-
Články
Top novinky
Reklama- Vzdělávání
- Časopisy
Top články
Nové číslo
- Témata
Top novinky
Reklama- Videa
- Podcasty
Nové podcasty
Reklama- Kariéra
Doporučené pozice
Reklama- Praxe
Top novinky
ReklamaImmune Modulation with Sulfasalazine Attenuates Immunopathogenesis but Enhances Macrophage-Mediated Fungal Clearance during Pneumonia
Although T cells are critical for host defense against respiratory fungal infections, they also contribute to the immunopathogenesis of Pneumocystis pneumonia (PcP). However, the precise downstream effector mechanisms by which T cells mediate these diverse processes are undefined. In the current study the effects of immune modulation with sulfasalazine were evaluated in a mouse model of PcP-related Immune Reconstitution Inflammatory Syndrome (PcP-IRIS). Recovery of T cell-mediated immunity in Pneumocystis-infected immunodeficient mice restored host defense, but also initiated the marked pulmonary inflammation and severe pulmonary function deficits characteristic of IRIS. Sulfasalazine produced a profound attenuation of IRIS, with the unexpected consequence of accelerated fungal clearance. To determine whether macrophage phagocytosis is an effector mechanism of T cell-mediated Pneumocystis clearance and whether sulfasalazine enhances clearance by altering alveolar macrophage phagocytic activity, a novel multispectral imaging flow cytometer-based method was developed to quantify the phagocytosis of Pneumocystis in vivo. Following immune reconstitution, alveolar macrophages from PcP-IRIS mice exhibited a dramatic increase in their ability to actively phagocytose Pneumocystis. Increased phagocytosis correlated temporally with fungal clearance, and required the presence of CD4+ T cells. Sulfasalazine accelerated the onset of the CD4+ T cell-dependent alveolar macrophage phagocytic response in PcP-IRIS mice, resulting in enhanced fungal clearance. Furthermore, sulfasalazine promoted a TH2-polarized cytokine environment in the lung, and sulfasalazine-enhanced phagocytosis of Pneumocystis was associated with an alternatively activated alveolar macrophage phenotype. These results provide evidence that macrophage phagocytosis is an important in vivo effector mechanism for T cell-mediated Pneumocystis clearance, and that macrophage phenotype can be altered to enhance phagocytosis without exacerbating inflammation. Immune modulation can diminish pulmonary inflammation while preserving host defense, and has therapeutic potential for the treatment of PcP-related immunopathogenesis.
Published in the journal: . PLoS Pathog 6(8): e32767. doi:10.1371/journal.ppat.1001058
Category: Research Article
doi: https://doi.org/10.1371/journal.ppat.1001058Summary
Although T cells are critical for host defense against respiratory fungal infections, they also contribute to the immunopathogenesis of Pneumocystis pneumonia (PcP). However, the precise downstream effector mechanisms by which T cells mediate these diverse processes are undefined. In the current study the effects of immune modulation with sulfasalazine were evaluated in a mouse model of PcP-related Immune Reconstitution Inflammatory Syndrome (PcP-IRIS). Recovery of T cell-mediated immunity in Pneumocystis-infected immunodeficient mice restored host defense, but also initiated the marked pulmonary inflammation and severe pulmonary function deficits characteristic of IRIS. Sulfasalazine produced a profound attenuation of IRIS, with the unexpected consequence of accelerated fungal clearance. To determine whether macrophage phagocytosis is an effector mechanism of T cell-mediated Pneumocystis clearance and whether sulfasalazine enhances clearance by altering alveolar macrophage phagocytic activity, a novel multispectral imaging flow cytometer-based method was developed to quantify the phagocytosis of Pneumocystis in vivo. Following immune reconstitution, alveolar macrophages from PcP-IRIS mice exhibited a dramatic increase in their ability to actively phagocytose Pneumocystis. Increased phagocytosis correlated temporally with fungal clearance, and required the presence of CD4+ T cells. Sulfasalazine accelerated the onset of the CD4+ T cell-dependent alveolar macrophage phagocytic response in PcP-IRIS mice, resulting in enhanced fungal clearance. Furthermore, sulfasalazine promoted a TH2-polarized cytokine environment in the lung, and sulfasalazine-enhanced phagocytosis of Pneumocystis was associated with an alternatively activated alveolar macrophage phenotype. These results provide evidence that macrophage phagocytosis is an important in vivo effector mechanism for T cell-mediated Pneumocystis clearance, and that macrophage phenotype can be altered to enhance phagocytosis without exacerbating inflammation. Immune modulation can diminish pulmonary inflammation while preserving host defense, and has therapeutic potential for the treatment of PcP-related immunopathogenesis.
Introduction
Pneumocystis (Pc) is an opportunistic fungal respiratory pathogen that causes life-threatening pneumonia in patients suffering from defects in cell-mediated immunity, including those with acquired immunodeficiency syndrome (AIDS) and immunosuppression secondary to chemotherapy or organ transplantation. Pneumocystis pneumonia (PcP) remains a leading cause of death among HIV-infected patients and a significant cause of AIDS-related mortality and morbidity [1]. For example, mortality rates of 50% or higher have been reported for AIDS patients with severe PcP [2], [3], and despite major advances in health care, the mortality associated with PcP has changed little over the past 25 years. In addition, as more powerful anti-inflammatory treatments are developed for various underlying diseases, more cases of PcP are occurring in non-HIV patients and in previously unreported clinical settings [4]–[6]. Recent studies also indicate that Pc colonization can exacerbate chronic obstructive pulmonary disease [7]. Therefore, improving the treatment of patients suffering from both HIV - and non HIV-related PcP remains a central concern of the health care community.
Although the direct pathogenic capabilities of the Pneumocystis organism itself are poorly understood, the role of the host's immune response as a major contributor to PcP-related lung injury has come to the forefront. In patients, the clinical severity of PcP is dictated by the degree of pulmonary inflammation, rather than by the organism lung burden [8]–[14]. Specifically, T cell and neutrophilic responses have been linked to PcP-related lung injury in patients [10], [15]. A clinical example of the immunopathogenic nature of PcP is the severe disease that has been reported in AIDS patients following successful anti-retroviral treatment [16]–[18]. This distinct clinical syndrome, termed Immune Reconstitution Inflammatory Syndrome (IRIS) or Immunorestitution Disease (IRD), occurs when CD4+ T cell-mediated immunity is restored following a period of immunosuppression. The recovery of immune function restores protective adaptive immunity, but does so at the cost of initiating a severe immunopathological response to a pre-existing Pc infection. An IRIS-like presentation of PcP has also been described in non-HIV infected patients following the successful tapering of steroid therapy or bone marrow engraftment [19], [20]. Importantly, patients with non-HIV presentations of PcP and IRIS seem to develop a more fulminant and acutely immunopathogenic disease than patients with a classical AIDS-related presentation in which CD4+ T cell function is chronically and profoundly depressed [10], [21]–[24].
The immunopathogenesis of PcP has been confirmed by controlled studies in Pc-infected severe combined immunodeficient (SCID) mice. Following adoptive transfer of normal splenocytes these mice develop disease that is pathologically similar to clinical reports of IRIS. When the host's immune system is restored, an intense T cell-mediated immune response brings about organism clearance with the undesired consequence of severe lung damage and respiratory deterioration [25]–[30]. Our laboratory has demonstrated that CD4+ T cells predominate in the lungs at the time of maximal injury and that depleting this population prevents the onset of acute disease [28], [31]. Other studies have demonstrated that CD4+ T cells are robustly pathogenic in the setting of immune recovery and PcP [26], [27], [32], [33]. While existing evidence unquestionably demonstrates that T cell responses are directly involved in both the clearance of Pc and the generation of immune-mediated lung injury, the specific downstream effector mechanisms have not been elucidated. Alveolar macrophages (AMs) are likely involved in both of these processes, but their in vivo role remains incompletely defined.
Sulfasalazine (SSZ) is a potent anti-inflammatory drug commonly used to treat the inflammatory consequences of Crohn's disease and Rheumatoid Arthritis [34]–[36]. SSZ modulates immune responses by altering macrophage and T cell responses [37]–[39]. Many effects of SSZ are related to its function as a potent inhibitor of NF-κB [39], [40], a signaling pathway that is important for inflammatory responses to Pc [38], [41], [42], [43]. Therefore, we hypothesized that the potent immunomodulatory properties of SSZ could alleviate lung injury and improve outcome in a mouse model of PcP-related IRIS. SSZ was highly effective for attenuating the immune-mediated lung injury associated with PcP, with the unexpected finding that SSZ also accelerated fungal clearance. Moreover, we developed a multispectral imaging flow cytometer-based method to assess Pc phagocytosis in vivo. Using this technology we established that the macrophage is the downstream effector for CD4+ T cell-dependent clearance of Pc from the lung, and that SSZ enhances clearance by promoting AM phagocytosis.
Results
Sulfasalazine markedly reduces the severity of PcP
SSZ is used clinically to treat conditions in which inflammation is integral to pathogenesis. To test the efficacy of SSZ for reducing the severity of PcP-related IRIS, infected SCID mice were immunologically reconstituted with wild type splenocytes, and then treated with either SSZ or PBS vehicle beginning at day 1 post-reconstitution (PR). Respiratory rates and body weights were monitored non-invasively, and dynamic lung compliance and resistance were measured at 13, 18 and 25 days PR. These times correspond to the early, peak, and resolution phases of PcP in this model. As expected, the PBS-treated mice with IRIS exhibited progressive disease that was characterized by dramatic weight loss and elevated respiratory rates. These mice lost an average of 17±2% body weight by day 12, and 24±3% by day 16 PR. Thereafter, the mice began to gain weight coincident with the resolution of disease (Figure 1A). These mice also exhibited elevated respiratory rates, which increased dramatically to an average of 441±9 respirations per minute at day 12 and 487±13 respirations per minute by day 17 PR (Figure 1B). In contrast, the SSZ-treated mice exhibited only slight variations in body weight and respiratory rate over this same period, and had a generally healthy appearance.
Fig. 1. SSZ treatment reduces the severity of PcP-related IRIS. 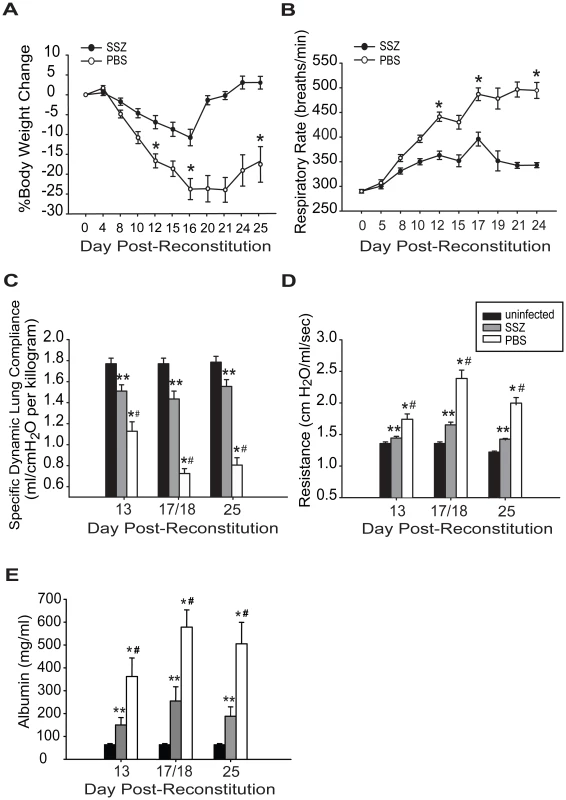
Pc-infected SCID mice were immunologically reconstituted and then treated with PBS or SSZ beginning at day 1 post-reconstitution. (A and B) Body weight and respiratory rates were monitored non-invasively. (C, D, E) Dynamic lung compliance, lung resistance and BAL albumin content were measured at 13, 18, and 25 days post-reconstitution. Values are mean ±1 standard error measurement (SEM) (n≥15 for day 13, n≥37 for day 18, and n≥15 for day 25). ** and #, P<0.05 as compared to uninfected mice at the same time point. *, P<0.05 as compared to SSZ treated mice at the same time point. Mean represents combined data from eight independent experiments. Direct pulmonary function measurements were taken at days 13, 18, and 25 PR. Dynamic lung compliance and lung resistance are derived from pressure and volume measurements recorded on live ventilated mice. Lung compliance is a measure of the lungs ability to stretch during the respiratory cycle, and is considered a measure of alveolar health. Mice with PcP have reduced compliance compared to healthy mice, indicating that the lungs are less elastic and generate greater pressure during respiration. Lung resistance is a measure of air flow limitation to and from the gas exchange surface, and can be negatively affected by airway and alveolar inflammation. Mice with PcP have increased resistance compared to healthy mice. Both of these measures are good indicators of the severity of PcP. PBS-treated mice with IRIS developed a drastic deterioration of pulmonary function over the course of the study. A severe reduction in dynamic lung compliance was observed by day 13 PR, and by day 18 these mice demonstrated a 59% deficit in lung compliance (Figure 1C). In contrast, the SSZ-treated mice suffered only a 19% reduction in lung compliance over this same period, and recovered to nearly normal pulmonary function by day 25 PR. Similarly, a dramatic difference in resistance values was observed between the SSZ - and PBS-treated IRIS mice (Figure 1D). The SSZ-treated mice exhibited significantly lower lung resistance than the vehicle group at all time points, supporting the conclusion that SSZ decreases the magnitude of the pulmonary function deficits associated with PcP, and attenuates overall disease severity.
