4B and S4F), but no significant increase in Cyclin E expression (Supplemental Figure S4A and S4E), indicating that Hippo signaling makes little if any contribution to the cellular overproliferation seen in the mutants.
In addition, we assessed activation of other growth control pathways, including the EGFR, Insulin receptor, Hedgehog, and JAK/STAT pathways, by monitoring levels of their respective downstream markers dpERK, pAkt, Cubitus Interruptus, and pSTAT in lace and ACC mutant wing disc clones. Loss of lace or ACC function was not associated with any obvious disruptions in the levels or subcellular localizations of these pathway markers (Supplemental Figure S5A–L). These findings support the idea that Notch and Wnt signaling are relatively more sensitive to the loss of Lace and ACC enzyme activities, although due to the limitations of these antibody probes, we cannot exclude the possibility that some activities of these other pathways are also subtly perturbed.
Discussion
The importance of lipid metabolism for the formation and maintenance of cell membranes is well established [24], [25], [49]. Both serine palmitoyltransferase (SPT) and acetyl-CoA carboxylase (ACC) are critical enzymes that control different steps of lipid metabolism, and are highly conserved in diverse animal species. Genetic elimination of ACC1 or the SPT subunits Sptlc1 or Sptlc2 cause early embryonic lethality in mice [50], [51], although the cellular basis for this lethality is unknown. In D. melanogaster, RNA-interfering disruption of ACC activity in the fat body results in reduced triglyceride storage and increased glycogen accumulation, and in oenocytes leads to loss of watertightness of the tracheal spiracles causing fluid entry into the respiratory system [52]. Here we demonstrate that D. melanogaster mutants lacking functional SPT or ACC exhibit endosomal trafficking defects, causing Notch, Wingless, EGFR, and Patched to accumulate abnormally in endosomes and lysosomes. These effects are accompanied by significant alterations in Notch and Wingless signaling, as revealed by changes in downstream target gene activation for both pathways. However, the mutants do not fully inactivate these developmental signaling pathways, and instead display phenotypes consistent with more complex, pleiotropic effects on Notch, Wingless, and potentially additional pathways in different tissues. Our findings reinforce the importance of lipid metabolism for the maintenance of proper developmental signaling, a concept that has also emerged from studies demonstrating that D. melanogaster mutants for phosphocholine cytidylyltransferase alter endosomal trafficking and signaling of Notch and EGFR [15], mutants for alpha-1,4-N-acetylgalactosaminyltransferase-1 affect endocytosis and activity of the Notch ligands Delta and Serrate [16], mutants for the ceramide synthase gene shlank disrupt Wingless endocytic trafficking and signaling [21], and mutants for the glycosphingolipid metabolism genes egghead and brainiac modify the extracellular gradient of the EGFR ligand Gurken [53].
Most strikingly, our newly characterized lace and ACC mutants also display prominent tissue overgrowth phenotypes. These tissue overgrowth effects are linked to changes in Notch and Wingless signaling outputs, and they involve gamma-secretase, Su(H), and Armadillo activities, suggesting that the overgrowth reflects an interplay of Wingless inactivation and Notch hyperactivation. Consistent with our findings, both Notch and Wingless regulate cell proliferation and imaginal disc size in D. melanogaster [54]. Moreover, several observations indicate that Notch and Wingless are jointly regulated by endocytosis, with opposing effects on their respective downstream pathway activities, a dynamic process that might be especially sensitive to perturbations in membrane lipid constituents [55]. Wingless itself exerts opposing effects on disc size that might depend on the particular developmental stage or disc territory. For example, hyperactivation of Wingless or inactivation of its negative regulators cause overproliferation [56]–[58], but Wingless activity can also constrain wing disc growth [59]. Similar spatiotemporal effects might underlie the variability we detected in our studies with lace and ACC mutant clones, in which both tissue overgrowth and developmentally arrested discs were observed. Although we did not detect obvious changes in downstream signaling for several other cell growth pathways that were examined, the trafficking abnormalities seen for other membrane proteins aside from Notch, Delta, and Wingless, as well as the incomplete suppression of the overgrowth phenotypes by blockage of Notch and Wingless signaling, suggest that other pathways might also be dysregulated in lace and ACC mutants, possibly contributing to the observed tissue overgrowth.
Wingless is modified by lipid addition [19], [20], and lipoprotein vesicles have been suggested to control Wingless diffusion [60]. In D. melanogaster embryos, endocytosis of Wingless limits its diffusion and ability to act as a long-range morphogen [61]. Endocytosis can also affect Wingless signaling in receiving cells, where endocytosis both promotes signal downregulation [61], [62] and positively facilitates signaling [63]. The apparently normal diffusion ranges for overaccumulated Wingless in lace and ACC mutant clones, yet reduced downstream target gene expression, is consistent with the idea that SPT and ACC act by promoting endocytic trafficking of Wingless in receiving cells rather than influencing the secretion and/or diffusion of Wingless from signal-sending cells.
