-
Články
Top novinky
Reklama- Vzdělávání
- Časopisy
Top články
Nové číslo
- Témata
Top novinky
Reklama- Videa
- Podcasty
Nové podcasty
Reklama- Volná místa
Doporučené pozice
Reklama- Praxe
Top novinky
ReklamaThe Deadly Chytrid Fungus: A Story of an Emerging Pathogen
article has not abstract
Published in the journal: . PLoS Pathog 6(1): e32767. doi:10.1371/journal.ppat.1000550
Category: Pearls
doi: https://doi.org/10.1371/journal.ppat.1000550Summary
article has not abstract
Emerging infectious diseases present a great challenge for the health of both humans and wildlife. The increasing prevalence of drug-resistant fungal pathogens in humans [1] and recent outbreaks of novel fungal pathogens in wildlife populations [2] underscore the need to better understand the origins and mechanisms of fungal pathogenicity. One of the most dramatic examples of fungal impacts on vertebrate populations is the effect of the amphibian disease chytridiomycosis, caused by the chytrid fungus Batrachochytrium dendrobatidis (Bd).
Amphibians around the world are experiencing unprecedented population losses and local extinctions [3]. While there are multiple causes of amphibian declines, many catastrophic die-offs are attributed to Bd [4],[5]. The chytrid pathogen has been documented in hundreds of amphibian species, and reports of Bd's impact on additional species and in additional geographic regions are accumulating at an alarming rate (e.g., see http://www.spatialepidemiology.net/bd). Bd is a microbial, aquatic fungus with distinct life stages. The motile stage, called a zoospore, swims using a flagellum and initiates the colonization of frog skin. Within the host epidermal cells, a zoospore forms a spherical thallus, which matures and produces new zoospores by dividing asexually, renewing the cycle of infection when zoospores are released to the skin surface (Figure 1). Bd is considered an emerging pathogen, discovered and described only a decade ago [6],[7]. Despite intensive ecological study of Bd over the last decade, a number of unanswered questions remain. Here we summarize what has been recently learned about this lethal pathogen.
Fig. 1. Life cycle of the pathogenic chytrid fungus Batrachochytrium dendrobatidis. 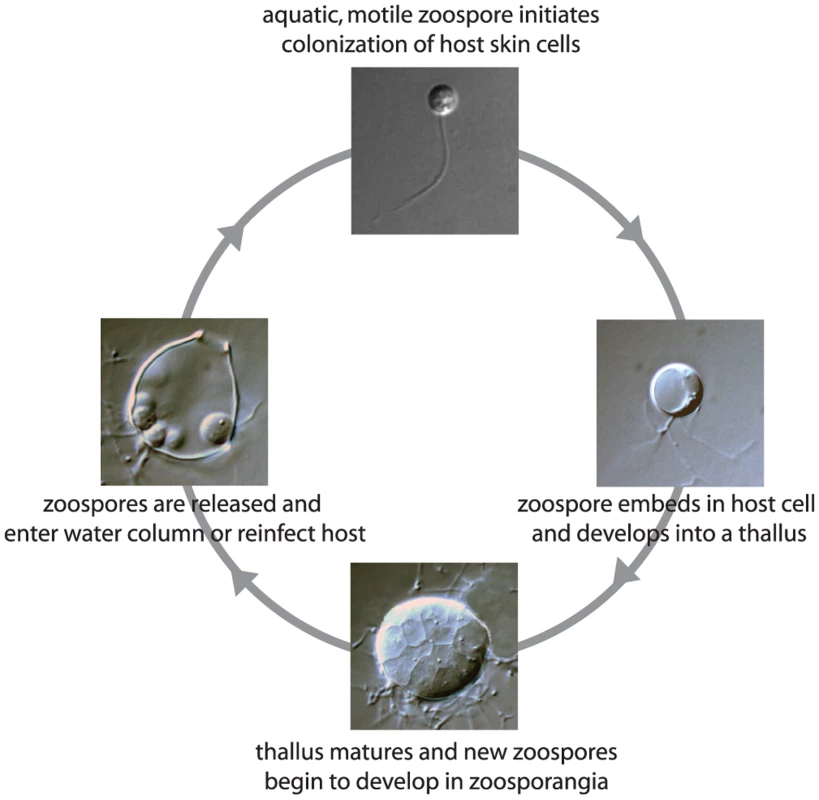
Images were taken of Bd in pure culture grown in 1% tryptone media. How Is Bd Related to Other Fungi?