To determine the effect of SSZ treatment on epithelial damage and alveolar permeability during PcP, albumin content was measured in the bronchoalveolar lavage (BAL) fluid. Elevated levels of albumin in the BAL fluid indicates damage to the tight junctions between alveolar epithelial cells and serves as a marker for the severity of PcP. PBS-treated mice with PcP had significantly elevated albumin levels on days 13 and 18 PR as compared with normal uninfected mice (Figure 1E). In contrast, the SSZ-treated mice had lower albumin levels than PBS-treated mice at both time points (Figure 1E). While albumin levels returned toward baseline in both groups by day 25, they remained significantly lower in the SSZ-treated mice. These data demonstrate that SSZ attenuates damage to the alveolar-capillary barrier, which contributes to the preservation of pulmonary function during PcP.
Sulfasalazine reduces pulmonary inflammation during PcP-related IRIS
Total cell counts, differentials, and flow cytometry were also performed on BAL cells from experimental mice. PBS-treated IRIS mice had significantly elevated numbers of total BAL cells compared to SSZ-treated mice at all time points (Table 1). Differential staining revealed that the reduced number of cells in SSZ-treated mice relative to PBS-treated mice was mainly a reflection of fewer lymphocytes and neutrophils at all time points (Table 1). As neutrophil numbers are predictive of disease severity, fewer neutrophils in SSZ-treated mice was also an indicator of less severe disease. Despite a reduction in total cells, it was notable that SSZ-treated mice had more AMs than PBS-treated mice at day 18. Flow cytometry analyses revealed that SSZ-treated mice had fewer CD4+ and CD8+ T cells than PBS-treated mice at 13 and 18 days PR. By day 25 PR the reduced BAL cells in SSZ-treated mice was mainly a reflection of reduced CD4−/CD8− lymphocytes (possibly B cells) and neutrophils.
Tab. 1. Cellular composition of BAL fluid* from SSZ- and PBS-treated IRIS mice. 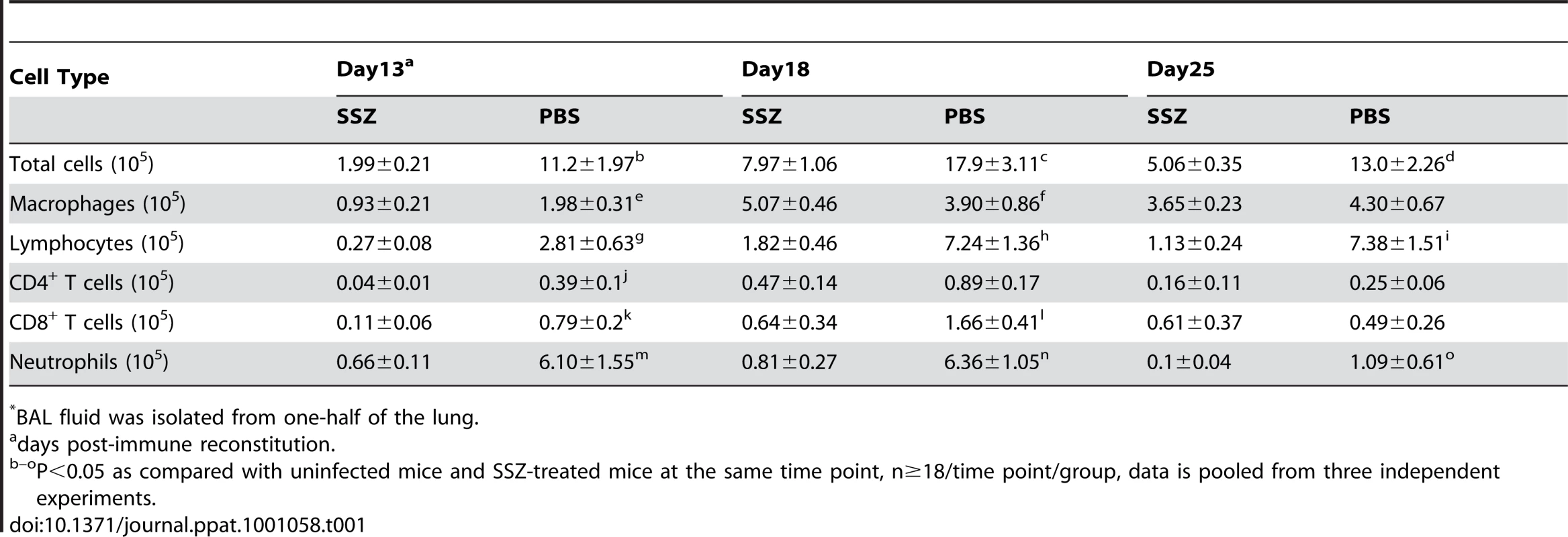
BAL fluid was isolated from one-half of the lung. Histological examination of hematoxylin and eosin stained lung sections showed that the PBS-treated mice exhibited a more intense, wide-spread inflammatory response than the SSZ-treated mice. Pulmonary inflammation in PBS-treated mice was characterized by the accumulation of monocytic and polymorphonuclear aggregates throughout the alveoli, although the inflammation appeared less intense by day 25 (Figure 2A–C). In contrast, evidence of inflammation in the SSZ-treated mice at days 13 and 18 PR was mostly limited to perivascular and peribronchial regions, and the alveoli were mainly free from cellular infiltrates (Figure 2D, E). Furthermore, the inflammation was almost completely resolved by day 25 in the SSZ-treated mice (Figure 2F).
Fig. 2. SSZ reduces pulmonary inflammation during PcP-related IRIS. 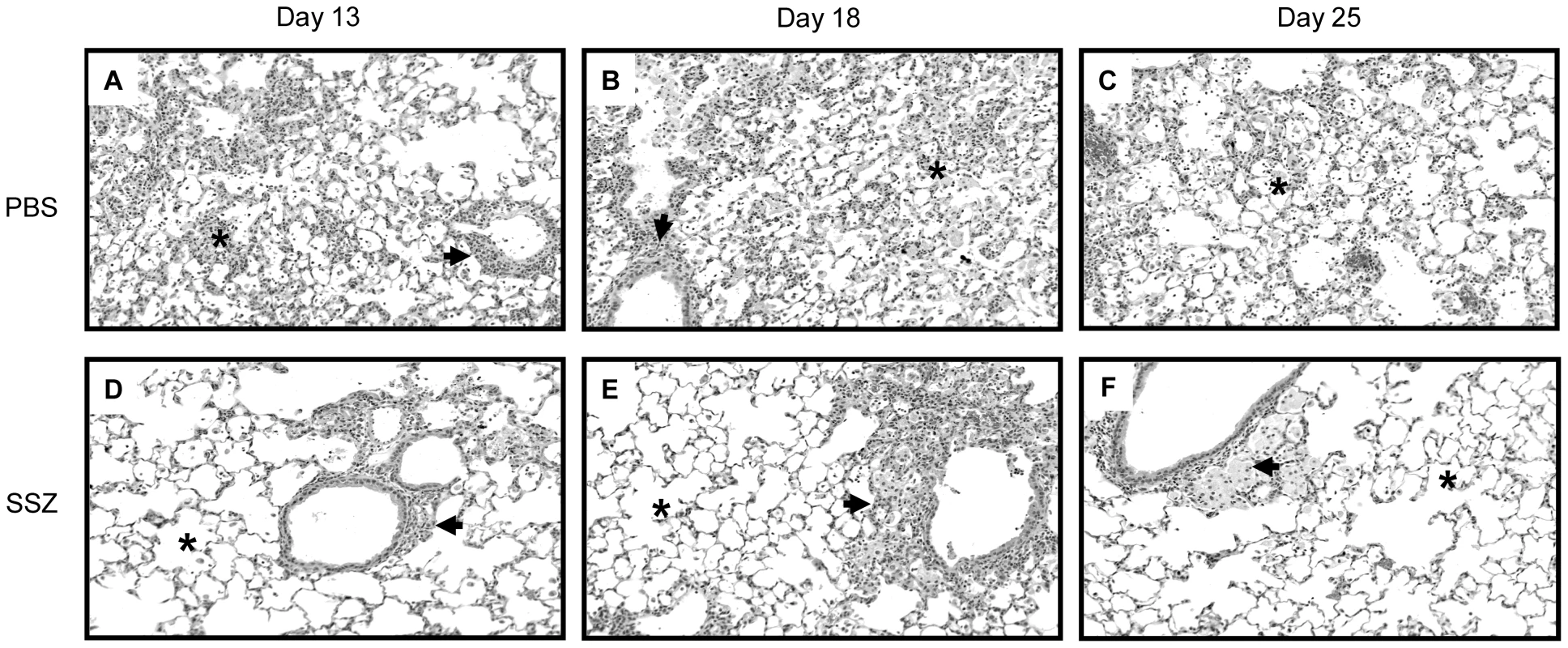
Pc-infected SCID mice were immunologically reconstituted, and then treated with PBS (Panels A–C) or SSZ (Panels D–F) beginning at day 1 post-reconstitution. Lung sections from mice at days 13 (A, D), 18 (B, E) or 25 (c, F) post-reconstitution were stained with Hematoxylin and Eosin. Representative pictures were taken by microscopy under 40× magnification. Arrows denote peribronchiolar regions in each section, while asterisks denote alveolar regions. Sulfasalazine inhibits lung chemokine and cytokine production during PcP
Elevated cytokine production is a characteristic of PcP-related inflammation. To determine whether the reduced severity of PcP observed in SSZ-treated mice was associated with blunted chemokine and cytokine production in the lung, MCP-1, RANTES, TNF-α, and IFN-γ levels were measured in the BAL fluid. Cytokine and chemokine levels were elevated above control levels in the lungs of PBS-treated IRIS mice, consistent with the physiological and histological data showing severe inflammatory disease. In contrast, lung levels of MCP-1, RANTES, TNF-α and IFN-γ in the SSZ-treated group were all significantly lower than the PBS-treated group at all time points (Figure 3A–D). Reduced cytokine levels in the SSZ-treated mice were associated with reduced inflammatory disease in these mice.
Fig. 3. SSZ reduces inflammatory chemokine and cytokine production during PcP-related IRIS. 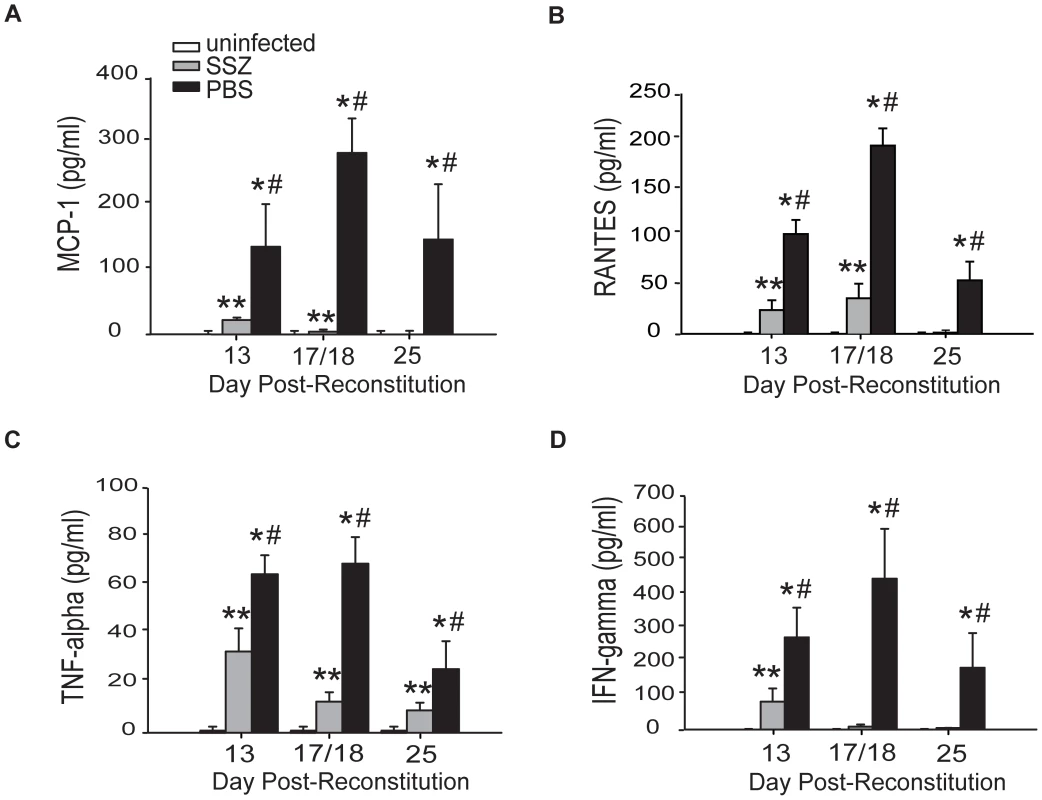
Pc-infected SCID mice were immunologically reconstituted, and then treated with PBS or SSZ. (A, B, C, and D) MCP-1, RANTES, TNF-α, and IFN-γ levels were measured in the BAL fluid. Values are mean ±1 SEM (n≥15/time point/group). ** and #, P<0.05 as compared to uninfected mice at the same time point. *, P<0.05 as compared to SSZ treated mice at the same time point. Mean represents combined data from three independent experiments. Sulfasalazine enhances the host's ability to clear the Pc infection
To study the effect of SSZ on fungal clearance, the lungs of experimental mice were examined for Pc burden using real-time PCR. Both PBS - and SSZ-treated mice had significant lung Pc burdens on day 13 PR (Figure 4A). Although the PBS-treated mice showed little reduction in Pc burden by day 18 (log 6.77±0.07), the SSZ-treated mice exhibited a 4-log reduction between days 13 and 18 PR (log 2.25±0.42) (Figure 4A). By day 25 PR, Pc burdens in the PBS-treated IRIS mice were reduced by an average of 2 logs, while Pc was undetectable in nearly all individual SSZ-treated animals (Figure 4A). The above data were combined from 8 independent experiments. The enhanced clearance of Pc in SSZ-treated mice was confirmed by enumerating Pc cysts in the lung homogenates with Gomori's methenamine silver stain (data not shown). Therefore, SSZ accelerates the fungal clearance kinetics in this model.