Our finding that lace and ACC mutant overgrowth phenotypes are also partially Notch-dependent is reminiscent of similar overproliferation phenotypes seen in certain D. melanogaster endocytic mutants, such as vps25, and tsg101 [11]–[14]. The overproliferation of disc tissue in these mutants is attributable to Notch hyperactivation, reflecting the fact that non-ligand-bound Notch receptors that are normally targeted for recycling or degradation are instead retained and signal from endosomes. Analogous effects are likely to contribute to the lace and ACC mutant overgrowth, where we observe significant Notch overaccumulation throughout the endosomal-lysosomal routing pathway. Some ectopic Notch signaling might emanate from the lysosomal compartment, which is enlarged and accumulates particularly high levels of Notch in lace and ACC mutant clones. Analysis of D. melanogaster HOPS and AP-3 mutants, which affect protein delivery to lysosomes, has identified a lysosomal pool of Notch that is able to signal in a ligand-independent, gamma-secretase-dependent manner [64].
How do SPT and ACC contribute to endosomal trafficking of Notch and other proteins? In the yeast SPT mutant lcb1, an early step of endocytosis is impaired due to defective actin attachment to endosomes, a phenotype that is suppressed by addition of sphingoid base [65]. However, the trafficking abnormalities seen in lace and ACC mutants do not resemble those in the yeast lcb1 mutant, perhaps because endocytic vesicle fission is primarily dependent upon dynamin in D. melanogaster and mammals, instead of actin as in yeast [66]. Nevertheless, the requirement for SPT and ACC in D. melanogaster endosomal compartments might reflect possible functions in endosome-cytoskeleton interactions. Another possibility is that the defective endosomal trafficking seen in lace and ACC mutants is caused by the inability to synthesize specific phospholipids needed for normal membrane homeostasis. Finally, lace and ACC might be important for the formation and/or function of lipid rafts, specialized membrane microdomains that have been implicated in both signaling and protein trafficking [67], [68].
A remarkable feature of the lace and ACC mutant phenotypes that suggests an underlying defect in lipid biogenesis is the non-autonomous effect in mutant tissue clones, wherein nearby wildtype cells generate a secreted activity that diffuses several cell diameters into the mutant tissue and rescues the trafficking and signaling defects. One possibility is that these secreted activities are diffusible lipid biosynthetic products of SPT and ACC, which enter the mutant cells and serve as precursors for further biosynthetic steps that do not require SPT or ACC. An intriguing alternative is that the SPT and ACC enzymes are themselves secreted and taken up by the mutant cells. A precedent for this mechanism has recently been demonstrated for D. melanogaster ceramidase, a sphingolipid metabolic enzyme that is secreted extracellularly, delivered to photoreceptors, and internalized by endocytosis to regulate photoreceptor cell membrane turnover [69].
Recent work has highlighted the importance of lipid metabolism for oncogenic transformation, and ACC has been advanced as a promising target for cancer drug development [70]. ACC is upregulated in some cancers, possibly as a result of high demands for lipid biosynthesis during rapid cell divisions. Sphingolipids and their derivatives are also thought to influence the balance of apoptosis and cell proliferation during tissue growth, and thus have also garnered attention as potential cancer therapy targets [71]. Our findings regarding the requirements of SPT and ACC for proper trafficking and signaling of key developmental cell-surface signaling molecules, including Notch and Wingless, provide insights into how lipid metabolic enzymes might influence cell proliferation and tissue patterning in multicellular animals. Complex lipid biosynthesis is essential for the creation of the elaborate, interconnected, and highly specialized membrane compartments in which developmental pathways operate, and perturbations in lipid biosynthesis that are tolerated by the cell might nevertheless exert significant pleiotropic effects on developmental patterning, cell proliferation, and other cellular processes. Exploration of lipid metabolic enzymes as pharmacological targets must therefore take into account potentially unfavorable effects on critical signaling pathways controlling development and organogenesis.
Materials and Methods
Constructs
A full-length ACC cDNA (GH12002; obtained from the Drosophila Genomics Resource Center, Indiana University) was subcloned into the XbaI and SmaI sites of pBluescript II SK−, introducing a new NotI site between KpnI and HindIII. The resulting NotI fragment was excised and inserted into the NotI site of the pUAST vector. Transformants of UAS-ACC were obtained according to standard protocols.
D. melanogaster genetics
Mutagenesis was performed using standard protocols by administering 35 mM ethylmethanesulfonate to isogenic male flies of genotype y w; P{ry[+t7.2] = neoFRT}40A P{w[+mW.hs] = FRT(whs)}G13, which were used to establish candidate mutant stocks. For screening of 3335 mutagenized second chromosome arms, these stocks were mated to marked 2L and 2R FRT stocks to yield progeny bearing homozygous candidate mutant wing clones using the FLP/FRT method [29]. 10 wing discs of each candidate mutant line were harvested and analyzed for abnormal Notch accumulation by direct immunofluorescence using Notch antibody C17.9C6 as described below.
