-
Články
Top novinky
Reklama- Vzdělávání
- Časopisy
Top články
Nové číslo
- Témata
Top novinky
Reklama- Videa
- Podcasty
Nové podcasty
Reklama- Kariéra
Doporučené pozice
Reklama- Praxe
Top novinky
ReklamaRegulation of Flowering by the Histone Mark Readers MRG1/2 via Interaction with CONSTANS to Modulate Expression
The photoperiodic flowering in Arabidopsis requires the key regulator CO and its target gene FT. However, how CO regulates FT expression in the context of chromatin remains largely obscure. In this work, we present Arabidopsis MRG1/2 as novel chromatin effectors directly involved in the CO-FT photoperiodic flowering. Firstly, MRG1/2 proteins are identified as recognition factors of H3K4 and H3K36 methylation via their chromodomains. The mrg1 mrg2 double mutant shows a late-flowering phenotype only under long-day conditions through down-regulation of FT but not of CO. MRG2 can directly target in vivo the FT promoter chromatin in a H3K4me3/H3K36me3-level dependent manner. More importantly, MRG2 and CO physically interact and enhance each other's binding to the FT promoter in planta. Determination of co-crystal structures of MRG2 with H3K4me3/H3K36me3 peptides and mutagenesis of a key amino acid residue involved in structural interaction demonstrate that MRG2 reader activity is essential for in planta function. Taken together, our findings uncover a novel mechanism of FT activation in flowering promotion and provide a striking example of mutual interplay between a transcription factor and a histone methylation reader in transcription regulation.
Published in the journal: . PLoS Genet 10(9): e32767. doi:10.1371/journal.pgen.1004617
Category: Research Article
doi: https://doi.org/10.1371/journal.pgen.1004617Summary
The photoperiodic flowering in Arabidopsis requires the key regulator CO and its target gene FT. However, how CO regulates FT expression in the context of chromatin remains largely obscure. In this work, we present Arabidopsis MRG1/2 as novel chromatin effectors directly involved in the CO-FT photoperiodic flowering. Firstly, MRG1/2 proteins are identified as recognition factors of H3K4 and H3K36 methylation via their chromodomains. The mrg1 mrg2 double mutant shows a late-flowering phenotype only under long-day conditions through down-regulation of FT but not of CO. MRG2 can directly target in vivo the FT promoter chromatin in a H3K4me3/H3K36me3-level dependent manner. More importantly, MRG2 and CO physically interact and enhance each other's binding to the FT promoter in planta. Determination of co-crystal structures of MRG2 with H3K4me3/H3K36me3 peptides and mutagenesis of a key amino acid residue involved in structural interaction demonstrate that MRG2 reader activity is essential for in planta function. Taken together, our findings uncover a novel mechanism of FT activation in flowering promotion and provide a striking example of mutual interplay between a transcription factor and a histone methylation reader in transcription regulation.
Introduction
The timing of floral transition from vegetative to reproductive development is a critical event in the plant life cycle and is coordinated by internal and environmental cues [1]–[3]. In Arabidopsis, the photoperiodic flowering pathway is regulated by the transcription factor CONSTANS (CO) and the florigen FLOWERING LOCUS T (FT) [4]. Circadian-clock regulated CO mRNA and light-dependent stabilization of CO protein are crucial for activation of FT expression in leaves under long days (LDs) but not short days (SDs); the FT protein is then translocated to the shoot apical meristem, where it promotes flowering [4]. The CO protein can bind to specific cis-elements in the FT promoter either by itself [5] or in a complex with CCAAT-binding factors [6].
Histone lysine methylation is an important epigenetic mechanism for the regulation of gene expression. Recent studies have revealed that chromatin mechanisms play important roles in flowering time by regulating the expression of key flowering-regulatory genes [7]. For example, FT expression is affected by several factors, including H3K27 methyltransferase CLF [8]–[10], H3K27 demethylase JMJ12/REF6 [11], H3K4 demethylases ELF6 and JMJ14/JMJ4 [12]–[14], and AFR1/2-HDAC histone deacetylase [15]. More recently, other histone methyltransferases (ATX1, ATX2, SDG8, SDG25) and demethylases (LDL1/2) were reported to affect histone methylation status at the FT chromatin [16].