Fig. 4. SSZ enhances Pc clearance through a CD4-dependent mechanism. 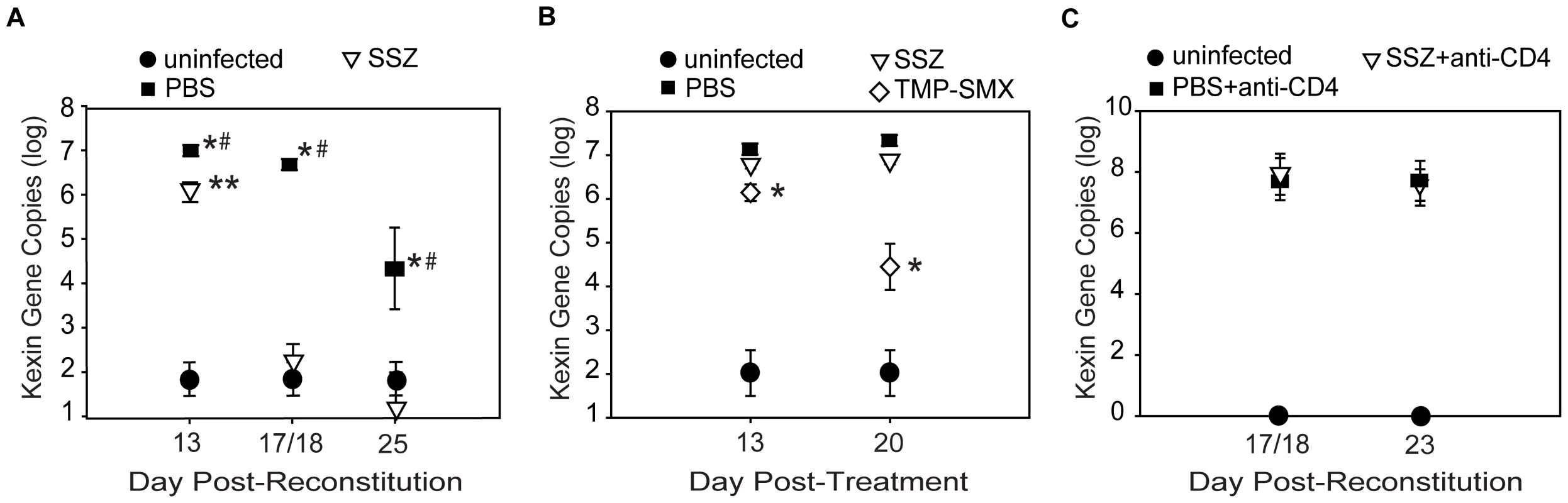
(A) Pc burden in immune reconstituted SCID mice that received SSZ- (∇) or PBS (▪). Values are mean ±1 SEM (n≥15 for day 13, n≥37 for day 18, and n≥15 for day 25). ** and #, P<0.05 as compared to uninfected mice at the same time point. *, P<0.05 as compared to SSZ treated mice at the same time point. Mean represents combined data from eight independent experiments. (B) Pc burden in non-reconstituted SCID mice after 13 and 20 days of SSZ (∇), PBS (▪), or TMP-SMX (◊) treatment. Values are mean ±1 SEM (n≥11 for day 13, and n≥17 for day 20). *, P<0.05 as compared to SSZ and PBS treated mice at the same time point. Mean represents combined data from three independent experiments. (C) Pc burden in immune reconstituted, CD4-depleted SCID mice receiving SSZ (∇) or PBS (▪). Values are mean ±1 SEM (n≥8 for day 18 and n≥14 for day 23). Mean represents combined data from four independent experiments. Enhanced Pc clearance in SSZ-treated mice requires CD4+ T cell mediated immunity
Adaptive immunity is critical for the clearance of Pc from the lungs. To determine whether the mechanism by which SSZ enhances Pc clearance requires the adaptive immune system, non-reconstituted Pc-infected SCID mice were administered either PBS vehicle, SSZ (200 mg/kg/day), or trimethoprim sulfamethoxazole (TMP-SMX) (16 mg/kg/day). As expected, PBS-treated SCID mice had high Pc burdens at both 13 and 20 days post-treatment (Figure 4B). Similarly, the SSZ-treated group also had high Pc burdens at both time points and did not clear the Pc infection (Figure 4B), demonstrating that SSZ had no direct Pc killing effect. In contrast, the TMP-SMX-treated group showed reduced Pc burden at both time points (Figure 4B). The role of CD4+ T cells in host defense against Pc has been well-documented. To determine whether the mechanism of SSZ-enhanced clearance is CD4+ T cell-dependent, Pc-infected SCID mice were immune reconstituted and then CD4+ T cell depleted. The Pc burdens of PBS - and SSZ-treated mice were determined at days 18 and 25 PR. Neither PBS - nor SSZ-treated mice cleared the Pc infection in the absence of CD4+ T cells (Figure 4C). Thus, the mechanism by which SSZ enhances Pc clearance requires a CD4+ T cell response. These data demonstrate that SSZ does not have direct anti-Pc activity, and that SSZ-enhanced clearance arises from modulation of the CD4+ T cell-mediated immune response.
Sulfasalazine enhances CD4+ T cell-dependent AM phagocytosis
Although it has been inferred that AMs are responsible for CD4+ T cell-mediated clearance of Pc, there is little in vivo evidence to support this. Therefore, a multispectral imaging flow cytometry-based method was developed to quantify AM phagocytosis of Pc in vivo. The Amnis ImageStream, which combines digital imaging with traditional flow cytometry, allowed for dual staining of AM surface markers and internalized Pc. Because of the large number of cells that can be rapidly evaluated, a quantitative assessment of AM internalization of Pc was generated. This method was used to assess AM phagocytic activity in SSZ and PBS treated animals at various time points. CD11c was used as a marker for AMs, and a pool of five monoclonal antibodies that recognize the surface of mouse Pc were used to determine internalization of Pc relative to the AM. Figure 5 (Panel A) shows representative images of brightfield (BF), CD11c (green), Pc (red), and CD11c-Pc merged. The no internalization control is a representative cell with CD11c signal, but no Pc internalization (Figure 5A). Control staining was performed without inclusion of anti-Pc antibodies. As expected, no red signal was observed in these cells. The flow cytometer data were then quantified for AMs from individual SSZ or PBS treated mice at days 13, 17/18, and 21 PR. The percentage of AMs with internalized Pc in SSZ-treated mice was significantly higher than in PBS-treated mice at day 17/18 PR (Figure 5B). Furthermore, when the absolute number of AM with internalized Pc was determined, the difference was even more striking. SSZ-treated mice had a 9-fold greater number of AM with internalized Pc than PBS-treated mice at days 17/18 PR (Figure 5C). Importantly the number of AM with internalized Pc increased dramatically in PBS-treated mice at day 21 PR, coincident with Pc clearance and recovery from infection. These data indicate that AM-mediated clearance also occurs in PBS-treated mice, but that SSZ treatment accelerates this T cell-dependent process.
Fig. 5. SSZ enhances CD4+ T cell-dependent AM phagocytosis of Pc. 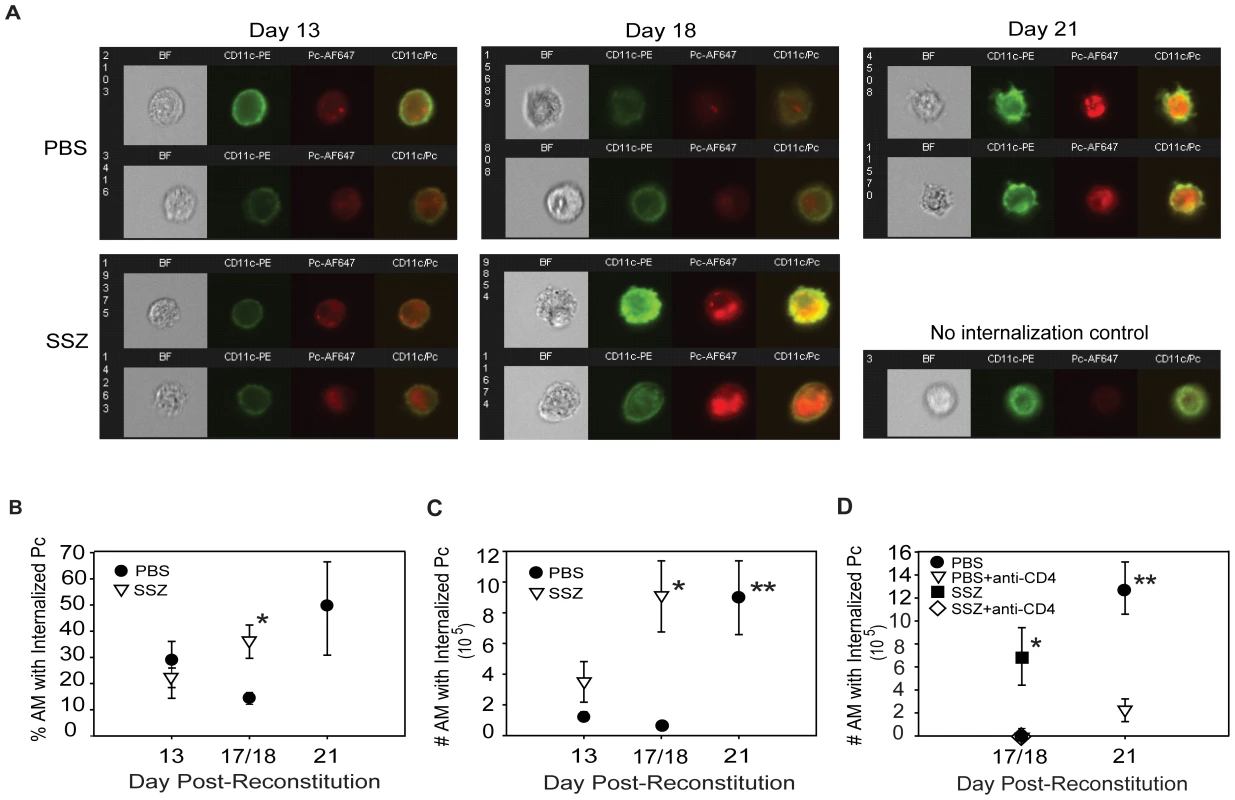
BAL cells were collected from PBS- and SSZ- treated mice at day 13, 17 or 18 (17/18) and 21 post-reconstitution. Cells were stained with antibodies specific for CD11c (green) and Pc (red). (A) Imaging flow cytometry was used to quantify Pc internalization by AMs. Representative images of brightfield (BF), CD11c, Pc, and merged CD11c/Pc are shown for SSZ or PBS-treated mice following immune reconstitution. The no internalization control is a representative CD11c+ cell without internalized Pc. ImageStream IDEAS software was used to quantify (B) percentile and (C) number of AMs with Internalized Pc in immune reconstituted SCID mice treated with SSZ (∇) or PBS (•). Values are mean ±1 SEM (n≥5 for day 13, n≥9 for day 17/18, and n≥9 for day 21). *, P<0.05 as compared to PBS-treated mice at the same time point. **, P<0.05 as compared to PBS-treated mice at day 17/18. Mean represents combined data from three independent experiments. (D) Quantification of in vivo Pc phagocytosis in the presence or absence of CD4+ T cells. Reconstituted SCID mice were treated with PBS- (•), PBS + anti-CD4 (∇), SSZ (▪), or SSZ + anti-CD4 (⋄). Phagocytosis was measured at 17/18 or 21 days post-reconstitution. Values are mean +1 SEM (n≥5 for day 13, n≥9 for day 17/18, and n≥8 for day 21). *, P<0.05 as compared to PBS treated, PBS + anti-CD4-treated, and SSZ + anti-CD4-treated mice at the same time point. **, P<0.05 as compared to PBS + anti-CD4-treated mice at the same time point. Mean represents combined data from three independent experiments. Note that values for PBS treated, PBS + anti-CD4-treated, and SSZ + anti-CD4-treated mice at day 17/18 are all indistinguishable, and the symbols overlap. A CD4+ T cell response was required for Pc clearance in both PBS and SSZ-treated animals. To determine whether CD4+ T cells are required for AM phagocytosis of Pc, infected SCID mice were immune reconstituted and given SSZ, SSZ plus anti-CD4 antibody, PBS, or PBS plus anti-CD4 antibody. As expected, SSZ-treated mice exhibited a large increase in AM phagocytosis of Pc at day 17/18 PR (Figure 5D), and also exhibited significant fungal clearance at this time with an average Pc burden of log 2.8±1.1 (as assessed by kexin gene copies). In contrast, SSZ-treated mice that were depleted of CD4+ T cells displayed nearly undetectable levels of AM phagocytosis of Pc (Figure 5D), and maintained high average Pc lung burdens of log 7.5±0.1 (p<0.05 compared to SSZ-treated mice). Likewise, PBS-treated animals displayed increased AM phagocytosis at day 21 PR, but phagocytosis was nearly undetectable in the absence of CD4+ T cells (Figure 5D). Consistent with these results, PBS-treated mice had lower average Pc burdens than the PBS plus anti-CD4 group (log 6.7±0.5 versus log 7.6±0.01; p<0.05). Together, these data demonstrate that AM phagocytosis is the effector mechanism for CD4+ T cell-dependent clearance of Pc from the lung, and that SSZ alters the lung immune response in a manner that accelerates the macrophage-mediated phagocytosis of Pc.