D. melanogaster stocks used included Oregon-R as wild type, thj5C8 as a DIAP1-lacZ marker (Bloomington Drosophila Stock Center), UAS-Rab5-YFP, UAS-Rab7-YFP, UAS-Rab11-YFP [34], UAS-LAMP-HRP [35], UAS-EGFP-clc [72], sqh-EYFP-Golgi [73], and PDI-GFP [74] as intracellular compartment markers, UAS-laceHA [32] as a lace rescue transgene, UAS-armS10 [75] to express activated Armadillo, vgBE-lacZ [42] and Gbe+Su(H)m8 [43] as Notch target gene reporters, da-GAL4 and hh-GAL4 (courtesy of Dr. Jin Jiang) as GAL4 drivers, fz3-lacZ (fz3J29 [76]) as a Wingless signal reporter, P{Ubi-GFP(S65T)nls}2L FRT40A, M(2)24F1 P{πM}36F FRT40A, P{arm-lacZ.V}36BC FRT40A, FRTG13 P{Ubi-GFP.nls}2R1 P{Ubi-GFP.nls}2R2, FRT42D P{πM}45F M(2)531, and FRT42D P{arm-lacZ.V}51D as FLP-FRT clone makers, with P{hsFLP}12 or P{UAS-FLP.Exel}1 as FLP sources.
Double mutant clones of lace2 aph-1D35 and lace2 Su(H)k07904 were produced by the FLP/FRT method following recombination of aph-1D35 [44] and Su(H)k07904 [77] onto the FRT40A lace2 chromosome. To check Notch intracellular localization, hs-FLP; lace2 FRT40A/tub-GAL80 FRT40A; da-GAL4 combined with either UAS-driven Rab5-YFP, 7-YFP, 11-YFP, EGFP-clc, or LAMP-HRP, or hs-FLP; FRTG13 ACC1/FRTG13 tub-GAL80 da-GAL4 combined with either UAS-driven Rab5-YFP, 7-YFP, 11-YFP, EGFP-clc, or LAMP-HRP larvae were dissected and stained with anti-Notch and anti-GFP antibodies described below.
Immunohistology
Wing imaginal discs were dissected, fixed, and immunostained [44] using the following primary antibodies: mouse Notch intracellular domain antibody C17.9C6 (1∶1000; [78]; DSHB, University of Iowa); mouse Notch extracellular domain antibody C458.2H (1∶500; [79]; DSHB, University of Iowa); rat Notch3 (1∶1000; courtesy of Dr. Spyros Artavanis-Tsakonas); mouse Delta antibody C594.9B (1∶1000; [80]; DSHB, University of Iowa); mouse Cut 2B10 (1∶1000; DSHB, University of Iowa); rat ELAV antibody 7E8A10 (1∶500; DSHB, University of Iowa); mouse Wg antibody 4D4 (1∶500; [81]; DSHB, University of Iowa); guinea pig Sens antibody (1∶1000; [45]; courtesy of Dr. Hugo Bellen); mouse Distalless antibody DMDll.1 (1∶400; [82]; courtesy of Dr. Ian Duncan); mouse Engrailed antibody 4D9 (1∶500; [83]; DSHB, University of Iowa); rat CycE antibody (1∶1000; [84]; courtesy of Dr. Helena Richardson); rabbit phosphorylated Mad antibody PS1 (1∶500; [47]; courtesy of Dr. Carl H. Heldin); rabbit Spalt antibody (1∶400; [85]; courtesy of Dr. Rosa Barrio); rabbit Sara antibody (1∶500; [86]; courtesy of Dr. Franck Coumailleau); rat DE-Cadherin antibody DCAD2 (1∶20; [87]; DSHB, University of Iowa); mouse Discs large 4F3 (1∶10; [88]; DSHB, University of Iowa); rabbit PKCξ antibody C20 (1∶1000; Santa Cruz); mouse Armadillo antibody N2 7A1 (1∶500; [89]; DSHB, University of Iowa); mouse CD2 antibody MCA154GA (1∶1000; AbD Serotech); chicken Myc antibody NB600-334 (1∶1000; Novus Biologicals); rat Myc antibody JAC6 (1∶500; Novus Biologicals); rabbit GFP antibody 598 (1∶1000; MBL); rat GFP antibody GF090R (1∶500; NacalaiTesque); mouse ß-galactosidase antibody Z378A (1∶1000; Promega); chicken ß-galactosidase antibody XW-7591 (1∶1000; ProSci); mouse HRP antibody 2H11 (1∶500; Santa Cruz Biotechnology); goat anti-Egfr antibody dC-20 (1∶500; Santa Cruz Biotechnology), mouse anti-Patched antibody (1/500; [90]; DSHB, University of Iowa), mouse anti-Active(dp) MAPK antibody A3713 (1∶500; Sigma), rabbit anti-phospho-Drosophila Akt (Ser505) Antibody (Cell Signaling Technology), rat anti-Cubitus Interruptus 2A1 (1/500; [91]; DSHB, University of Iowa), rabbit anti-phosphohistone H3 Ser10 antibody (Upstate), rabbit anti-phosphorylated STAT (Cell Signaling Technology). Confocal images were acquired using LSM510META and LSM700 (Zeiss) confocal microscopes, and fluorescent intensity was measured using ImageJ software.