Bd is a member of a basal group of fungi, the Chytridiomycota, and is the only known member of its order (the Rhizophydiales) to parasitize vertebrates. Bd is phylogenetically distant from any of the other ∼1,000 chytrid species [8], and the lack of close relatives capable of parasitizing vertebrates suggests that Bd pathogenicity evolved relatively recently. Further, population genetic data on Bd isolates collected from different amphibian populations around the world suggest that Bd is a recently spread pathogen rather than being endemic with altered relationships with hosts due to environmental change [9],[10].
How Has Bd Spread around the World So Quickly?
Africa was initially proposed as the geographic origin because the earliest evidence of Bd is from skin samples from African clawed frogs (Xenopus laevis) collected in 1938. African clawed frogs were traded globally for decades (from the 1930s–1960s) for pregnancy assays in humans [11]. Although based on a small sample size, recent population genetic work shows reduced genetic diversity of isolates from African clawed frogs and, instead, high allelic diversity in North American isolates collected from bullfrogs (Rana catesbeiana) [10],[12]. Although additional genetic work is needed, these studies suggest that Bd's origin may not be in Africa. Anthropogenic spread of Bd is a plausible explanation for at least some introductions [11],[13]. Some amphibian species that are traded globally may serve as disease reservoirs because they can carry Bd infections without morbidity. A number of mysteries remain about how Bd has dispersed to and persisted in remote pristine environments where anthropogenic introduction is unlikely. If Bd can survive independently of amphibian hosts, it must use non-amphibian organic materials as nutrient resources. Although Bd DNA has been detected in water bodies [14] and on rocks [15], conclusive evidence of Bd persistence in the environment is lacking.
How Does Bd Kill Frogs?
In infected amphibians, Bd is found in the cells of the epidermis and pathological abnormalities include a thickening of the outer layer of skin [6]. Cutaneous fungal infections in other vertebrates are not typically lethal, but amphibian skin is unique because it is physiologically active, tightly regulating the exchange of respiratory gases, water, and electrolytes. Thus, the physiological importance of the skin makes amphibians particularly vulnerable to skin infections. It has been hypothesized that Bd disrupts normal regulatory functioning of frog skin, and evidence suggests that electrolyte depletion and osmotic imbalance that occurs in amphibians with severe chytridiomycosis are sufficient to cause mortality [16],[17].
What Factors Are Implicated in Bd Pathogenicity/Virulence?
The molecular factors influencing Bd pathogenicity and virulence have yet to be conclusively identified. Some evidence suggests that Bd enzymatic activity directly influences pathogenesis. The initial penetration of Bd into amphibian epidermal cells likely requires digestive enzymes. In culture, Bd secretes extracellular proteases that degrade casein and gelatin [18],[19]. At the molecular level, genomic research into Bd is revealing intriguing expression patterns in genes such as those for serine protease and fungalysin metallopeptidase [20], two gene families involved in pathogenesis in other fungal pathogens. Full genomes of two Bd isolates have recently been sequenced, providing new resources for the study of molecular mechanisms of pathogenicity [21].
Are There Differences in Bd Isolate Virulence?
Several studies have shown variation in virulence among Bd isolates. In experimental infections, differences in frog survival have been observed when exposed to different Bd isolates (e.g., [22],[23]). Initial proteomic work suggests that Bd isolates differ in their proteome profiles [23]. However, controlled infection experiments with reciprocal host isolate treatments and paired genomic and proteomic studies are necessary to identify the functional determinants of Bd virulence.
Do All Frogs Respond Similarly to Bd?
Species, populations, and individuals vary widely in susceptibility to chytridiomycosis. Mortality rates in laboratory infection experiments can range from 0% to 100%, depending on the species (e.g., [22],[24]), age of animals [25], and temperature regime [26]. In the wild, some species and populations are extirpated while others, those that survive initial declines, persist with various levels of infection (e.g., [27],[28]). While the disease dynamics are undoubtedly influenced by local environmental conditions, particularly temperature, inherent differences in host susceptibility and behavior are also important. Colonization by Bd and subsequent disease development may be influenced by host defense mechanisms, such as secretions of antimicrobial peptides [29] or bacterial commensals with anti-fungal properties [30]. Some species-specific behavioral characteristics such as microhabitat selection, basking, aggregating in retreat sites, or association with water bodies may also affect the likelihood of infection and disease [31],[32].
How Can We Stem the Tide of Bd-Related Declines?