In order to validate the ImageStream data, confocal microscopy was used to confirm the internalization and localization of Pc within AMs. BAL cells were recovered from PBS - and SSZ-treated mice at time points when AMs are actively phagocytosing Pc. Based on the data in Figure 5, the time points chosen were day 21 PR for PBS-treated mice, and day 17 PR for SSZ-treated mice. Cells were stained with DAPI (blue), anti-Pc antibody (green), anti-CD11c antibody (gray), and anti - lysosomal-associated membrane protein-1 (LAMP-1) antibody (red). LAMP-1 is a lysosome-specific protein, and was used to co-localize Pc staining to the phagolysosome inside AMs. As shown in Figure 6, we found that Pc signal co-localized with LAMP-1 signal inside CD11c+ AMs from both PBS-(Day 21) and SSZ-(Day 17) treated animals. The “Control” Panel shows a direct comparison between an AM with Pc and one without Pc next to each other in the same field. The cell without Pc (top) shows more diffuse LAMP-1 staining, while the bottom cell shows more focal LAMP-1 staining that co-localizes with Pc signal. This panel demonstrates differential staining and specificity of LAMP-1 and Pc antibodies. These data validate the quantitative ImageStream data as a measure of Pc internalization.
Fig. 6. Co-localization of internalized Pc with the lysosomal protein LAMP-1. 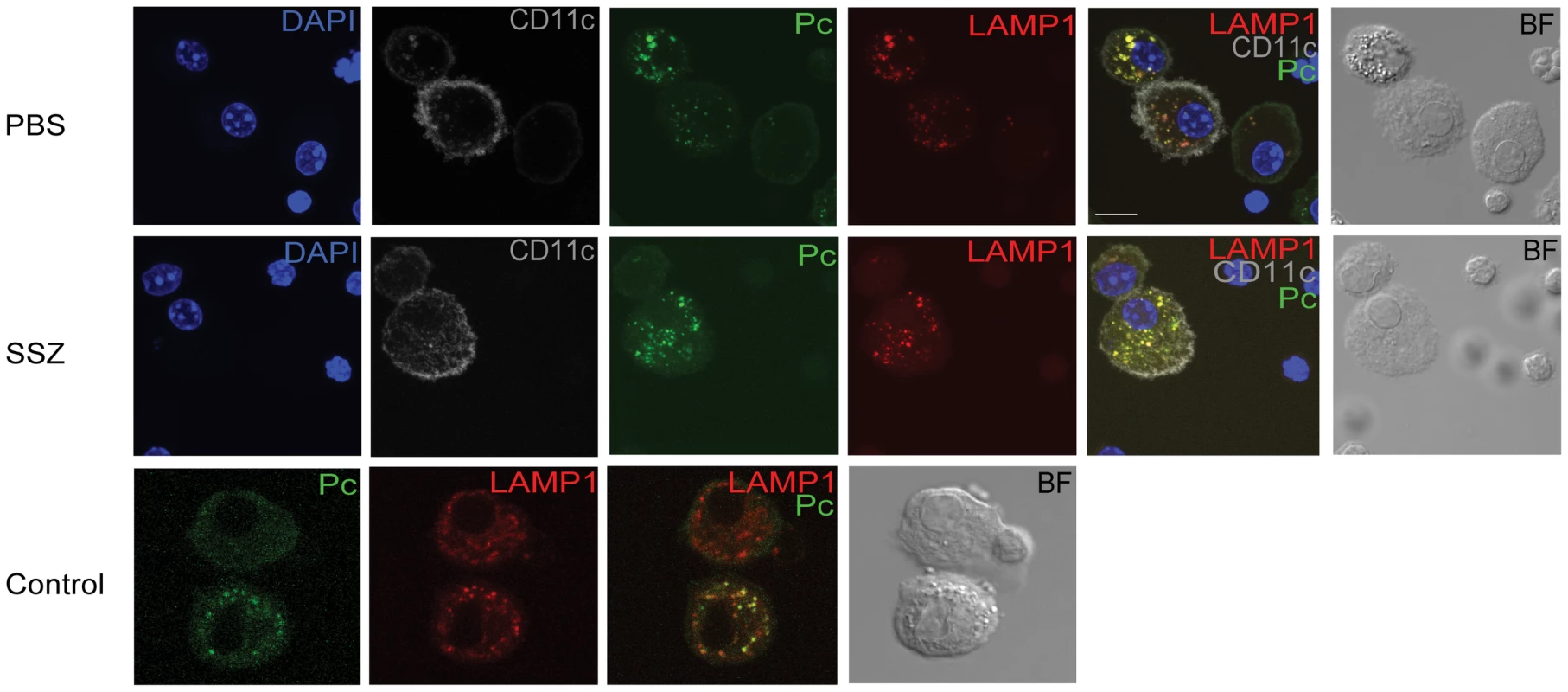
BAL cells were collected from PBS-treated mice at day 21 and SSZ-treated mice at day 17 post-reconstitution. Cells were stained with antibodies specific for CD11c (gray), Pc (green) and LAMP-1 (red). Nuclei were stained with DAPI (blue). Control image compares two AM in the same field; one with internalized Pc and one without. The scale bar represents 10 µM. Images are representative of two independent experiments. Sulfasalazine promotes a TH2 lung cytokine environment and alternative activation of alveolar macrophages
Macrophages are immune effector cells for T cell-dependent responses, and distinct macrophage phenotypes with differential effects on host defense and inflammation have been identified [44], [45]. Classically activated macrophages (CAM) are induced by TH1 cytokines, produce inducible nitric oxide synthase (INOS), and are pro-inflammatory. In contrast, alternatively activated macrophages (AAM) are induced by TH2 cytokines, produce arginase (ARG), are highly phagocytic, and produce anti-inflammatory mediators. Our studies have demonstrated that SSZ has profound effects on CD4+ T cell-dependent macrophage responses to Pc. Therefore, to determine whether SSZ alters PcP-related IRIS by modulating the polarity of the T helper response and subsequent AM effector phenotype, TH cytokine levels and macrophage activation state were assessed in experimental mice. SSZ treatment caused a dramatic decrease in lung IFN-γ production (Figure 3), with a concomitant increase in lung IL-4 production compared to PBS-treated mice (Figure 7A). Thus SSZ produced a significant shift in the IL-4 to IFN-γ ratio in the lungs (Figure 7B), effectively creating a pro-TH2 lung cytokine environment. In contrast, PBS-treated IRIS mice exhibited a pro-TH1 lung cytokine environment.
Fig. 7. SSZ promotes a TH2 cytokine environment during PcP-related IRIS. 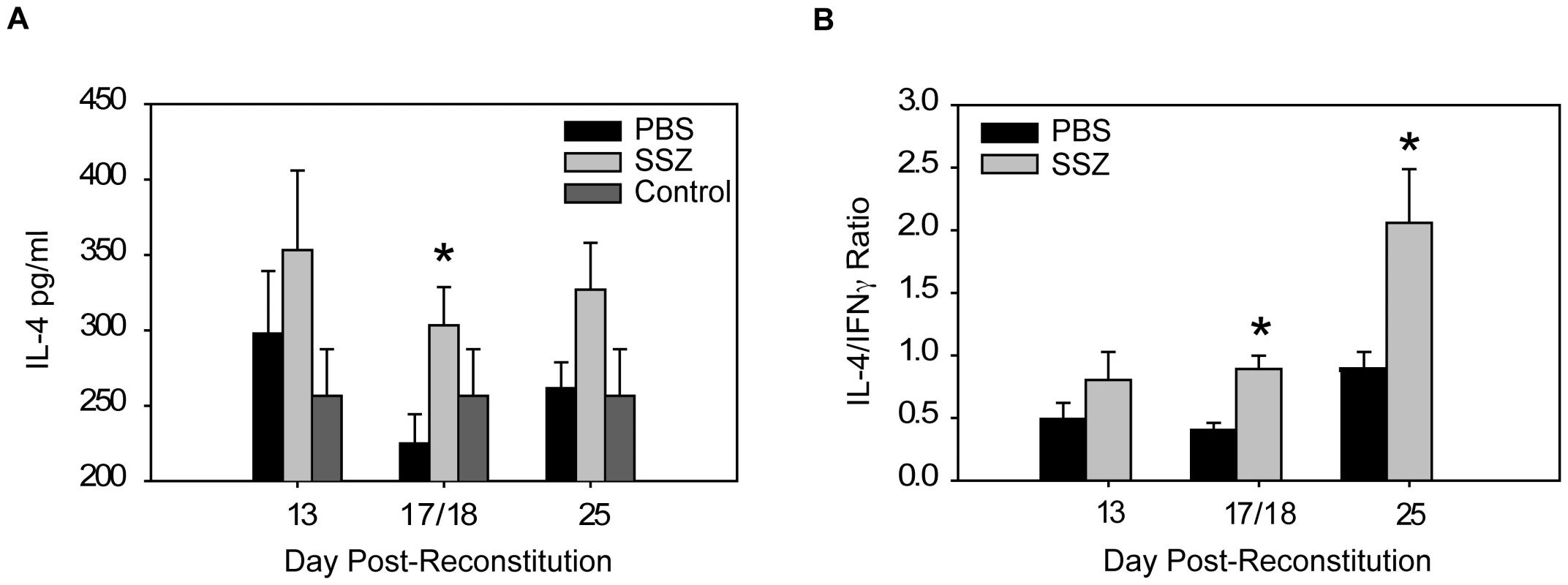
Pc-infected SCID mice were immunologically reconstituted, and then treated with PBS or SSZ. (A) IL-4 levels and (B) IL-4/IFN-γ ratios were measured in the lung homogenates of experimental mice at indicated time points. Values are mean ±1 SEM (n≥10/time point/group). *, P<0.05 as compared to PBS treated mice at the same time point. Mean represents combined data from five independent experiments. To determine whether SSZ treatment altered AM phenotype, AMs from SSZ - and PBS-treated IRIS mice at day 13 PR were assessed for INOS and ARG protein expression. Because other cell types were present in the BAL fluid from mice with PcP, CD11c was used as a surface marker for AMs. CD11c positive AMs from PBS-treated IRIS mice stained intensely for INOS (Figure 8A), but weakly for ARG (Figure 8B). In contrast, CD11c positive AMs from SSZ-treated mice stained weakly for INOS (Figure 8A), but intensely for ARG (Figure 8B). Measurement of mean fluorescent intensity of INOS and ARG staining in CD11c positive cells was used to quantify the differential expression of CAM and AAM markers in SSZ - and PBS-treated mice (Figure 8C, D). These data demonstrate that SSZ promotes alternative activation of AMs, which is associated with reduced immunopathogenesis but enhanced phagocytosis and accelerated fungal clearance.