For live tissue labeling, dissected wing discs were incubated with antibody for 40 min in S2 cell culture medium (Gibco), washed three times for 10 min with S2 medium, fixed and processed further as above. Hoechst-33342 trihydrochloride trihydrate (Invitrogen; 1∶1000 dilution) and Phalloidin-Alexa546 (Molecular Probes; 1∶20 dilution) stainings were performed for 1 hr at room temperature following immunostaining.
Supporting Information
Zdroje
1. KopanR, IlaganMX (2009) The canonical Notch signaling pathway: unfolding the activation mechanism. Cell 137 : 216–233.
2. FortiniME (2009) Notch signaling: the core pathway and its posttranslational regulation. Dev Cell 16 : 633–647.
3. CleversH (2006) Wnt/β-catenin signaling in development and disease. Cell 127 : 469–480.
4. GagliardiM, PiddiniE, VincentJP (2008) Endocytosis: a positive or a negative influence on Wnt signalling? Traffic 9 : 1–9.
5. BraySJ (2006) Notch signalling: a simple pathway becomes complex. Nat Rev Mol Cell Biol 7 : 678–689.
6. KovallRA (2008) More complicated than it looks: assembly of Notch pathway transcription complexes. Oncogene 27 : 5099–5109.
7. YamamotoS, CharngWL, BellenHJ (2010) Endocytosis and intracellular trafficking of Notch and its ligands. Curr Top Dev Biol 92 : 165–200.
8. SeugnetL, SimpsonP, HaenlinM (1997) Requirement for dynamin during Notch signaling in Drosophila neurogenesis. Dev Biol 192 : 585–598.
9. GordonWR, Vardar-UluD, HistenG, Sanchez-IrizarryC, AsterJC, et al. (2007) Structural basis for autoinhibition of Notch. Nat Struct Mol Biol 14 : 295–300.
10. LuH, BilderD (2005) Endocytic control of epithelial polarity and proliferation in Drosophila. Nat Cell Biol 7 : 1232–1239.
11. VaccariT, BilderD (2005) The Drosophila tumor suppressor vps25 prevents nonautonomous overproliferation by regulating Notch trafficking. Dev Cell 9 : 687–698.
12. ThompsonBJ, MathieuJ, SungHH, LoeserE, RørthP, et al. (2005) Tumor suppressor properties of the ESCRT-II complex component Vps25 in Drosophila. Dev Cell 9 : 711–720.
13. MobergKH, SchelbleS, BurdickSK, HariharanIK (2005) Mutations in erupted, the Drosophila ortholog of mammalian tumor susceptibility gene 101, elicit non-cell-autonomous overgrowth. Dev Cell 9 : 699–710.
14. HerzHM, ChenZ, ScherrH, LackeyM, BolducC, et al. (2006) vps25 mosaics display non-autonomous cell survival and overgrowth, and autonomous apoptosis. Development 133 : 1871–1880.
15. WeberU, ErogluC, MlodzikM (2003) Phospholipid membrane composition affects EGF receptor and Notch signaling through effects on endocytosis during Drosophila development. Dev Cell 5 : 559–570.
16. HamelS, FantiniJ, SchweisguthF (2010) Notch ligand activity is modulated by glycosphingolipid membrane composition in Drosophila melanogaster. J Cell Biol 188 : 581–594.
17. BehrensJ, von KriesJP, KuhlM, BruhnL, WedlichD, et al. (1996) Functional interaction of β-catenin with the transcription factor LEF-1. Nature 382 : 638–642.
18. van de WeteringM, CavalloR, DooijesD, van BeestM, van EsJ, et al. (1997) Armadillo coactivates transcription driven by the product of the Drosophila segment polarity gene dTCF. Cell 88 : 789–799.
19. WillertK, BrownJD, DanenbergE, DuncanAW, WeissmanIL, et al. (2003) Wnt proteins are lipid-modified and can act as stem cell growth factors. Nature 423 : 448–452.
20. ZhaiL, ChaturvediD, CumberledgeS (2004) Drosophila Wnt-1 undergoes a hydrophobic modification and is targeted to lipid rafts, a process that requires Porcupine. J Biol Chem 279 : 33220–33227.
21. PepperlJ, ReimG, LüthiU, KaechA, HausmannG, et al. (2013) Sphingolipid depletion impairs endocytic traffic and inhibits Wingless signaling. Mech Dev 130 : 493–505.
22. HanadaK (2003) Serine palmitoyltransferase, a key enzyme of sphingolipid metabolism. Biochim Biophys Acta 1632 : 16–30.
23. PerryDK (2002) Serine palmitoyltransferase: role in apoptotic de novo ceramide synthesis and other stress responses. Biochim Biophys Acta 1585 : 146–152.
24. LajoieP, NabiIR (2010) Lipid rafts, caveolae, and their endocytosis. Int Rev Cell Mol Biol 282 : 135–163.
25. SillenceDJ (2007) New insights into glycosphingolipid functions–storage, lipid rafts, and translocators. Int Rev Cytol 262 : 151–189.