It was proposed that chromatin with methylated histone residues can be specifically recognized by the chromatin effectors that act as readers, which might ultimately direct downstream functions [17], [18]. The Arabidopsis histone methylation reader proteins ORC1 [19], AtING [20], AL [20], [21], WDR5a [22], SDG8/ASHH2 [23], [24], LHP1/TFL2 [25], [26], PICKLE [27], and rice protein CHR729 [28] were found to interact with methylated H3K4 or/and H3K27, and also affect many aspects of plant development from mutant analyses. Although chromatin effectors related to several lysine residues were identified, the proteins recognizing H3K36 methylation remain unknown in plant. In addition, histone mark readers that are directly involved in CO-FT regulatory pathway have not been identified.
To identify H3K36 methylation readers, we performed an in vitro peptide pull-down experiment using Arabidopsis nuclear extracts, and identified among proteins detected with mass spectrometry the Arabidopsis Morf Related Gene (MRG) group protein MRG2 as a binding protein of the histone H3 N-terminal tail methylated at lysine 36. MRG1 and MRG2 belong to the MRG protein family, with highly conserved members in fungi, plants, and animals. Several family members, such as Esa1p-associated factor-3 (EAF3), MRG on chromosomes 15 (MRG15), and male-specific lethal (MSL3) have been found in yeast and animals as recognition factors of H3K36 methylation [29]–[31]. The yeast eaf3 deletion causes no obvious growth phenotypes and only has a very modest effect on transcription [32], [33], while a mutation in the Drosophila MSL3 gene led to male lethality [34], and overexpression of human MRG15 results in abnormal nuclear morphologies and cell death [35], making further studies of in vivo MRG functions difficult in animals.
We show here using viable Arabidopsis mutants that MRG1/2 promote photoperiodic flowering in Arabidopsis. MRG1/2 proteins act as novel chromatin effectors directly involved in the CO-dependent FT activation in the photoperiodic flowering pathway. The mrg1 mrg2 double mutant exhibits reduced FT mRNA level, with a normal CO mRNA level, and is late-flowering only under LDs. We further demonstrate that MRG2 and CO interact with each other physically and enhance each other's binding to the FT promoter region, thereby activating FT transcription. Furthermore, the co-crystal structures of MRG2 with H3K4me3/36me3 peptides reveal the residues important for peptide binding and a site-specific MRG2 mutation abolishes both histone mark binding activity and flowering time regulation, providing a direct link between the biochemical activity of MRG1/2 proteins and their in vivo biological functions.
Results
Identification of MRG1/2 as specific binding proteins for methylated H3K4 and H3K36
An in vitro peptide pull-down assay was used to identify the proteins that bind to histone H3 tail methylated at K36. Biotinylated histone H3 peptides (amino acids 21 to 44), with either unmethylated (control) or tri-methylated K36, were immobilized on the streptavidin-coated beads and incubated with Arabidopsis nuclear extracts. Mass spectrometry of proteins bound to the H3 peptides tri-methylated at K36 identified MRG2, which is a member of the MRG protein family. Members in this group have the same domain organization, with a chromodomain (chromatin organization modifier domain) near the N-terminus, and a MRG domain near the C-terminus (Figure S1). MRG2 has a homolog in Arabidopsis, MRG1, which shares with MRG2 49% identity and 65% similarity at the amino acid sequence level.