Fig. 8. SSZ promotes an alternatively activated macrophage phenotype during PcP-related IRIS. 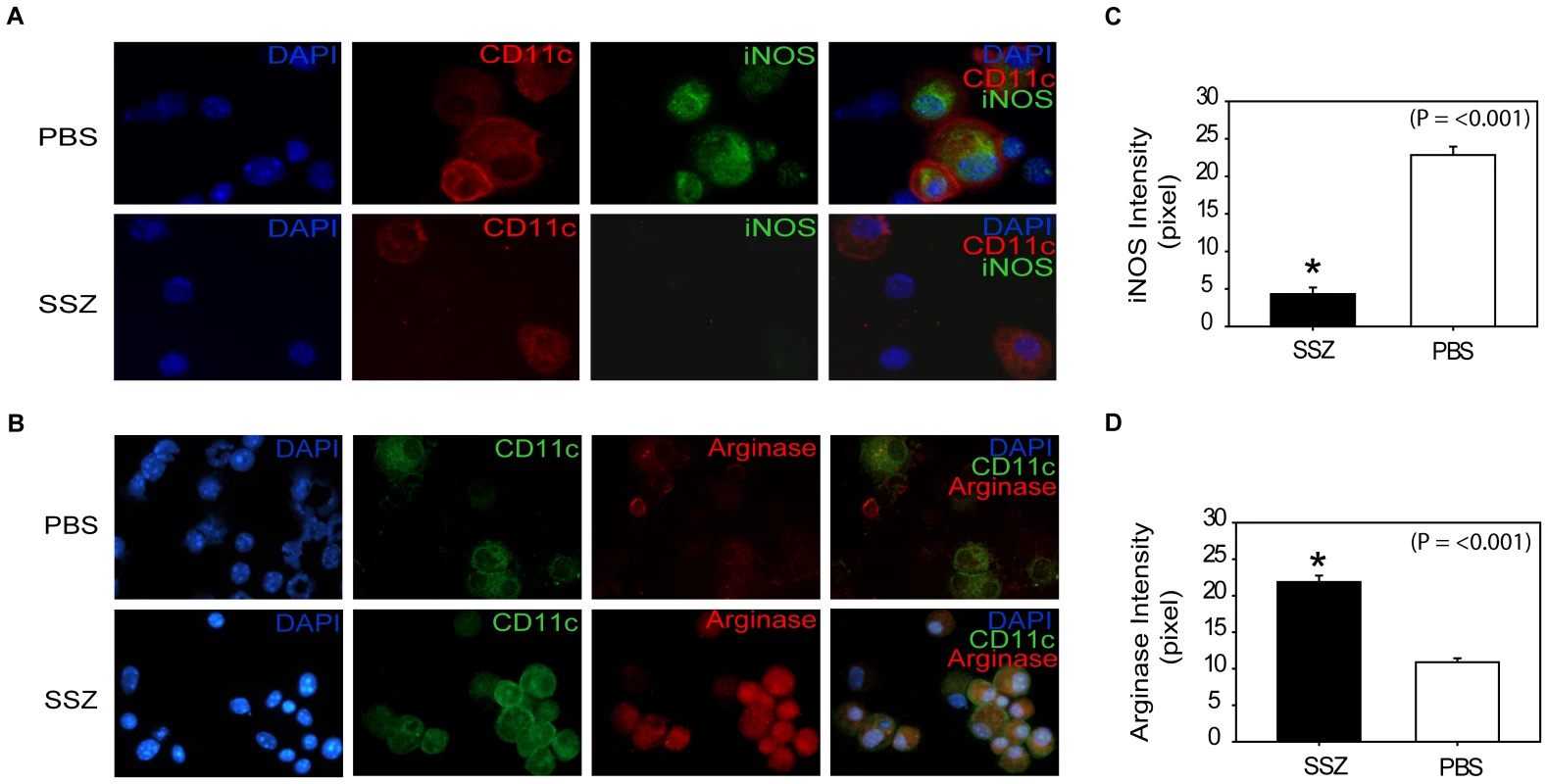
BAL cells were collected from PBS- and SSZ- treated mice at day 13 post-reconstitution. (A) Cells were stained with antibodies specific for CD11c (red) and iNOS (green). (B) Cells were stained with antibodies specific for CD11c (green) and Arginase (red). Nuclei were stained with DAPI (blue). Images shown are 40× magnification. ImageJ software was used to quantify the mean fluorescent intensity of iNOS (C) and Arginase (D) staining of CD11c positive cells. Values are mean ±1 SEM (Panel A, n = 23 for PBS and n = 58 for SSZ; Panel B, n = 44 for PBS and n = 105 for SSZ). *, P<0.05 as compared to PBS treated mice at the same time point. Mean represents combined data from two independent experiments. Discussion
IRIS is a clinical manifestation of PcP that occurs in certain patients when cell-mediated immunity is restored following a period of immune suppression and infection [16], [18], [19], [46], [47]. The resulting acute pulmonary inflammatory response restores host defense, but also produces severe pathology. The current study demonstrates that modulation of the immune response dramatically reduces the severity of PcP-related IRIS. The SSZ-mediated reduction in physiological impairment was associated with abrogated cytokine responses, reduced immune cell recruitment to the lung, and reduced histological evidence of inflammation. Unexpectedly, SSZ did not impair, but actually enhanced the host's ability to clear the Pc infection, indicating that the immune pathways leading to injury are at least partly independent of the pathways leading to clearance. Macrophages are equipped to recognize and eliminate pathogens as well as promote or resolve inflammation. To test the hypothesis that SSZ enhances Pc clearance via downstream functional alteration of AMs, a multispectral imaging flow cytometry-based method was developed to assess and quantify the in vivo Pc phagocytic activity of AM. This technology demonstrated that AM are effector cells for CD4+ T-cell mediated Pc clearance, and that SSZ enhances clearance by accelerating the AM phagocytic response. Subsequent studies found that SSZ promotes alternative activation of AMs, which is associated with reduced immunopathogenesis, but enhanced phagocytosis and fungal clearance. These data demonstrate that AM can be phenotypically modified to enhance fungal phagocytosis and clearance without ehnancing their pro-inflammatory properties, and also provide in vivo evidence that macrophage phagocytosis is the mechanism of CD4+ T cell-dependent Pc clearance from the lung.
SSZ is a clinically important immunomodulatory therapy for inflammatory diseases such as inflammatory bowel disease and rheumatoid arthritis [34]–[36]. Most of the beneficial effects of SSZ are attributed to its function as a potent inhibitor of NF-κB. SSZ directly inhibits the activity of Inhibitor of κB Kinase (IKK), effectively preventing downstream κB-dependent transcriptional events [39], [40]. Recent clinical studies have confirmed that the beneficial effects of SSZ in patients with ulcerative colitis are in fact related to inhibition of NF-κB activation in the mucosa, which results in reduced cytokine production, and less severe inflammation [48]. In addition to IKK inhibition, other mechanisms of SSZ action have been described. SSZ inhibits 5-aminoimidazole-4-carboxamidoribonucleotide transformylase causing the release of adenosine [49], [50], which controls inflammation at least partly through inhibition of NF-κB signaling [51]. Other investigators found that SSZ may alleviate inflammation in a mouse model of inflammatory bowel disease by interacting with PPAR (peroxisome proliferator-activated receptor) nuclear receptors [52]. It is noteworthy that a common mechanism of all of these interactions is related to NF-κB inhibition, and it seems likely that SSZ-mediated blockade of NF-κB is central to the beneficial effects observed in our model. In fact, a highly specific IKK inhibitor, BMS-345541, mimicked the beneficial effects of SSZ on PcP-related lung injury and pulmonary dysfunction, suggesting that NF-κB plays an important role in the immune cascade leading to the development of PcP. However, BMS-345541 did not enhance pathogen clearance. Therefore, SSZ may have other, IKK-independent, immunomodulatory properties that account for the beneficial effects on AM phagocytosis and pathogen clearance.
The beneficial action of SSZ may result from its effects on a single cell type, or more likely, from its combined effects on several cell types that contribute to injury and disease. Potential lymphocyte targets of SSZ include CD4+, CD8+, and B lymphocytes. SSZ has pro-apoptotic effects on activated T cells [37], which may contribute to the reduced T cell numbers and inflammation found in PcP-related IRIS. SSZ also influences macrophage function through the induction of apoptosis, as well as alteration of macrophage inflammatory responses [53], [54]. SSZ blocked TNF production and also abrogated IL-12 expression and NO production by stimulated macrophages [55]. Modification of macrophage IL-12 may represent a mechanism by which SSZ alters the nature of the T cell response during IRIS. NF-κB is also involved in pulmonary epithelial cell inflammatory responses to Pc [38], [41], [56], providing another potential target for the action of SSZ. While the immunopathology associated with PcP and IRIS requires T cells, other cell types likely contribute to the overall disease process, and therefore the effectiveness of SSZ reported here likely results from multiple points of action.
Our studies have found that SSZ produces a TH2 shift in the lung cytokine environment during PcP-related IRIS, and that this shift is reflected in the phenotype of alveolar macrophages. TH2 cytokines lead to alternative activation of macrophages, and consistent with a TH2 cytokine shift we found that AMs isolated from SSZ-treated mice express high levels of the AAM marker ARG, but low levels of the CAM marker INOS (Figure 8). In contrast, AMs from PBS-treated IRIS mice display a CAM phenotype with high expression of INOS. It is notable that despite a well-documented role for INOS in host defense, these data suggest that enhanced Pc phagocytosis in SSZ-treated mice is associated with an alternatively activated AM phenotype with low expression of INOS. Based on our results, we believe that increased phagocytosis by alternatively activated macrophages is the mechanism of enhanced Pc clearance. However, a role for INOS in Pc killing cannot be excluded. Although we observe less staining in AMs from SSZ-treated mice, they are not totally devoid of INOS protein. More extensive studies will be required to determine the contribution of INOS in this model.
Although we have not demonstrated that the TH2 shift is solely responsible for the beneficial effects of SSZ during PcP, it is possible that TH2 cytokines acting through AAM effectors can increase fungal clearance while reducing immunopathogenesis. For example, TH2 cytokines enhance macrophage phagocytosis of Candida albicans by inducing macrophage expression of mannose receptor (MR) [57], [58] and dectin-1 [59]. These pattern recognition molecules are markers of the AAM phenotype, and have known roles in anti-fungal host defense. Similarly a TH2 shift in SSZ-treated mice could enhance phagocytosis of Pc by eliciting AAM with increased expression of MR and dectin-1. In addition, a TH2 shift may also attenuate the immunopathogenesis of PcP by reducing the production of pro-inflammatory TH1 cytokines, while enhancing production of anti-inflammatory TH2 cytokines. Elevated lung levels of the TH1 cytokines TNF-α and IFN-γ are associated with PcP-related lung injury and respiratory impairment [28]. In contrast, TH2-derived AAMs produce the potent anti-inflammatory cytokines IL-10 and TGF - β [44], which can dampen inflammatory responses and may contribute to the reduced inflammation and injury in SSZ-treated mice. Importantly, the anti-inflammatory potential of AAMs has been established in vivo by studies showing that the adoptive transfer of in vitro programmed AAMs attenuates immunopathogenesis in mouse models of inflammatory disease [60]. Although these findings are consistent with a SSZ-induced shift in the polarity of the T cell response, further studies are required to establish whether TH2 cytokines and alternative activation of AMs are directly responsible for the beneficial effects of SSZ during PcP.
Clinical studies have found that the severity of PcP correlates with the degree of pulmonary inflammation, but not with organism burden [9]–[14]. Controlled animal studies support these clinical observations, and have provided direct evidence that the immune response is a major pathogenic component of PcP [26], [28]–[31], [61]. Consequently, antibiotic treatment does not always produce rapid improvement of patients with severe PcP, because organisms and antigen may continue to drive the pathological immune response. The efficacy of SSZ in dramatically attenuating the severity of PcP supports the contention that adjunctive immunomodulatory therapy that target the T cell response is critical to optimal treatment of patients. Currently, adjunctive corticosteroids are commonly used for the clinical treatment of PcP. The broad anti-inflammatory and immunosuppressive properties of steroids are presumed to provide benefit, but concrete evidence that steroids improve survival is lacking. Our group has recently published a study demonstrating that specific disruption of the T cell response to Pc with anti-CD3 antibody has beneficial effects in a mouse model of PcP-related IRIS [62]. While both SSZ and anti-CD3 altered the T cell response to Pc and reduced immunopathogenesis, they produced differential outcomes with respect to fungal clearance. Anti-CD3 produced a profound inhibition of T cell responses which reduced disease, but also prevented the clearance of Pc from the lung. In contrast, SSZ dampened PcP-related immunopathogenesis without suppressing TH responses to a degree that prevented eradication of the organism. SSZ not only reduced T cell-mediated inflammation, but altered the nature of the T cell response by promoting TH2 lung cytokine environment and alternative activation of macrophages. It is likely that the preservation of TH2 responses combined with a shift in the polarization of AMs in SSZ-treated mice is responsible for the differential effects of SSZ and anti-CD3.