26. HannunYA, ObeidLM (2008) Principles of bioactive lipid signalling: lessons from sphingolipids. Nat Rev Mol Cell Biol 9 : 139–150.
27. TongL (2005) Acetyl-coenzyme A carboxylase: crucial metabolic enzyme and attractive target for drug discovery. Cell Mol Life Sci 62 : 1784–1803.
28. WakilSJ, Abu-ElheigaLA (2009) Fatty acid metabolism: target for metabolic syndrome. J Lipid Res 50 Suppl: S138–143.
29. XuT, HarrisonSD (1994) Mosaic analysis using FLP recombinase. Methods Cell Biol 44 : 655–681.
30. AshburnerM, ThompsonP, RooteJ, LaskoPF, GrauY, et al. (1990) The genetics of a small autosomal region of Drosophila melanogaster containing the structural gene for alcohol dehydrogenase. VII. Characterization of the region around the snail and cactus loci. Genetics 126 : 679–694.
31. TweedieS, AshburnerM, FallsK, LeylandP, McQuiltonP, et al. (2009) FlyBase: enhancing Drosophila Gene Ontology annotations. Nucleic Acids Res 37: D555–559.
32. Adachi-YamadaT, GotohT, SugimuraI, TatenoM, NishidaY, et al. (1999) De novo synthesis of sphingolipids is required for cell survival by down-regulating c-Jun N-terminal kinase in Drosophila imaginal discs. Mol Cell Biol 19 : 7276–7286.
33. PerizG, FortiniME (1999) Ca2+-ATPase function is required for intracellular trafficking of the Notch receptor in Drosophila. EMBO J 18 : 5983–5993.
34. ZhangJ, SchulzeKL, HiesingerPR, SuyamaK, WangS, et al. (2007) Thirty-one flavors of Drosophila rab proteins. Genetics 176 : 1307–1322.
35. LloydTE, AtkinsonR, WuMN, ZhouY, PennettaG, et al. (2002) Hrs regulates endosome membrane invagination and tyrosine kinase receptor signaling in Drosophila. Cell 108 : 261–269.
36. MorataG, RipollP (1975) Minutes: mutants of Drosophila autonomously affecting cell division rate. Dev Biol 42 : 211–221.
37. GoodeS, MelnickM, ChouTB, PerrimonN (1996) The neurogenic genes egghead and brainiac define a novel signaling pathway essential for epithelial morphogenesis during Drosophila oogenesis. Development 122 : 3863–3879.
38. ZhangH, AbrahamN, KhanLA, HallDH, FlemingJT, et al. (2011) Apicobasal domain identities of expanding tubular membranes depend on glycosphingolipid biosynthesis. Nat Cell Biol 13 : 1189–1201.
39. CousoJP, BishopSA, Martinez AriasA (1994) The Wingless signalling pathway and the patterning of the wing margin in Drosophila. Development 120 : 621–636.
40. NeumannCJ, CohenSM (1996) A hierarchy of cross-regulation involving Notch, wingless, vestigial and cut organizes the dorsal/ventral axis of the Drosophila wing. Development 122 : 3477–3485.
41. de CelisJF, BrayS (1997) Feed-back mechanisms affecting Notch activation at the dorsoventral boundary in the Drosophila wing. Development 124 : 3241–3251.
42. KimJ, SebringA, EschJJ, KrausME, VorwerkK, et al. (1996) Integration of positional signals and regulation of wing formation and identity by Drosophila vestigial gene. Nature 382 : 133–138.
43. FurriolsM, BrayS (2001) A model Notch response element detects Suppressor of Hairless-dependent molecular switch. Curr Biol 11 : 60–64.
44. HuY, FortiniME (2003) Different cofactor activities in gamma-secretase assembly: evidence for a Nicastrin-Aph-1 subcomplex. J Cell Biol 161 : 685–690.
45. NoloR, AbbottLA, BellenHJ (2000) Senseless, a Zn finger transcription factor, is necessary and sufficient for sensory organ development in Drosophila. Cell 102 : 349–362.
46. ZeccaM, BaslerK, StruhlG (1996) Direct and long-range action of a Wingless morphogen gradient. Cell 87 : 833–844.
47. TanimotoH, ItohS, ten DijkeP, TabataT (2000) Hedgehog creates a gradient of DPP activity in Drosophila wing imaginal discs. Mol Cell 5 : 59–71.
48. WuS, HuangJ, DongJ, PanD (2003) hippo encodes a Ste-20 family protein kinase that restricts cell proliferation and promotes apoptosis in conjunction with salvador and warts. Cell 114 : 445–456.
49. Van der HorstDJ, RoosendaalSD, RodenburgKW (2009) Circulatory lipid transport: lipoprotein assembly and function from an evolutionary perspective. Mol Cell Biochem 326 : 105–119.
50. HojjatiMR, LiZ, JiangXC (2005) Serine palmitoyl-CoA transferase (SPT) deficiency and sphingolipid levels in mice. Biochim Biophys Acta 1737 : 44–51.