To analyze the in vitro binding activity of MRG1 and MRG2, we tested whether their chromodomains could bind to H3 peptides containing different methylations at K4, K9, K27, or K36 using a pull-down assay. The chromodomains of MRG1 and MRG2 could bind to both the H3K4me2/me3 and H3K36me2/me3 peptides, but not to mono-methylated H3K4 and H3K36 peptides (Figure 1A). The two MRG proteins had a higher affinity to the tri-methylated peptides than to the di-methylated forms in the dot-blot binding assay (Figure 1A). In contrast, H3K9me3 and H3K27me3 were not recognized by either of these two proteins (Figure 1A), indicating a degree of MRG binding specificity. To further verify the specificity of the binding, an isothermal titration calorimetry (ITC) assay was performed using free-labeled histone peptides as substrates. H3K4me3 (residues 1–9) and H3K36me3 (residues 31–41) exhibited detectable binding to MRG2 chromodomain (residues 41–123) (Kd values: 0.8 mM for H3K4me3; 0.69 mM for H3K36me3) (Figure 1B). On the other hand, MRG2 chromodomain bound to H3K4me2 (residues 1–9), H3K36me2 (residues 31–41), H3K9me3 (residues 4–13) and H3K27me3 (residues 23–31) at a relatively low affinity (Kd values: 2.4 mM for H3K4me2; 2.6 mM for H3K36me2; 1.2 mM for H3K9me3; 2.5 mM for H3K27me3) (Figure S2). The ITC binding assay results were consistent with those of the in vitro pull-down assay, showing the binding specificity of MRG1/2 with tri-methylated H3K4 and H3K36 in vitro.
Fig. 1. Chromodomains of MRG1 and MRG2 specifically bind to tri-methylated H3K4 and H3K36 in vitro. 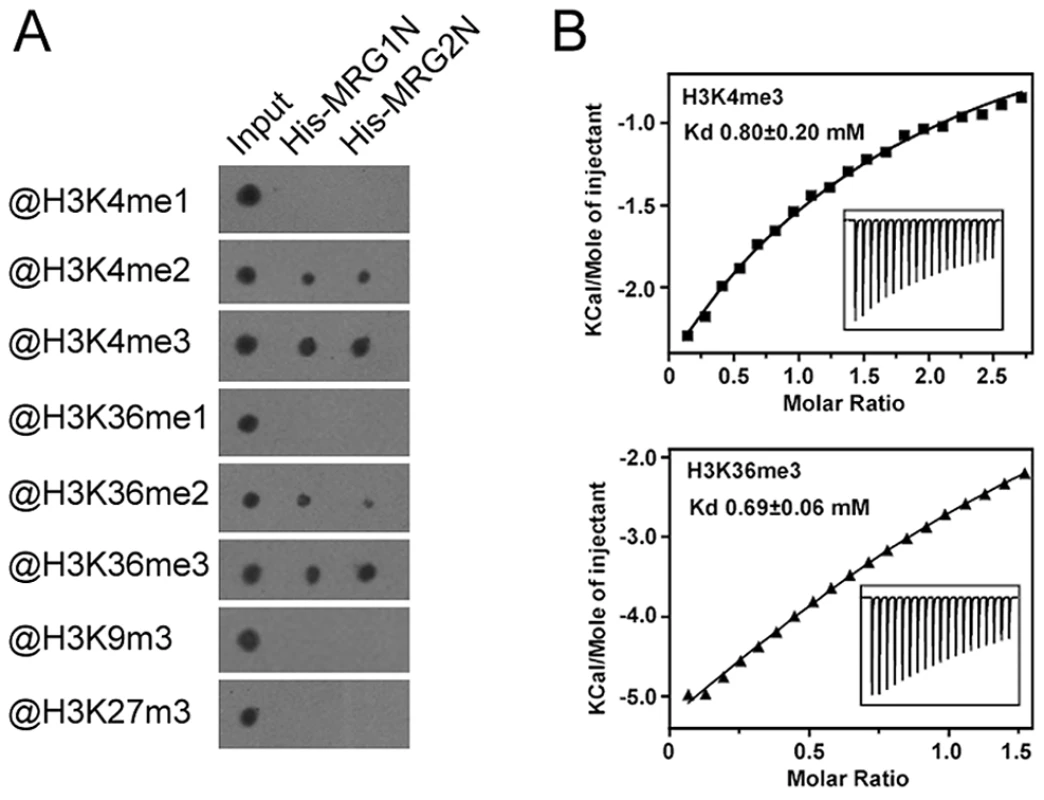
A. Binding assays of N-terminal His-tagged chromodomains of MRG1 (His-MRG1N) and MRG2 (His-MRG2N) with H3 peptides containing varying degrees of methylation at K4, K9, K27, or K36. B. ITC measurements of binding between the MRG2 chromodomain and histone peptides. Top panel, MRG2 and H3K4me3 peptide; bottom panel, MRG2 and H3K36me3 peptide. MRG1/2 promote photoperiodic flowering in Arabidopsis
To investigate the in vivo function of MRG1/2, the T-DNA insertional mutants mrg1 and mrg2, obtained from the Arabidopsis Biological Resource Center (ABRC, http://www.arabidopsis.org) and the Saskatoon collection [36] (http://aafc-aac.usask.ca/FST/), were used. The single mutants of mrg1 and mrg2 carry T-DNA insertions in the first intron and the second exon, respectively (Figure 2A). Although MRG1 and MRG2 transcripts were undetectable in both mutants (Figure 2B), their overall phenotypes were normal (Figure 2C). Next, we constructed and analyzed the mrg1 mrg2 double mutant, which showed late-flowering under long-day (LD) conditions (Figure 2C and 2E). Both MRG1 and MRG2 were mainly expressed in the vasculature of cotyledons and true leaves, and also in roots and inflorescences, as evidenced by histochemical GUS staining in PMRG1::MRG1-GUS and PMRG2::MRG2-GUS transgenic plants (Figure 2F). The late-flowering phenotype of mrg1 mrg2 double mutant could be fully rescued by introducing either PMRG1::MRG1-GUS or PMRG2::MRG2-GUS into the plants, indicating that MRG1 and MRG2 are functionally redundant in the control of flowering time in Arabidopsis. Additionally, the mrg1 mrg2 late-flowering phenotype was specific for LD conditions. Under short-day (SD) conditions, the double mutant showed a flowering time similar to the wild-type (Figure 2D and 2E), suggesting that the mrg1 mrg2 double mutant is defective in the photoperiodic flowering pathway.
Fig. 2. MRG1 and MRG2 act redundantly in flowering time control of Arabidopsis in the photoperiodic flowering pathway. 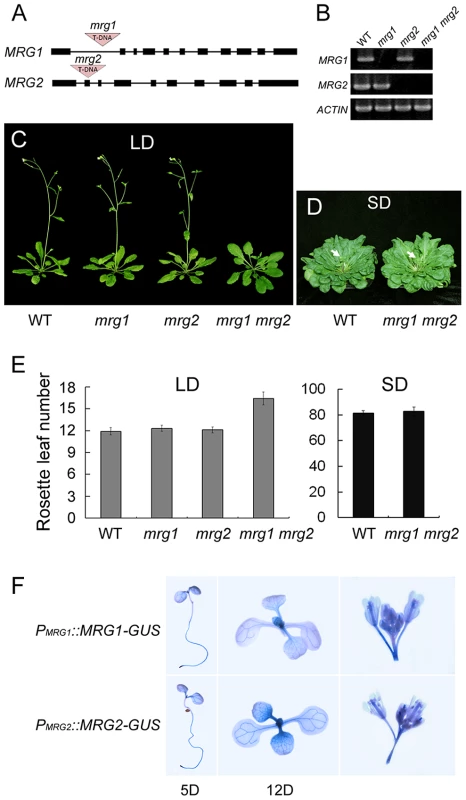
A. Gene structure of mrg1 and mrg2 mutant alleles. Dark boxes represent exons; lines represent introns; red triangles indicate T-DNA insertions. B. RT-PCR analysis of MRG1 and MRG2 expression in leaves of mrg1, mrg2, and mrg1 mrg2 plants. ACTIN2 was used as the internal control. C. Phenotypes of the wild-type (WT), the single mutants mrg1 and mrg2, and the double mutant mrg1 mrg2 grown under long-day photoperiods (LD; 16 h light: 8 h dark). D. Phenotypes of WT and mrg1 mrg2 plants grown under short-day photoperiods (SD; 8 h light: 16 h dark). E. Flowering time, as measured by rosette leaf number at bolting, in plants grown under LD and SD conditions. The mean value from 20 plants is shown. Error bars represent standard deviations. F. Tissue expression pattern analyses of MRG1 and MRG2 by histochemical GUS staining in PMRG1::MRG1-GUS and PMRG2::MRG2-GUS transgenic plants. Staining was performed at different times after seed germination (day 5 or 12), and in inflorescences. MRG1/2 are required for normal activation of FT expression