Another important aspect of our work is the development of a multispectral imaging flow cytometer-based method to assess the in vivo phagocytic activity of AM during a T cell-mediated immune response by quantifying the percentage of AMs that contain internalized Pc. Understanding the mechanisms controlling Pc phagocytosis is an area of great interest, and many investigators have utilized various techniques to perform in vitro assessments of Pc phagocytosis [63]–[70]. However, demonstrating an in vivo role for AM phagocytosis in the clearance of Pc has been more difficult. AM with associated Pc have been observed in the BAL fluid of patients and animals [71]–[74]. However, this was in the setting of active PcP, the level of phagocytosis appeared low, and the significance to organism clearance was not determined. Others have performed in vivo assessments of phagocytosis immediately (within 24 hours) following a bolus inoculation of labeled Pc [75], [76]. In addition, short-term depletion of AMs in rats reduced the clearance of Pc over the initial 24 hours post-inoculation [77]. While these studies were able to demonstrate a role for AM in vivo, the timing indicates that the investigators were evaluating the innate immune response to a bolus inoculation of Pc, rather than the CD4+ T cell-mediated response which is required for natural clearance of Pc from the lung. Using this new technology we were able to develop an assay to show that AMs are effector cells for the clearance of Pc during a natural CD4+ T cell-mediated immune response in vivo. The advantages of these ImageStream-based data are that: 1) internalized Pc was distinguished from attached Pc; 2) a large number of AM from each animal was rapidly assessed to provide quantification of the phagocytic response; 3) the dependence of phagocytic activity on the presence of CD4+ T cells was demonstrated; and 4) the CD4+ T cell-dependent increase in phagocytic activity correlated with the clearance kinetics of Pc. Importantly, the ImageStream data was validated using confocal microscopy to co-localize intracellular Pc with the lysosome protein LAMP-1. These data indicate that Pc is located within the phagolysosome of AM, consistent with phagocytosis of the pathogen. The multispectral imaging flow cytometry technology should provide a valuable tool for further study of Pc phagocytosis in vivo.
In summary, the results of this study indicate that the immune response to Pc can be modulated in a manner that reduces inflammatory consequences of PcP while enhancing the pathogen clearance through increased AM phagocytic capacity. We also developed a method for in vivo quantification of AM phagocytosis of Pc, and provide evidence that the macrophage is the ultimate effector for the CD4+ T cell-mediated clearance of Pc from the lungs. Immune modulation of T cell and AM functions should be considered potential therapeutic targets for the treatment of immune complications of PcP. Macrophages are equipped to recognize and eliminate pathogens as well as promote and/or resolve inflammation. Our results indicate that the phagocytic function of macrophages can be enhanced with a concomitant reduction in their pro-inflammatory properties. Enhancement of AM-mediated clearance of Pc may prove less inflammatory and generally superior to antibiotic therapy alone.
Methods
Pc source animals
Severe combined immunodeficient (SCID) mice on a C.B-17 background (C. B-Igh-1b/Icr Tac-Prkdcscid) were purchased from Taconic (Hudson, NY), or obtained from a breeding colony at the University of Rochester. The mice were housed using micro-isolator technology and fed sterilized food and water. To induce infection SCID mice were co-housed with Pc-infected SCID mice. Pc organisms were isolated as previously described [38]. Pc cysts were enumerated by standard Gomori's methenamine silver stain.
Sulfasalazine and trimethoprim-sulfamethoxazole administration
Sulfasalazine (SSZ) (Sigma, St. Louis, MO) was administered once daily by intra-peritoneal (i.p.) injection at a dose of 200 mg per kg of body weight. Trimethoprim Sulfamethoxazole (TMP-SMX) (SICOR Pharmaceuticals, Inc. Irvine, CA) was administered once daily i.p. at a dose calculated to give 16 mg per kg of body weight of the Trimethoprim component of the drug combination. This dose was based on the therapeutic dose given to humans for the treatment of PcP.
Mouse model of PcP-related IRIS
To induce infection SCID mice were intra-nasally inoculated with 1×105 purified Pc based on cyst count. Three weeks later the mice were immunologically reconstituted with 5×107 congenic spleen cells from normal C.B-17 mice.
In vivo CD4+ T cell depletion
CD4+ T cells were depleted by i.p. injection of monoclonal antibody specific for mouse CD4 (clone GK1.5, ATCC TIB207). Antibody injections (250 µg per mouse) were given one day prior to and one day after immune reconstitution. Thereafter, antibody was administrated every four days for the duration of the experiment.
Physiologic assessment of pulmonary function in live, ventilated mice
Lung compliance and resistance were measured in live ventilated mice using a whole body plethysmograph (BUXCO Electronics Inc., Wilmington, NC) connected to a Harvard rodent ventilator (Harvard Apparatus, Southnatic, MA) as previously described [78]. Dynamic lung compliance was normalized to the peak body weight of the animal. Respiratory rates were measured using whole body unrestrained chambers (BUXCO Electronics Inc). Data was collected and analyzed using the Biosystems XA software package (BUCXO Electronics Inc.).
Bronchoalveolar lavage (BAL) and lung tissue preparation
Right lung lobes were lavaged with four, one-ml aliquots of 1X Hank's balanced salt solution. Cell-free lavage fluid (approximately 3.5 ml per mouse) was frozen at −80°C. BAL cells were enumerated, and then differentials and multi-parameter flow cytometric analyses were performed. Anti-CD4-Fluorescein (clone RM4-4) and anti-CD8a-Peridinin Chlorophyll-α Protein (clone 53-6.7), were purchased from BD Biosciences (San Diego, CA). The anti-CD4 clone RM4-4 was used to confirm CD4+ cell depletion in vivo because it is not blocked by the CD4-depleting antibody (clone GK1.5). Cells were analyzed on a FACSCalibur (BD Biosciences, San Jose, CA), with at least 10,000 events routinely analyzed for each Pc-infected mouse. At least 5,000 events were analyzed from uninfected control mice.
For fixation the lungs were inflated with 15 cm gravity flow-pressure of 10% formalin (Sigma, St. Louis, MO). The lungs were fixed in situ for 10 minutes under gravity flow pressure, and then removed from the animal and placed in fixative for 16 h at 4°C. Lung tissue was embedded in paraffin and 4 µM sections were cut. Hematoxylin and eosin was used to visualize tissue.
Cytokine ELISAs of BAL fluid
Total protein concentration was determined in cell-free lavage by the colorimetric assay of Lowry. Albumin concentration was determined using the Mouse Albumin ELISA Quantitation kit from Bethyl Laboratories (Montgomery, TX). TNF-α, IFN-γ, MCP-1, and RANTES ELISA kits were used according to the manufacturer's instructions (R&D, Minneapolis, MN).
Real-time PCR assessment of lung Pc burden
Since Pc cannot be cultured, a real-time PCR method was used to quantify lung burden. For quantification of Pc burden in right lung lobes, quantitative PCR using TaqMan primer/fluorogenic probe chemistry and an Applied Biosystems Prism 7000 Sequence Detection System (Applied Biosystems, Foster City, CA) was performed with a primer/probe set specific for the mouse Pc kexin gene as previously described [78].
In vivo assessment of macrophage phagocytosis of Pc using ImageStream
For quantitation of Pc phagocytosis, an ImageStream multispectral imaging flow cytometer (Amnis Corporation, Seattle, WA) was used [79], [80]. With this technology the number of BAL AM with internalized Pc was directly quantified. CD11c was used as a surface marker to identify AM, while anti-Pc antibodies were used to stain internalized Pc. Whole lungs were lavaged and BAL cells were washed with ice cold PBS with 1%BSA (PBA), and incubated with mouse Fc Block (BD Biosciences, San Diego, CA) for 5 min on ice. Cells were then surface stained with anti-CD11c-phycoerythrin (clone HL3, BD Biosciences) for 30 minutes on ice and washed with PBA. The cells were then permeabilized with BD Cytofix/Cytoperm Fixation and Permeabilization Solution (BD Biosciences), and incubated with a pool of five different anti-Pneumocystis monoclonal antibodies for 30 minutes on ice. These antibodies were generated in our laboratory and were chosen because they recognize five different epitopes on the surface of Pc as determined by western blot and IFA (4F11, 2B5, 3D6, 1F1, 1F5). Characterization of antibody 4F11 has been published [81], but the remaining antibodies have not been further characterized. Following a wash step, the cells were incubated with Alexa Fluor 647 goat anti-mouse IgG (H+L) (Invitrogen Molecular Probes, OR) for 30 minutes on ice. Stained cells were washed, pelleted, and resuspended in 50 µl of ice cold 1% paraformaldehyde in PBS (Electron Microscopy Sciences, PA). Samples were stored at 4°C in the dark until analyzed. Twenty thousand to forty thousand event image files were collected for each sample on an ImageStream100 using 200 mW of 488 nm and 90 mW of 658 nm laser power.
The data obtained were analyzed using the ImageStream Data Exploration and Analysis Software (IDEAS, Amnis), which quantifies morphometric and photometric parameters on a per-cell basis for large populations of collected events [79], [82]. Single AM cells were gated as those events with normal brightfield area, high brightfield aspect ratio and CD11c positive staining. Each analyzed file contained at least 5000 AM events, enabling routine statistical analysis. AM that had phagocytosed Pc were identified by gating on CD11c+ events with high Pc-AF647 Max Pixel values (discriminates punctuate Pc from diffuse background staining and autofluorescence) and high Pc Internalization values. The Internalization score is a ratio of the intensity of bright red staining inside the cell (defined by eroding the CD11c mask 6 pixels) to the bright red staining in the membrane (defined by subtracting the latter mask from the CD11c mask dilated 3 pixels), and the higher the score the greater the concentration of Pc inside the cell. In order to condition the measurement to Pc particles in sharp focus (and thus in the central focal plane of the cell), only the mean of the upper quartile pixel intensities, weighted by the max pixel intensity, is used to compute the ratio [83]. The percentage of AM with internalized Pc was derived directly from the ImageStream data. The absolute number of AM with internalized Pc that was recovered from each animal was calculated by multiplying this percentage by the total number of AM recovered.
Confocal fluorescence microscopy
Cells were stained with the identical antibodies under the identical conditions described for ImageStream analyses with the following exceptions. Anti-mouse CD11c-AlexaFluor 488 (Invitrogen Molecular Probes, OR) was used instead of CD11c-PE. Also, in some experiments, biotinylated anti-mouse CD107a (LAMP-1) (Biolegend, CA) was used to co-localize intracellular Pc with lysosomes. Cells were first stained with anti-CD11c and anti-Pc antibodies as described above. After a wash and second permeabilization step, cells were stained with biotinylated anti-CD107a antibody for 30 minutes on ice followed by streptavidin conjugated with PE-Texas Red (BD Bioscience, CA). After fixation, cells were centrifuged onto glass slides, mounted with anti-fade Vectashield (Vector Laboratories, CA), and coverslipped for optimal imaging. Cells were imaged using an FV1000 Olympus Laser Scanning Confocal Microscope using an I×81 inverted microscope and a 60× objective with zoom of 4. Lasers used were 405, 488, 559, and 635 optimized to reduce photo-bleaching and used sequentially. Differential Interference Contrast (DIC) was performed using the 559 laser. Pixel dwell times were 8 us/pixel and 1024×1024 pixel format for high resolution imaging. Parameters were maintained consistent throughout imaging. All the images presented are the originals without post-processing.
Analyses of alveolar macrophage phenotype
BAL cells were collected and centrifuged onto glass slides. Cells were fixed with 3% paraformaldehyde and initially stained with hamster anti-mouse CD11c (Abcam, MA) followed by either goat anti-hamster AF594 or goat anti-hamster AF488 (Invitrogen Molecular Probe, Oregon) secondary antibody. After permeabilization with 0.2% Triton ×−100 in phosphate buffered saline, cells were stained with rabbit anti-mouse iNOS (Abcam, MA) with goat anti-rabbit AF488 (Invitrogen Molecular Probe, Oregon) secondary antibody, or goat anti-mouse Arginase (Santa Cruz Biotechnology, CA) with donkey anti-goat AF546 (Invitrogen Molecular Probe, Oregon) secondary antibody. Slides were mounted with anti-fade Vectashield (Vector Laboratories, CA) and coverslipped for optimal imaging. A Nikon Eclipse E400 fluorescence microscope was used for photomicroscopy. All photographs for a given protein were taken with identical exposure settings. The ImageJ software (National Institutes of Health) was used to quantify the mean fluorescent intensity of iNOS and Arginase staining in CD11c positive alveolar macrophages.