51. Abu-ElheigaL, MatzukMM, KordariP, OhW, ShaikenovT, et al. (2005) Mutant mice lacking acetyl-CoA carboxylase 1 are embryonically lethal. Proc Natl Acad Sci U S A 102 : 12011–12016.
52. ParvyJP, NapalL, RubinT, PoldevinM, PerrinL, et al. (2012) Drosophila melanogaster acetyl-CoA-carboxylase sustains a fatty acid-dependent remote signal to waterproof the respiratory system. PLoS Genet 8: e1002925.
53. PizetteS, RabouilleC, CohenSM, ThérondP (2009) Glycosphingolipids control the extracellular gradient of the Drosophila EGFR ligand Gurken. Development 136 : 551–561.
54. HerranzH, MilanM (2008) Signalling molecules, growth regulators and cell cycle control in Drosophila. Cell Cycle 7 : 3335–3337.
55. Muñoz DescalzoS, Martinez AriasA (2012) The structure of Wntch signalling and the resolution of transition states in development. Sem Cell Dev Biol 23 : 443–449.
56. Baena-LopezLA, Franch-MarroX, VincentJP (2009) Wingless promotes proliferative growth in a gradient-independent manner. Sci Signal 2: ra60.
57. HerranzH, PerezL, MartinFA, MilanM (2008) A Wingless and Notch double-repression mechanism regulates G1-S transition in the Drosophila wing. EMBO J 27 : 1633–1645.
58. GiraldezAJ, CohenSM (2003) Wingless and Notch signaling provide cell survival cues and control cell proliferation during wing development. Development 130 : 6533–6543.
59. JohnstonLA, SandersAL (2003) Wingless promotes cell survival but constrains growth during Drosophila wing development. Nat Cell Biol 5 : 827–833.
60. PanákováD, SprongH, MaroisE, ThieleC, EatonS (2005) Lipoprotein particles are required for Hedgehog and Wingless signalling. Nature 435 : 58–65.
61. DuboisL, LecourtoisM, AlexandreC, HirstE, VincentJP (2001) Regulated endocytic routing modulates Wingless signaling in Drosophila embryos. Cell 105 : 613–624.
62. PiddiniE, MarshallF, DuboisL, HirstE, VincentJP (2005) Arrow (LRP6) and Frizzled2 cooperate to degrade Wingless in Drosophila imaginal discs. Development 132 : 5479–5489.
63. SetoES, BellenHJ (2006) Internalization is required for proper Wingless signaling in Drosophila melanogaster. J Cell Biol 173 : 95–106.
64. WilkinM, TongngokP, GenschN, ClemenceS, MotokiM, et al. (2008) Drosophila HOPS and AP-3 complex genes are required for a Deltex-regulated activation of Notch in the endosomal trafficking pathway. Dev Cell 15 : 762–772.
65. ZanolariB, FriantS, FunatoK, SutterlinC, StevensonBJ, et al. (2000) Sphingoid base synthesis requirement for endocytosis in Saccharomyces cerevisiae. EMBO J 19 : 2824–2833.
66. ConibearE (2010) Converging views of endocytosis in yeast and mammals. Curr Opin Cell Biol 22 : 513–518.
67. LingwoodD, SimonsK (2010) Lipid rafts as a membrane-organizing principle. Science 237 : 46–50.
68. PontierSM, SchweisguthF (2012) Glycosphingolipids in signaling and development: from liposomes to model organisms. Dev Dyn 241 : 92–106.
69. AcharyaJK, DasguptaU, RawatSS, YuanC, SanxaridisPD, et al. (2008) Cell-nonautonomous function of ceramidase in photoreceptor homeostasis. Neuron 57 : 69–79.
70. SwinnenJV, BrusselmansK, VerhoevenG (2006) Increased lipogenesis in cancer cells: new players, novel targets. Curr Opin Clin Nutr Metab Care 9 : 358–365.
71. SaddoughiSA, SongP, OgretmenB (2008) Roles of bioactive sphingolipids in cancer biology and therapeutics. Subcell Biochem 49 : 413–440.
72. ChangHC, NewmyerSL, HullMJ, EbersoldM, SchmidSL, et al. (2002) Hsc70 is required for endocytosis and clathrin function in Drosophila. J Cell Biol 159 : 477–487.
73. LaJeunesseDR, BucknerSM, LakeJ, NaC, PirtA, et al. (2004) Three new Drosophila markers of intracellular membranes. Biotechniques 36 : 784–788, 790.
74. BobinnecY, MarcaillouC, MorinX, DebecA (2003) Dynamics of the endoplasmic reticulum during early development of Drosophila melanogaster. Cell Motil Cytoskeleton 54 : 217–225.
75. PaiLM, OrsulicS, BejsovecA, PeiferM (1997) Negative regulation of Armadillo, a Wingless effector in Drosophila. Development 124 : 2255–2266.
76. SatoA, KojimaT, Ui-TeiK, MiyataY, SaigoK (1999) Dfrizzled-3, a new Drosophila Wnt receptor, acting as an attenuator of Wingless signaling in wingless hypomorphic mutants. Development 126 : 4421–4430.