Despite many gaps in our understanding of chytridiomycosis, we are beginning to unravel important elements of this lethal disease and make progress towards amphibian conservation. Multiple conservation strategies have been proposed and are currently being implemented to mitigate the threat of chytridiomycosis. These plans include efforts to limit the spread of the disease, invest in captive breeding programs for highly vulnerable amphibians, and advance basic disease research. Continued research on the biology of both the host and the pathogen is necessary, and efforts to catalog and preserve the Bd isolates for ongoing research are particularly important (see http://www.spatialepidemiology.net/bd/ and http://www.bdbank.org/, [33]). The conservation challenges we face with chytridiomycosis—and other emerging pathogens—are best confronted by increasing our knowledge of disease processes from both host and pathogen perspectives.
Zdroje
1. WarnockDW
2006 Fungal diseases: an evolving public health challenge. Med Mycol 44 697 705
2. BlehertDS
HicksAC
BehrM
MeteyerCU
Berlowski-ZierBM
2009 Bat white-nose syndrome: an emerging fungal pathogen? Science 323 227 227
3. StuartSN
ChansonJS
CoxNA
YoungBE
RodriguesASL
2004 Status and trends of amphibian declines and extinctions worldwide. Science 306 1783 1786
4. LipsKR
BremF
BrenesR
ReeveJD
AlfordRA
2006 Emerging infectious disease and the loss of biodiversity in a Neotropical amphibian community. Proc Natl Acad Sci U S A 103 3165 3170
5. SchloegelL
HeroJM
BergerL
SpeareR
McDonaldKR
2006 The decline of the sharp-snouted day frog (Taudactylus acutirostris): the first documented case of extinction by infection in a free-ranging wildlife species? EcoHealth 3 35 40
6. BergerL
SpeareR
DaszakP
GreenDE
CunninghamAA
1998 Chytridiomycosis causes amphibian mortality associated with population declines in the rain forests of Australia and Central America. Proc Natl Acad Sci U S A 95 9031 9036
7. LongcoreJE
PessierAP
NicholsDK
1999 Batrachochytrium dendrobatidis gen et sp nov, a chytrid pathogenic to amphibians. Mycologia 91 219 227
8. JamesTY
LetcherPM
LongcoreJE
Mozley-StandridgeSE
PorterD
2006 A molecular phylogeny of the flagellated fungi (Chytridiomycota) and description of a new phylum (Blastocladiomycota). Mycologia 98 860 871
9. MorehouseEA
JamesTY
GanleyARD
VilgalysR
BergerL
2003 Multilocus sequence typing suggests the chytrid pathogen of amphibians is a recently emerged clone. Mol Ecol 12 395 403
10. JamesTY
LitvintsevaAP
VilgalysR
MorganJAT
TaylorJW
2009 Rapid global expansion of the fungal disease chytridiomycosis into declining and healthy amphibian populations. PLoS Pathog e1000458 doi:10.1371/journal.ppat.1000458
11. WeldonC
du PreezLH
HyattAD
MullerR
SpeareR
2004 Origin of the amphibian chytrid fungus. Emerg Infect Dis 10 2100 2105
12. GokaK
YokoyamaJ
UneY
KurokiT
SuzukiK
NakaharaM
KobayashiA
InabaS
MizutaniT
HyattAD
2009 Amphibian chytridiomycosis in Japan: distribution, haplotypes and possible route of entry into Japan. Molecular Ecology 18 4757 4774
13. FisherMC
GarnerTWJ
2007 The relationship between the emergence of Batrachochytrium dendrobatidis, the international trade in amphibians and introduced amphibian species. Fungal Biol Rev 21 2 9
14. KirshteinJD
AndersonCW
WoodJS
LongcoreJE
VoytekMA
2007 Quantitative PCR detection of Batrachochytrium dendrobatidis DNA from sediments and water. Dis Aquat Org 77 11 15
15. WalkerSF
SalasMB
JenkinsD
GarnerTWJ
CunninghamAA
2007 Environmental detection of Batrachochytrium dendrobatidis in a temperate climate. Dis Aquat Org 77 105 112
16. VoylesJ
BergerL
YoungS
SpeareR
WebbR
2007 Electrolyte depletion and osmotic imbalance in amphibians with chytridiomycosis. Dis Aquat Org 77 113 118