Statistical analyses
One-way analysis of variance was performed with the SigmaStat 2.0 software (Jandel, San Rafael, Calif.). The Student-Newman-Keuls method was used for all pair-wise multiple comparisons.
Ethics statement
All animal protocols were pre-approved by University Committee on Animal Resources (UCAR) at the University of Rochester Medical Center according to the guidelines of the Association for Assessment and Accreditation of Laboratory Animal Care International.
Zdroje
1. SantosJ
PalaciosR
RuizJ
GonzalezM
MarquezM
2005 Study of patients diagnosed with advanced HIV in the HAART era—OMEGA Cohort. Int J STD AIDS 16 252 255
2. ForrestDM
SeminariE
HoggRS
YipB
RaboudJ
1998 The incidence and spectrum of AIDS-defining illnesses in persons treated with antiretroviral drugs. Clin Infect Dis 27 1379 1385
3. StaikowskyF
LafonB
GuidetB
DenisM
MayaudC
1993 Mechanical ventilation for Pneumocystis carinii pneumonia in patients with the acquired immunodeficiency syndrome. Is the prognosis really improved? Chest 104 756 762
4. ArendSM
KroonFP
van't WoutJW
1995 Pneumocystis carinii pneumonia in patients without AIDS, 1980 through 1993. An analysis of 78 cases. Arch Intern Med 155 2436 2441
5. KaurN
MahlTC
2004 Pneumocystis carinii pneumonia with oral candidiasis after infliximab therapy for Crohn's disease. Dig Dis Sci 49 1458 1460
6. SepkowitzKA
BrownAE
ArmstrongD
1995 Pneumocystis carinii pneumonia without acquired immunodeficiency syndrome. More patients, same risk. Arch Intern Med 155 1125 1128
7. MorrisA
SciurbaFC
LebedevaIP
GithaigaA
ElliottWM
2004 Association of Chronic Obstructive Pulmonary Disease Severity and Pneumocystis Colonization. Am J Respir Crit Care Med 170 408 413
8. BenfieldTL
van SteenwijkR
NielsenTL
DichterJR
LipschikGY
1995 Interleukin-8 and eicosanoid production in the lung during moderate to severe Pneumocystis carinii pneumonia in AIDS: a role of interleukin-8 in the pathogenesis of P. carinii pneumonia. Respir Med 89 285 290
9. JensenBN
LisseIM
GerstoftJ
BorgeskovS
SkinhojP
1991 Cellular profiles in bronchoalveolar lavage fluid of HIV-infected patients with pulmonary symptoms: relation to diagnosis and prognosis. AIDS 5 527 533
10. LimperAH
OffordKP
SmithTF
MartinWJ
1989 Pneumocystis carinii pneumonia. Differences in lung parasite number and inflammation in patients with and without AIDS. Am Rev Respir Dis 140 1204 1209
11. LipschikGY
DoerflerME
KovacsJA
TravisWD
AndrawisVA
1993 Leukotriene B4 and interleukin-8 in human immunodeficiency virus-related pulmonary disease. Chest 104 763 769
12. MasonGR
HashimotoCH
DickmanPS
FouttyLF
CobbCJ
1989 Prognostic implications of bronchoalveolar lavage neutrophilia in patients with Pneumocystis carinii pneumonia and AIDS. Am Rev Respir Dis 139 1336 1342
13. SmithRL
El-SadrWM
LewisML
1988 Correlation of bronchoalveolar lavage cell populations with clinical severity of Pneumocystis carinii pneumonia. Chest 93 60 64
14. BenfieldTL
VestboJ
JungeJ
NielsenTL
JensenAB
1995 Prognostic value of interleukin-8 in AIDS-associated Pneumocystis carinii pneumonia. Am J Respir Crit Care Med 151 1058 1062
15. VahidB
BibboM
MarikPE
2007 Role of CD8 lymphocytes and neutrophilic alveolitis in Pneumocystis jiroveci pneumonia. Scandinavian Journal of Infectious Diseases 39 612 614
16. BarrySM
LipmanMC
DeeryAR
JohnsonMA
JanossyG
2002 Immune reconstitution pneumonitis following Pneumocystis carinii pneumonia in HIV-infected subjects. HIV Med 3 207 211
17. DoreGJ
LiY
McDonaldA
ReeH
KaldorJM
2002 Impact of highly active antiretroviral therapy on individual AIDS-defining illness incidence and survival in Australia. J Acquir Immune Defic Syndr 29 388 395
18. WuAK
ChengVC
TangBS
HungIF
LeeRA
2004 The unmasking of Pneumocystis jiroveci pneumonia during reversal of immunosuppression: case reports and literature review. BMC Infect Dis 4 57
19. SharmaS
NadrousHF
PetersSG
TefferiA
LitzowMR
2005 Pulmonary complications in adult blood and marrow transplant recipients: autopsy findings. Chest 128 1385 1392
20. SlivkaA
WenPY
SheaWM
LoefflerJS
1993 Pneumocystis carinii pneumonia during steroid taper in patients with primary brain tumors. Am J Med 94 216 219
21. MansharamaniNG
BalachandranD
VernovskyI
GarlandR
KozielH
2000 Peripheral blood CD4 + T-lymphocyte counts during Pneumocystis carinii pneumonia in immunocompromised patients without HIV infection. Chest 118 712 720
22. BachelezH
SchremmerB
CadranelJ
MoulyF
SarfatiC
1997 Fulminant Pneumocystis carinii pneumonia in 4 patients with dermatomyositis. Arch Intern Med 157 1501 1503
23. SepkowitzKA
1993 Pneumocystis carinii pneumonia in patients without AIDS. Clin Infect Dis 17 Suppl 2 S416 S422
24. KovacsJA
HiemenzJW
MacherAM
StoverD
MurrayHW
1984 Pneumocystis carinii pneumonia: a comparison between patients with the acquired immunodeficiency syndrome and patients with other immunodeficiencies. Ann Intern Med 100 663 671
25. HoriS
CarvalhoTL
DemengeotJ
2002 CD25+CD4+ regulatory T cells suppress CD4+ T cell-mediated pulmonary hyperinflammation driven by Pneumocystis carinii in immunodeficient mice. Eur J Immunol 32 1282 1291
26. RothsJB
SidmanCL
1992 Both immunity and hyperresponsiveness to Pneumocystis carinii result from transfer of CD4+ but not CD8+ T cells into severe combined immunodeficiency mice. J Clin Invest 90 673 678
27. SwainSD
MeissnerNN
HarmsenAG
2006 CD8 T cells modulate CD4 T-cell and eosinophil-mediated pulmonary pathology in Pneumocystis pneumonia in B-cell-deficient mice. Am J Pathol 168 466 475
28. WrightTW
GigliottiF
FinkelsteinJN
McBrideJT
AnCL
1999 Immune-mediated inflammation directly impairs pulmonary function, contributing to the pathogenesis of Pneumocystis carinii pneumonia. J Clin Invest 104 1307 1317
29. WrightTW
JohnstonCJ
HarmsenAG
FinkelsteinJN
1999 Chemokine gene expression during Pneumocystis carinii-driven pulmonary inflammation. Infect Immun 67 3452 3460
30. WrightTW
NotterRH
WangZ
HarmsenAG
GigliottiF
2001 Pulmonary inflammation disrupts surfactant function during Pneumocystis carinii pneumonia. Infect Immun 69 758 764
31. BhagwatSP
GigliottiF
XuH
WrightTW
2006 Contribution of T cell subsets to the pathophysiology of Pneumocystis-related immunorestitution disease. Am J Physiol Lung Cell Mol Physiol 291 L1256 L1266
32. RothsJB
SidmanCL
1993 Single and combined humoral and cell-mediated immunotherapy of Pneumocystis carinii pneumonia in immunodeficient scid mice. Infect Immun 61 1641 1649
33. SwainSD
HanS
HarmsenA
ShampenyK
HarmsenAG
2007 Pulmonary Hypertension Can Be a Sequela of Prior Pneumocystis Pneumonia. Am J Pathol 171 790 799
34. O'DellJR
PetersenK
LeffR
PalmerW
SchnedE
2006 Etanercept in combination with sulfasalazine, hydroxychloroquine, or gold in the treatment of rheumatoid arthritis. J Rheumatol 33 213 218
35. PloskerGL
CroomKF
2005 Sulfasalazine: a review of its use in the management of rheumatoid arthritis. Drugs 65 1825 1849
36. BrookesMJ
GreenJR
2004 Maintenance of remission in Crohn's disease: current and emerging therapeutic options. Drugs 64 1069 1089
37. LiptayS
BachemM
HackerG
AdlerG
DebatinKM
1999 Inhibition of nuclear factor kappa B and induction of apoptosis in T-lymphocytes by sulfasalazine. Br J Pharmacol 128 1361 1369
38. WangJ
GigliottiF
MaggirwarS
JohnstonC
FinkelsteinJN
2005 Pneumocystis carinii activates the NF-kappaB signaling pathway in alveolar epithelial cells. Infect Immun 73 2766 2777
39. WeberCK
LiptayS
WirthT
AdlerG
SchmidRM
2000 Suppression of NF-kappaB activity by sulfasalazine is mediated by direct inhibition of IkappaB kinases alpha and beta. Gastroenterology 119 1209 1218
40. WahlC
LiptayS
AdlerG
SchmidRM
1998 Sulfasalazine: a potent and specific inhibitor of nuclear factor kappa B. J Clin Invest 101 1163 1174
41. EvansSE
HahnPY
McCannF
KottomTJ
PavlovicZV
2005 Pneumocystis cell wall beta-glucans stimulate alveolar epithelial cell chemokine generation through nuclear factor-kappaB-dependent mechanisms. Am J Respir Cell Mol Biol 32 490 497
42. LebronF
VassalloR
PuriV
LimperAH
2003 Pneumocystis carinii cell wall beta-glucans initiate macrophage inflammatory responses through NF-kappaB activation. J Biol Chem 278 25001 25008
43. ZhangJ
ZhuJ
ImrichA
CushionM
KinaneTB
2004 Pneumocystis activates human alveolar macrophage NF-kappaB signaling through mannose receptors. Infect Immun 72 3147 3160
44. MartinezFO
HelmingL
GordonS
2008 Macrophage activation and polarization. Annu Rev Immuno 27 451 483
45. GordonS
2007 The macrophage: past, present and future. Eur J Immunol 37 Suppl 1 S9 17
46. ChengVC
HungIF
WuAK
TangBS
ChuCM
2004 Lymphocyte surge as a marker for immunorestitution disease due to Pneumocystis jiroveci pneumonia in HIV-negative immunosuppressed hosts. Eur J Clin Microbiol Infect Dis 23 512 514
47. ChenCS
BoeckhM
SeidelK
ClarkJG
KansuE
2003 Incidence, risk factors, and mortality from pneumonia developing late after hematopoietic stem cell transplantation. Bone Marrow Transplant 32 515 522
48. GanHT
ChenYQ
OuyangQ
2005 Sulfasalazine inhibits activation of nuclear factor-kappaB in patients with ulcerative colitis. J Gastroenterol Hepatol 20 1016 1024
49. BaggottJE
MorganSL
HaT
VaughnWH
HineKJ
1992 Inhibition of folate-dependent enzymes by non-steroidal anti-infalmmatory drugs. Biochem J 282 197 202
50. GadangiP
LongakerM
NaimeD
LevinRI
RechtPA
1996 The anti-inflammatory mechanism of sulfasalazine is related to adenosine release at inflamed sites. J Immunol 156 1937 1941
51. LukashevD
OhtaA
ApasovS
ChenJF
SitkovskyM
2004 Cutting Edge: Physiologic Attenuation of Proinflammatory Transcription by the Gs Protein-Coupled A2A Adenosine Receptor In Vivo. J Immunol 173 21 24
52. RousseauxC
LefebvreB
DubuquoyL
LefebvreP
RomanoO
2005 Intestinal antiinflammatory effect of 5-aminosalicylic acid is dependent on peroxisome proliferator-activated receptor-{gamma}. J Exp Med 201 1205 1215
53. SalhB
AssiK
HuangS
O'BrienL
SteinbrecherU
2002 Dissociated ROS production and ceramide generation in sulfasalazine-induced cell death in Raw 264.7 cells. J Leukoc Biol 72 790 799
54. OshikawaK
SugiyamaY
2003 Regulation of toll-like receptor 2 and 4 gene expression in murine alveolar macrophages. Exp Lung Res 29 401 412
55. HaskoG
SzaboC
NemethZH
DeitchEA
2001 Sulphasalazine inhibits macrophage activation: inhibitory effects on inducible nitric oxide synthase expression, interleukin-12 production and major histocompatibility complex II expression. Immunology 103 473 478
56. WangJ
GigliottiF
BhagwatSP
MaggirwarSB
WrightTW
2007 Pneumocystis stimulates MCP-1 production by alveolar epithelial cells through a JNK-dependent mechanism. Am J Physiol Lung Cell Mol Physiol 292 L1495 L1505
57. CosteA
DubourdeauM
LinasMD
CassaingS
LepertJC
2003 PPARgamma promotes mannose receptor gene expression in murine macrophages and contributes to the induction of this receptor by IL-13. Immunity 19 329 339
58. CosteA
LaganeC
FilipeC
AuthierH
GalesA