77. RochF, SerrasF, CifuentesFJ, CorominasM, AlsinaB, et al. (1998) Screening of larval/pupal P-element induced lethals on the second chromosome in Drosophila melanogaster: clonal analysis and morphology of imaginal discs. Mol Gen Genet 257 : 103–112.
78. FehonRG, KoohPJ, RebayI, ReganCL, XuT, et al. (1990) Molecular interactions between the protein products of the neurogenic loci Notch and Delta, two EGF-homologous genes in Drosophila. Cell 61 : 523–534.
79. DiederichRJ, MatsunoK, HingH, Artavanis-TsakonasS (1994) Cytosolic interaction between deltex and Notch ankyrin repeats implicates deltex in the Notch signaling pathway. Development 120 : 473–481.
80. QiH, RandMD, WuX, SestanN, WangW, et al. (1999) Processing of the Notch ligand Delta by the metalloprotease Kuzbanian. Science 283 : 91–94.
81. BrookWJ, CohenSM (1996) Antagonistic interactions between Wingless and Decapentaplegic responsible for dorsal-ventral pattern in the Drosophila leg. Science 273 : 1373–1377.
82. DuncanDM, BurgessEA, DuncanI (1998) Control of distal antennal identity and tarsal development in Drosophila by spineless-aristapedia, a homolog of the mammalian dioxin receptor. Genes Dev 12 : 1290–1303.
83. PatelNH, Martin-BlancoE, ColemanKG, PooleSJ, EllisMC, et al. (1989) Expression of engrailed proteins in arthropods, annelids, and chordates. Cell 58 : 955–968.
84. CrackD, SecombeJ, CoombeM, BrumbyA, SaintR, et al. (2002) Analysis of Drosophila Cyclin EI and II function during development: identification of an inhibitory zone within the morphogenetic furrow of the eye imaginal disc that blocks the function of Cyclin EI but not Cyclin EII. Dev Biol 241 : 157–171.
85. KühnleinRP, FrommerG, FriedrichM, Gonzalez-GaitanM, WeberA, et al. (1994) spalt encodes an evolutionarily conserved zinc finger protein of novel structure which provides homeotic gene function in the head and tail region of the Drosophila embryo. EMBO J 13 : 168–179.
86. CoumailleauF, FürthauerM, KnoblichJA, González-GaitánM (2009) Directional Delta and Notch trafficking in Sara endosomes during asymmetric cell division. Nature 458 : 1051–1055.
87. OdaH, UemuraT, HaradaY, IwaiY, TakeichiM (1994) A Drosophila homolog of Cadherin associated with Armadillo and essential for embryonic cell-cell adhesion. Dev Biol 165 : 716–726.
88. ParnasD, HaghighiAP, FetterRD, KimSW, GoodmanCS (2001) Regulation of postsynaptic structure and protein localization by the Rho-type guanine nucleotide exchange factor dPix. Neuron 32 : 415–424.
89. PeiferM, SweetonD, CaseyM, WieschausE (1994) wingless signal and Zeste-white 3 kinase trigger opposing changes in the intracellular distribution of Armadillo. Development 120 : 369–380.
90. CapdevilaJ, ParienteF, SampedroJ, AlonsoJL, GuerreroI (1994) Subcellular localization of the segment polarity protein Patched suggests an interaction with the Wingless reception complex in Drosophila embryos. Development 120 : 987–998.
91. MotznyCK, HolmgrenR (1995) The Drosophila Cubitus Interruptus protein and its role in the Wingless and Hedgehog signal transduction pathways. Mech Dev 52 : 137–150.
Štítky
Genetika Reprodukční medicína
Článek Ribosome Synthesis and MAPK Activity Modulate Ionizing Radiation-Induced Germ Cell Apoptosis inČlánek Fission Yeast Shelterin Regulates DNA Polymerases and Rad3 Kinase to Limit Telomere Extension
Článek vyšel v časopisePLOS Genetics
Nejčtenější tento týden
2013 Číslo 11
-
Všechny články tohoto čísla
- Molecular Recognition by a Polymorphic Cell Surface Receptor Governs Cooperative Behaviors in Bacteria
- The Light Skin Allele of in South Asians and Europeans Shares Identity by Descent
- Ribosome Synthesis and MAPK Activity Modulate Ionizing Radiation-Induced Germ Cell Apoptosis in
- Retrotransposon Silencing During Embryogenesis: Cuts in LINE
- Roles of XRCC2, RAD51B and RAD51D in RAD51-Independent SSA Recombination
- Parallel Evolution of Chordate Regulatory Code for Development
- A Genetic Approach to the Recruitment of PRC2 at the Locus
- Deletion of the Murine Cytochrome P450 Locus by Fused BAC-Mediated Recombination Identifies a Role for in the Pulmonary Vascular Response to Hypoxia
- Elevated Mutagenesis Does Not Explain the Increased Frequency of Antibiotic Resistant Mutants in Starved Aging Colonies
- Deletion of an X-Inactivation Boundary Disrupts Adjacent Gene Silencing
- Interplay between Active Chromatin Marks and RNA-Directed DNA Methylation in