To investigate the effect of MRG1/2 on flowering time in response to changes of photoperiod, we traced expression of the key genes CO and FT over a 24-h period in wild-type and mrg1 mrg2 double mutant plants. The expression of CO and FT shows different diurnal patterns with their transcript levels [37]. Under LDs, high levels of wild-type CO expression are detected from late afternoon through the first half of the night, while induction of FT transcription occurs at the end of the day (16 h after dawn, ZT16) (Figure 3A). While the CO expression pattern in mrg1 mrg2 was similar to that of the wild-type, the FT transcript level in mrg1 mrg2 was lower than that of wild-type plants (Figure 3A), suggesting that MRG1 and MRG2 may be involved in activation of FT transcription in the photoperiodic flowering pathway.
Fig. 3. MRG1/2 are required for normal activation of FT expression. 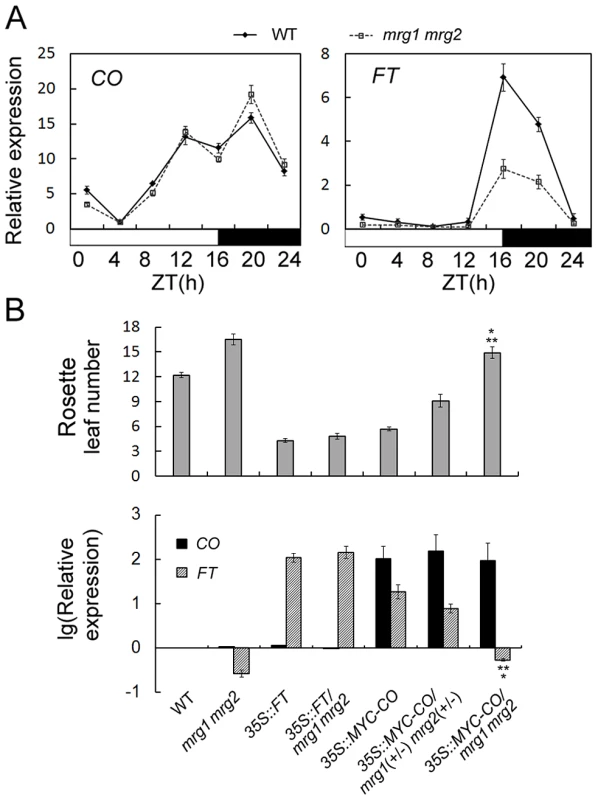
A. Diurnal expression of CO and FT genes in wild-type (WT) and mrg1 mrg2 double mutant. Time is expressed as hours from dawn (ZT, zeitgeber time). Values are normalized to ACTIN2. Error bars show standard deviation from three biological replicates. B. Top panel, flowering time of the wild-type (WT), mrg1 mrg2 double mutant, transgenic 35S::FT plants in wild-type (35S::FT) or in mrg1 mrg2 (35S::FT/mrg1 mrg2), transgenic 35S::MYC-CO plants in wild-type (35S::MYC-CO), in heterozygous for mrg1 mrg2 (35S::MYC-CO/mrg1(+/−) mrg2(+/−)), or in homozygous for mrg1 mrg2 (35S::MYC-CO/mrg1 mrg2) grown in LDs. The mean value from 20 plants is shown. Error bars represent standard deviations. Bottom panel, Relative expression of CO and FT in indicated genotypes at ZT16. Values are presented as logarithmic mode (lg) of relative expression normalized to ACTIN2. Error bars show standard deviation from three biological replicates. A single asterisk indicates the statistically significant difference between 35S::MYC-CO/mrg1 mrg2 plants and the wild-type (P<0.05), and double asterisks indicate the statistically significant difference between 35S::MYC-CO/mrg1 mrg2 plants and the mrg1 mrg2 double mutants (P<0.05). To further test where in the photoperiodic pathway MRG1/2 act, we introduced 35S::FT and 35S::MYC-CO constructs into the late-flowering mrg1 mrg2 double mutant through introgression of the transgenes. Transgenic plants overexpressing FT or CO in the wild-type flowered significantly earlier (Figure 3B) [37], [38]. Overexpression of FT fully rescued