17. VoylesJ
YoungS
BergerL
CampbellC
VoylesWF
DinudomA
Cook
WebbR
AlfordRA
SkerrattLF
SpeareR
2009 Pathogenesis of chytridiomycosis, a cause of catastrophic amphibian declines. Science 326 582 585
18. PiotrowskiJS
AnnisSL
LongcoreJE
2004 Physiology of Batrachochytrium dendrobatidis, a chytrid pathogen of amphibians. Mycologia 96 9 15
19. SymondsEP
TrottDJ
BirdPS
MillsP
2008 Growth characteristics and enzyme activity in Batrachochytrium dendrobatidis isolates. Mycopathologia 166 143 147
20. RosenblumEB
StajichJE
MaddoxN
EisenMB
2008 Global gene expression profiles for life stages of the deadly amphibian pathogen Batrachochytrium dendrobatidis. Proc Natl Acad Sci U S A 105 17034 17039
21. RosenblumEB
FisherMC
JamesTY
StajichJE
LongcoreJE
GentryLR
PoortenTJ
2009 A molecular perspective: biology of the emerging pathogen Batrachochytrium dendrobatidis. Dis Aquat Org doi:10.3354/dao02179
22. BergerL
MarantelliG
SkerrattLL
SpeareR
2005 Virulence of the amphibian chytrid fungus Batrachochytrium dendrobatidis varies with the strain. Dis Aquat Org 68 47 50
23. FisherMC
BoschJ
YinZ
SteadDA
WalkerJ
2009 Proteomic and phenotypic profiling of the amphibian pathogen Batrachochytrium dendrobatidis shows that genotype is linked to virulence. Mol Ecol 18 415 429
24. DaszakP
StriebyA
CunninghamAA
LongcoreJE
BrownCC
2004 Experimental evidence that the bullfrog (Rana catesbeiana) is a potential carrier of chytridiomycosis, an emerging fungal disease of amphibians. Herpetolog J 14 201 207
25. LamirandeEW
NicholsDK
2002 Effects of host age on susceptibility to cutaneous chytridiomycosis in blue-and-yellow poison dart frogs (Dendrobates tinctorius). St. Paul, Minnesota. 3 13 In Proceedings of the Sixth International Symposium on the Pathology of Reptiles and Amphibians, 18–19 April 2001
26. WoodhamsDC
AlfordRA
MarantelliG
2003 Emerging disease of amphibians cured by elevated body temperature. Dis Aquat Org 55 65 67
27. RetallickRWR
McCallumH
SpeareR
2004 Endemic infection of the amphibian chytrid fungus in a frog community post-decline. PLoS Biol 2 e351 doi:10.1371/journal.pbio.0020351
28. BriggsCJ
VredenburgVT
KnappRA
RachowiczLJ
2005 Investigating the population-level effects of chytridiomycosis: An emerging infectious disease of amphibians. Ecology 86 3149 3159
29. WoodhamsDC
ArdipradjaK
AlfordRA
MarantelliG
ReinertLK
2007 Resistance to chytridiomycosis varies among amphibian species and is correlated with skin peptide defenses. Anim Conserv 10 409 417
30. HarrisRN
JamesTY
LauerA
SimonMA
PatelA
2006 Amphibian pathogen Batrachochytrium dendrobatidis is inhibited by the cutaneous bacteria of Amphibian species. Ecohealth 3 53 56
31. LipsKR
ReeveJD
WittersLR
2003 Ecological traits predicting amphibian population declines in Central America. Conserv Biol 17 1078 1088
32. RowleyJJL
AlfordRA
2007 Behaviour of Australian rainforest stream frogs may affect the transmission of chytridiomycosis. Dis Aquat Org 77 1 9
33. VoylesJ
CashinsSD
RosenblumEB
PushendorfR
2009 Preserving pathogens for wildlife conservation: a case for action on amphibian declines. Oryx 43 527 529
Štítky
Hygiena a epidemiologie Infekční lékařství Laboratoř
Článek Polyoma Virus-Induced Osteosarcomas in Inbred Strains of Mice: Host Determinants of MetastasisČlánek Nutrient Availability as a Mechanism for Selection of Antibiotic Tolerant within the CF AirwayČlánek Type I Interferon Induction Is Detrimental during Infection with the Whipple's Disease Bacterium,
Článek vyšel v časopisePLOS Pathogens
Nejčtenější tento týden
2010 Číslo 1- Jak souvisí postcovidový syndrom s poškozením mozku?
- Měli bychom postcovidový syndrom léčit antidepresivy?