- Recombinogenic Conditions Influence Partner Choice in Spontaneous Mitotic Recombination
- Crosstalk between NSL Histone Acetyltransferase and MLL/SET Complexes: NSL Complex Functions in Promoting Histone H3K4 Di-Methylation Activity by MLL/SET Complexes
- A New Role for the GARP Complex in MicroRNA-Mediated Gene Regulation
- RNAi-Dependent and Independent Control of LINE1 Accumulation and Mobility in Mouse Embryonic Stem Cells
- Loss of DNMT1o Disrupts Imprinted X Chromosome Inactivation and Accentuates Placental Defects in Females
- Inhibition of the Smc5/6 Complex during Meiosis Perturbs Joint Molecule Formation and Resolution without Significantly Changing Crossover or Non-crossover Levels
- Disruption of Lipid Metabolism Genes Causes Tissue Overgrowth Associated with Altered Developmental Signaling
- Translation Initiation Factors eIF3 and HCR1 Control Translation Termination and Stop Codon Read-Through in Yeast Cells
- Recruitment of TREX to the Transcription Machinery by Its Direct Binding to the Phospho-CTD of RNA Polymerase II
- MYB97, MYB101 and MYB120 Function as Male Factors That Control Pollen Tube-Synergid Interaction in Fertilization
- Oct4 Is Required ∼E7.5 for Proliferation in the Primitive Streak
- Contrasted Patterns of Crossover and Non-crossover at Meiotic Recombination Hotspots
- Transposable Prophage Mu Is Organized as a Stable Chromosomal Domain of
- Ash1l Methylates Lys36 of Histone H3 Independently of Transcriptional Elongation to Counteract Polycomb Silencing
- Fine-Mapping the Genetic Association of the Major Histocompatibility Complex in Multiple Sclerosis: HLA and Non-HLA Effects
- Genomic Mechanisms Accounting for the Adaptation to Parasitism in Nematode-Trapping Fungi
- Decoding a Signature-Based Model of Transcription Cofactor Recruitment Dictated by Cardinal Cis-Regulatory Elements in Proximal Promoter Regions
- Removal of Misincorporated Ribonucleotides from Prokaryotic Genomes: An Unexpected Role for Nucleotide Excision Repair
- Fission Yeast Shelterin Regulates DNA Polymerases and Rad3 Kinase to Limit Telomere Extension
- Activin Signaling Targeted by Insulin/dFOXO Regulates Aging and Muscle Proteostasis in
- Activin-Like Kinase 2 Functions in Peri-implantation Uterine Signaling in Mice and Humans
- Demographic Divergence History of Pied Flycatcher and Collared Flycatcher Inferred from Whole-Genome Re-sequencing Data
- Recurrent Tissue-Specific mtDNA Mutations Are Common in Humans
- The Histone Variant His2Av is Required for Adult Stem Cell Maintenance in the Testis
- The Maternal-to-Zygotic Transition Targets Actin to Promote Robustness during Morphogenesis
- Reconstructing the Population Genetic History of the Caribbean
- and Are Required for Growth under Iron-Limiting Conditions
- Whole Genome, Whole Population Sequencing Reveals That Loss of Signaling Networks Is the Major Adaptive Strategy in a Constant Environment
- Neuron-Specific Feeding RNAi in and Its Use in a Screen for Essential Genes Required for GABA Neuron Function
- RNA∶DNA Hybrids Initiate Quasi-Palindrome-Associated Mutations in Highly Transcribed Yeast DNA
- Mouse BAZ1A (ACF1) Is Dispensable for Double-Strand Break Repair but Is Essential for Averting Improper Gene Expression during Spermatogenesis
- Genetic and Functional Studies Implicate Synaptic Overgrowth and Ring Gland cAMP/PKA Signaling Defects in the Neurofibromatosis-1 Growth Deficiency
- DUX4 Binding to Retroelements Creates Promoters That Are Active in FSHD Muscle and Testis
- Pathways-Driven Sparse Regression Identifies Pathways and Genes Associated with High-Density Lipoprotein Cholesterol in Two Asian Cohorts
- PLOS Genetics
- Archiv čísel
- Aktuální číslo
- Informace o časopisu
Nejčtenější v tomto čísle- and Are Required for Growth under Iron-Limiting Conditions
- Genetic and Functional Studies Implicate Synaptic Overgrowth and Ring Gland cAMP/PKA Signaling Defects in the Neurofibromatosis-1 Growth Deficiency
- The Light Skin Allele of in South Asians and Europeans Shares Identity by Descent
- RNA∶DNA Hybrids Initiate Quasi-Palindrome-Associated Mutations in Highly Transcribed Yeast DNA
Kurzy
Zvyšte si kvalifikaci online z pohodlí domova
Současné možnosti léčby obezity
nový kurzAutoři: MUDr. Martin Hrubý
Všechny kurzyPřihlášení#ADS_BOTTOM_SCRIPTS#Zapomenuté hesloZadejte e-mailovou adresu, se kterou jste vytvářel(a) účet, budou Vám na ni zaslány informace k nastavení nového hesla.
- Vzdělávání