the late-flowering phenotype of the mrg1 mrg2 double mutant (Figure 3B), indicating that MRG1/2 indeed act upstream of FT. In contrast, overexpression of CO failed to induce FT transcription in mrg1 mrg2 background, and the plants still displayed late-flowering phenotype comparing with the wild-type plants, although these 35S::MYC-CO/mrg1 mrg2 plants showed slightly higher FT expression and slightly earlier flowering time comparing with the mrg1 mrg2 double mutant (Figure 3B). It's to be noted that the significantly increased expression levels of CO (around 100 folds of that in the wild-type) were similar in the 35S::MYC-CO transgenic plants in wild-type (2 copies of 35S::MYC-CO), heterozygous (1 copy of 35S::MYC-CO) or homozygous mrg1 mrg2 (2 copies of 35S::MYC-CO) backgrounds. To further verify this phenotype, another transgene 35S::FLAG-CO was also constructed and introduced into the mrg1 mrg2 double mutant. As shown in Figure S3, transgenic plants overexpressing FLAG-CO in the wild-type exhibited high level of FT transcription and early-flowering phenotype. However, similar to 35S::MYC-CO/mrg1 mrg2, overexpression of FLAG-CO also failed to induce FT transcription in mrg1 mrg2 background (with similar expression level of CO as that in the wild-type background, Figure S3) and the plants still flowered a little bit later than the wild-type, suggesting that the CO function in promoting flowering requires MRG1/2 proteins. When constructing 35S::MYC-CO/mrg1 mrg2 plants, we found that the 35S::MYC-CO transgenic plants in wild-type, heterozygous (1 copy of MRG1 and 1 copy of MRG2) or homozygous mrg1 mrg2 backgrounds exhibited different FT transcription levels and flowering time (Figure 3B), noting that the plants with either 1 copy or 2 copies of 35S::MYC-CO showed similar expression levels of CO. The heterozygous 35S::MYC-CO/mrg1(+/−) mrg2(+/−) plants, with about 30% reduction of MRG1 or MRG2 (Figure S4), showed FT activation and earlier flowering comparing with the wild-type plants, but their flowering time was still later than that of the 35S::MYC-CO plants in wild-type background, indicating that the requirement of MRG1/2 in the effect of CO overexpression is in a dosage-dependent manner. These results imply an important role for MRG1/2 in the photoperiod flowering pathway in Arabidopsis, and MRG1/2 are involved in the FT transcriptional activation.