- Farmakovigilanční studie perorálních antivirotik indikovaných v léčbě COVID-19
- 10 bodů k očkování proti COVID-19: stanovisko České společnosti alergologie a klinické imunologie ČLS JEP
-
Všechny články tohoto čísla
- CD8+ T Cell Control of HIV—A Known Unknown
- The Deadly Chytrid Fungus: A Story of an Emerging Pathogen
- Characterization of the Oral Fungal Microbiome (Mycobiome) in Healthy Individuals
- Polyoma Virus-Induced Osteosarcomas in Inbred Strains of Mice: Host Determinants of Metastasis
- Within-Host Evolution of in Four Cases of Acute Melioidosis
- The Type III Secretion Effector NleE Inhibits NF-κB Activation
- Protease-Sensitive Synthetic Prions
- Histone Deacetylases Play a Major Role in the Transcriptional Regulation of the Life Cycle
- Parasite-Derived Plasma Microparticles Contribute Significantly to Malaria Infection-Induced Inflammation through Potent Macrophage Stimulation
- β-Neurexin Is a Ligand for the MSCRAMM SdrC
- Structure of the HCMV UL16-MICB Complex Elucidates Select Binding of a Viral Immunoevasin to Diverse NKG2D Ligands
- Nutrient Availability as a Mechanism for Selection of Antibiotic Tolerant within the CF Airway
- Like Will to Like: Abundances of Closely Related Species Can Predict Susceptibility to Intestinal Colonization by Pathogenic and Commensal Bacteria
- Importance of the Collagen Adhesin Ace in Pathogenesis and Protection against Experimental Endocarditis
- N-glycan Core β-galactoside Confers Sensitivity towards Nematotoxic Fungal Galectin CGL2
- Two Plant Viral Suppressors of Silencing Require the Ethylene-Inducible Host Transcription Factor RAV2 to Block RNA Silencing
- A Small-Molecule Inhibitor of Motility Induces the Posttranslational Modification of Myosin Light Chain-1 and Inhibits Myosin Motor Activity
- Temporal Proteome and Lipidome Profiles Reveal Hepatitis C Virus-Associated Reprogramming of Hepatocellular Metabolism and Bioenergetics
- Marburg Virus Evades Interferon Responses by a Mechanism Distinct from Ebola Virus
- B Cell Activation by Outer Membrane Vesicles—A Novel Virulence Mechanism
- Killing a Killer: What Next for Smallpox?
- PPARγ Controls Dectin-1 Expression Required for Host Antifungal Defense against
- TRIM5α Modulates Immunodeficiency Virus Control in Rhesus Monkeys
- Immature Dengue Virus: A Veiled Pathogen?
- Panton-Valentine Leukocidin Is a Very Potent Cytotoxic Factor for Human Neutrophils
- In Vivo CD8+ T-Cell Suppression of SIV Viremia Is Not Mediated by CTL Clearance of Productively Infected Cells
- Placental Syncytiotrophoblast Constitutes a Major Barrier to Vertical Transmission of
- Type I Interferon Induction Is Detrimental during Infection with the Whipple's Disease Bacterium,
- The M/GP Glycoprotein Complex of Porcine Reproductive and Respiratory Syndrome Virus Binds the Sialoadhesin Receptor in a Sialic Acid-Dependent Manner
- Social Motility in African Trypanosomes
- Melanoma Differentiation-Associated Gene 5 (MDA5) Is Involved in the Innate Immune Response to Infection In Vivo
- Protection of Mice against Lethal Challenge with 2009 H1N1 Influenza A Virus by 1918-Like and Classical Swine H1N1 Based Vaccines
- Upregulation of xCT by KSHV-Encoded microRNAs Facilitates KSHV Dissemination and Persistence in an Environment of Oxidative Stress
- Persistent ER Stress Induces the Spliced Leader RNA Silencing Pathway (SLS), Leading to Programmed Cell Death in
- Evolutionary Trajectories of Beta-Lactamase CTX-M-1 Cluster Enzymes: Predicting Antibiotic Resistance
- Nucleoporin 153 Arrests the Nuclear Import of Hepatitis B Virus Capsids in the Nuclear Basket
- CD8+ Lymphocytes Control Viral Replication in SIVmac239-Infected Rhesus Macaques without Decreasing the Lifespan of Productively Infected Cells
- PLOS Pathogens
- Archiv čísel
- Aktuální číslo
- Informace o časopisu
Nejčtenější v tomto čísle- Panton-Valentine Leukocidin Is a Very Potent Cytotoxic Factor for Human Neutrophils
- CD8+ T Cell Control of HIV—A Known Unknown
- Polyoma Virus-Induced Osteosarcomas in Inbred Strains of Mice: Host Determinants of Metastasis
- The Deadly Chytrid Fungus: A Story of an Emerging Pathogen
Kurzy
Zvyšte si kvalifikaci online z pohodlí domova
Současné možnosti léčby obezity
nový kurzAutoři: MUDr. Martin Hrubý
Všechny kurzyPřihlášení#ADS_BOTTOM_SCRIPTS#Zapomenuté hesloZadejte e-mailovou adresu, se kterou jste vytvářel(a) účet, budou Vám na ni zaslány informace k nastavení nového hesla.
- Vzdělávání