2008 IL-13 attenuates gastrointestinal candidiasis in normal and immunodeficient RAG-2(-/-) mice via peroxisome proliferator-activated receptor-gamma activation. J Immunol 180 4939 4947
59. GalesA
ConducheA
BernadJ
LefevreL
OlagnierD
2010 PPARgamma controls dectin-1 expression required for host antifungal defense against Candida albicans. PLoS Pathog 6(1) e1000714
60. HunterMM
WangA
ParharKS
JohnstonMJ
VanRN
2010 In vitro-derived alternatively activated macrophages reduce colonic inflammation in mice. Gastroenterology 138 1395 1405
61. GautP
DaarES
1999 Editorial response: Prophylaxis for Pneumocystis carinii pneumonia—an evolving tale of two populations. Clin Infect Dis 29 784 786
62. BhagwatSP
WrightTW
GigliottiF
2010 Anti-CD3 antibody decreases inflammation and improves outcome in a murine model of Pneumocystis pneumonia. J Immunol 184 497 502
63. EzekowitzRA
WilliamsDJ
KozielH
ArmstrongMY
WarnerA
1991 Uptake of Pneumocystis carinii mediated by the macrophage mannose receptor. Nature 351 155 158
64. ForteM
RaheluM
StubberfieldC
TomkinsL
PithieA
1991 In-vitro interaction of human macrophages with Pneumocystis carinii. Int J Exp Pathol 72 589 598
65. KozielH
EichbaumQ
KruskalBA
PinkstonP
RogersRA
1998 Reduced binding and phagocytosis of Pneumocystis carinii by alveolar macrophages from persons infected with HIV-1 correlates with mannose receptor downregulation. J Clin Invest 102 1332 1344
66. LasburyME
LinP
TschangD
DurantPJ
LeeCH
2004 Effect of bronchoalveolar lavage fluid from Pneumocystis carinii-infected hosts on phagocytic activity of alveolar macrophages. Infect Immun 72 2140 2147
67. LimperAH
StandingJE
HoyteJS
1996 The role of alveolar macrophages in Pneumocystis carinii elimination from the lower respiratory tract. J Eukaryot Microbiol 43 12S
68. MasurH
JonesTC
1978 The interaction in vitro of Pneumocystis carinii with macrophages and L-cells. J Exp Med 147 157 170
69. SteeleC
MarreroL
SwainS
HarmsenAG
ZhengM
2003 Alveolar macrophage-mediated killing of Pneumocystis carinii f. sp. muris involves molecular recognition by the Dectin-1 beta-glucan receptor. J Exp Med 198 1677 1688
70. ZhangJ
ZhuJ
BuX
CushionM
KinaneTB
2005 Cdc42 and RhoB activation are required for mannose receptor-mediated phagocytosis by human alveolar macrophages. Mol Biol Cell 16 824 834
71. BaskervilleA
DowsettAB
CookRW
DennisMJ
CranageMP
1991 Pneumocystis carinii pneumonia in simian immunodeficiency virus infection: immunohistological and scanning and transmission electron microscopical studies. J Pathol 164 175 184
72. ChenW
MillsJW
HarmsenAG
1992 Development and resolution of Pneumocystis carinii pneumonia in severe combined immunodeficient mice: a morphological study of host inflammatory responses. Int J Exp Pathol 73 709 720
73. WehleK
BlankeM
KoenigG
PfitzerP
1991 The cytological diagnosis of Pneumocystis carinii by fluorescence microscopy of Papanicolaou stained bronchoalveolar lavage specimens. Cytopathology 2 113 120
74. WehleK
SchirmerM
Dunnebacke-HinzJ
KupperT
PfitzerP
1993 Quantitative differences in phagocytosis and degradation of Pneumocystis carinii by alveolar macrophages in AIDS and non-HIV patients in vivo. Cytopathology 4 231 236
75. EmpeyKM
HollifieldM
SchuerK
GigliottiF
GarvyBA
2004 Passive immunization of neonatal mice against Pneumocystis carinii f. sp. muris enhances control of infection without stimulating inflammation. Infect Immun 72 6211 6220
76. WeaverT
HallCL
KachelDL
WardRP
WilliamsMD
1996 Assessment of in vivo attachment/phagocytosis by alveolar macrophages. J Immunol Methods 193 149 156
77. LimperAH
HoyteJS
StandingJE
1997 The role of alveolar macrophages in Pneumocystis carinii degradation and clearance from the lung. J Clin Invest 99 2110 2117
78. WrightTW
PryhuberGS
ChessPR
WangZ
NotterRH
2004 TNF receptor signaling contributes to chemokine secretion, inflammation, and respiratory deficits during Pneumocystis pneumonia. J Immunol 172 2511 2521
79. GeorgeTC
BasijiDA
HallBE
LynchDH
OrtynWE
2004 Distinguishing modes of cell death using the ImageStream multispectral imaging flow cytometer. Cytometry A 59 237 245
80. OrtynWE
HallBE
GeorgeTC
FrostK
BasijiDA
2006 Sensitivity measurement and compensation in spectral imaging. Cytometry A 69 852 862
81. WellsJ
GigliottiF
Simpson-HaidarisPJ
HaidarisCG
2004 Epitope mapping of a protective monoclonal antibody against Pneumocystis carinii with shared reactivity to Streptococcus pneumoniae surface antigen PspA. Infect Immun 72 1548 1556
82. GeorgeTC
FanningSL
Fitzgeral-BocarslyP
MedeirosRB
HighfillS
2006 Quantitative measurement of nuclear translocation events using similarity analysis of multispectral cellular images obtained in flow. Journal of Immunological Methods 311 117 129
83. MegjugoracNJ
JacobsES
IzaguirreAG
GeorgeTC
GuptaG
2007 Image-Based Study of Interferongenic Interactions between Plasmacytoid Dendritic Cells and HSV-Infected Monocyte-Derived Dendritic Cells*. Immunological Investigations 36 739 761
Štítky
Hygiena a epidemiologie Infekční lékařství Laboratoř
Článek vyšel v časopisePLOS Pathogens
Nejčtenější tento týden
2010 Číslo 8- Jak souvisí postcovidový syndrom s poškozením mozku?
- Měli bychom postcovidový syndrom léčit antidepresivy?
- Farmakovigilanční studie perorálních antivirotik indikovaných v léčbě COVID-19
- 10 bodů k očkování proti COVID-19: stanovisko České společnosti alergologie a klinické imunologie ČLS JEP
-
Všechny články tohoto čísla
- Dissecting the Genetic Architecture of Host–Pathogen Specificity
- The Battle for Iron between Bacterial Pathogens and Their Vertebrate Hosts
- Global Genotype-Phenotype Correlations in
- Burkholderia Type VI Secretion Systems Have Distinct Roles in Eukaryotic and Bacterial Cell Interactions
- Chitin Synthases from Are Involved in Tip Growth and Represent a Potential Target for Anti-Oomycete Drugs
- Distinct Merkel Cell Polyomavirus Molecular Features in Tumour and Non Tumour Specimens from Patients with Merkel Cell Carcinoma
- Biological and Structural Characterization of a Host-Adapting Amino Acid in Influenza Virus
- Functional Characterisation and Drug Target Validation of a Mitotic Kinesin-13 in
- CTCF Prevents the Epigenetic Drift of EBV Latency Promoter Qp
- The Human Fungal Pathogen Escapes Macrophages by a Phagosome Emptying Mechanism That Is Inhibited by Arp2/3 Complex-Mediated Actin Polymerisation
- Bim Nuclear Translocation and Inactivation by Viral Interferon Regulatory Factor
- Cyst Wall Protein 1 Is a Lectin That Binds to Curled Fibrils of the GalNAc Homopolymer
- Reciprocal Analysis of Infections of a Model Reveal Host-Pathogen Conflicts Mediated by Reactive Oxygen and imd-Regulated Innate Immune Response
- A Subset of Replication Proteins Enhances Origin Recognition and Lytic Replication by the Epstein-Barr Virus ZEBRA Protein
- Damaged Intestinal Epithelial Integrity Linked to Microbial Translocation in Pathogenic Simian Immunodeficiency Virus Infections
- Kaposin-B Enhances the PROX1 mRNA Stability during Lymphatic Reprogramming of Vascular Endothelial Cells by Kaposi's Sarcoma Herpes Virus
- Direct Interaction between Two Viral Proteins, the Nonstructural Protein 2C and the Capsid Protein VP3, Is Required for Enterovirus Morphogenesis
- A Novel CCR5 Mutation Common in Sooty Mangabeys Reveals SIVsmm Infection of CCR5-Null Natural Hosts and Efficient Alternative Coreceptor Use
- Micro RNAs of Epstein-Barr Virus Promote Cell Cycle Progression and Prevent Apoptosis of Primary Human B Cells
- Enterohemorrhagic Requires N-WASP for Efficient Type III Translocation but Not for EspF-Mediated Actin Pedestal Formation
- Host Imprints on Bacterial Genomes—Rapid, Divergent Evolution in Individual Patients
- UNC93B1 Mediates Host Resistance to Infection with
- The Transcription Factor Rbf1 Is the Master Regulator for -Mating Type Controlled Pathogenic Development in
- Protective Efficacy of Cross-Reactive CD8 T Cells Recognising Mutant Viral Epitopes Depends on Peptide-MHC-I Structural Interactions and T Cell Activation Threshold
- Bacteriophage Lysin Mediates the Binding of to Human Platelets through Interaction with Fibrinogen
- Insecticide Control of Vector-Borne Diseases: When Is Insecticide Resistance a Problem?
- Immune Modulation with Sulfasalazine Attenuates Immunopathogenesis but Enhances Macrophage-Mediated Fungal Clearance during Pneumonia
- PKC Signaling Regulates Drug Resistance of the Fungal Pathogen via Circuitry Comprised of Mkc1, Calcineurin, and Hsp90
- A Multi-Step Process of Viral Adaptation to a Mutagenic Nucleoside Analogue by Modulation of Transition Types Leads to Extinction-Escape
- “Everything You Always Wanted to Know about Sex (but Were Afraid to Ask)” in after Two Decades of Laboratory and Field Analyses
- Norovirus Gastroenteritis, Carbohydrate Receptors, and Animal Models
- Variations in TcdB Activity and the Hypervirulence of Emerging Strains of
- SWAN-1 Binds to EGL-9 and Regulates HIF-1-Mediated Resistance to the Bacterial Pathogen PAO1
- Conformational Adaptation of Asian Macaque TRIMCyp Directs Lineage Specific Antiviral Activity
- The Proteasome Active Site Threonine Is Essential for Persistence Yet Dispensable for Replication and Resistance to Nitric Oxide
- Characterization of Oseltamivir-Resistant 2009 H1N1 Pandemic Influenza A Viruses
- The Pneumococcal Serine-Rich Repeat Protein Is an Intra-Species Bacterial Adhesin That Promotes Bacterial Aggregation and in Biofilms
- Regulatory T Cell Suppressive Potency Dictates the Balance between Bacterial Proliferation and Clearance during Persistent Infection
- Structural Alterations in a Component of Cytochrome Oxidase and Molecular Evolution of Pathogenic in Humans
- A Limited Number of Antibody Specificities Mediate Broad and Potent Serum Neutralization in Selected HIV-1 Infected Individuals
- Spliced Leader Trapping Reveals Widespread Alternative Splicing Patterns in the Highly Dynamic Transcriptome of
- Contribution of Coagulases towards Disease and Protective Immunity
- Early Severe Inflammatory Responses to Uropathogenic Predispose to Chronic and Recurrent Urinary Tract Infection
- PLOS Pathogens
- Archiv čísel
- Aktuální číslo
- Informace o časopisu
Nejčtenější v tomto čísle- The Transcription Factor Rbf1 Is the Master Regulator for -Mating Type Controlled Pathogenic Development in
- PKC Signaling Regulates Drug Resistance of the Fungal Pathogen via Circuitry Comprised of Mkc1, Calcineurin, and Hsp90
- Contribution of Coagulases towards Disease and Protective Immunity
- Early Severe Inflammatory Responses to Uropathogenic Predispose to Chronic and Recurrent Urinary Tract Infection
Kurzy
Zvyšte si kvalifikaci online z pohodlí domova
Současné možnosti léčby obezity
nový kurzAutoři: MUDr. Martin Hrubý
Všechny kurzyPřihlášení#ADS_BOTTOM_SCRIPTS#Zapomenuté hesloZadejte e-mailovou adresu, se kterou jste vytvářel(a) účet, budou Vám na ni zaslány informace k nastavení nového hesla.
- Vzdělávání