MRG2 binds the chromatin at the FT promoter
To obtain evidence for the molecular mechanisms of regulation of FT by MRG1/2 proteins, we performed a Chromatin Immuno-Precipitation (ChIP) assay to analyze possible in vivo binding of MRG1/2 proteins to the FT locus. The antibody against MRG2 (Figure 4A) was used in ChIP assays in wild-type and mrg1 mrg2 plants. The endogenous MRG2 protein was obviously enriched in the FT promoter regions in wild-type comparing with that in mrg1 mrg2 double mutant (Figure 4B), indicating that the MRG2 protein targets directly to the FT promoter. To test whether MRG2 binding to the FT promoter region was dependent on the H3K4 and H3K36 methylation status of the chromatin, we used loss-of-function mutants atx1 and sdg8, which affect, respectively, the ATX1 gene encoding a H3K4 methyltransferase and the SDG8 gene encoding a H3K36 methyltransferase [39], [40]. As a control, the atx1 and sdg8 mutations did not affect the expression levels of MRG1/2 (Figure 4A and 4C). ChIP analysis indicated that atx1 and sdg8 mutants showed reduced H3K4me3 and H3K36me3 levels, respectively, in most FT chromatin regions, when compared with wild-type plants (Figure 4B). A detectable reduced H3K4me3 levels in some FT promoter regions were observed in sdg8, while the H3K36me3 levels of FT chromatin were not affected in atx1 comparing with wild-type. The overall H3K4me3 and H3K36me3 levels of FT in mrg1 mrg2 double mutant were similar as those in the wild-type plants, indicating that the deletions of MRG1 and MRG2 do not change the H3K4me3 and H3K36me3 levels of FT chromatin. Along with reduced H3K4me3 and H3K36me3 levels, MRG2 enrichment at the FT promoter in atx1 and sdg8 mutants was decreased (Figure 4B), suggesting that MRG2 may directly bind to the FT promoter via recognition of methylated H3K4 and H3K36 in planta.
Fig. 4. MRG2 binds the chromatin at the FT promoter. 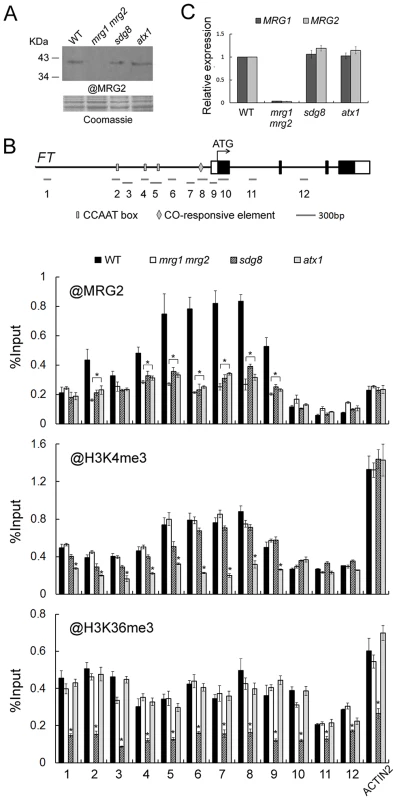
A. Protein levels of MRG2 in WT, mrg1 mrg2, sdg8, and atx1 plants at ZT16 verified by the antibody against MRG2. B. ChIP analysis using @MRG2, @H3K4me3 or @H3K36me3 at FT chromatin in WT, mrg1 mrg2, sdg8, and atx1 plants at ZT16. Error bars show standard deviation from three biological replicates. Asterisks indicate statistically significant differences between the indicated genotypes and the wild-type (P<0.01). C. Relative expression levels of MRG1 and MRG2 in indicated genotypes at ZT16. Values are normalized to ACTIN2. Error bars show standard deviation from three biological replicates. MRG2 and CO interact physically and enhance each other's binding to the FT promoter
The FT promoter contains several canonical CCAAT boxes and one CO-responsive element (CORE), these are essential for CO-mediated FT activation. Given that the CCAAT boxes and CORE sequence are within the MRG2-enriched regions of the FT promoter, we hypothesized that MRG1/2 proteins might be involved in the CO-dependent regulation of FT expression. To test this hypothesis, firstly we introduced a co mutant into the mrg1 mrg2 double mutant and found that the co mrg1 mrg2 triple mutant flowered at nearly the same time as co in LDs (Figure 5A), suggesting that MRG1/2 may act to regulate FT in a CO-dependent genetic pathway.
Fig. 5. MRG2 physically interacts with CO. 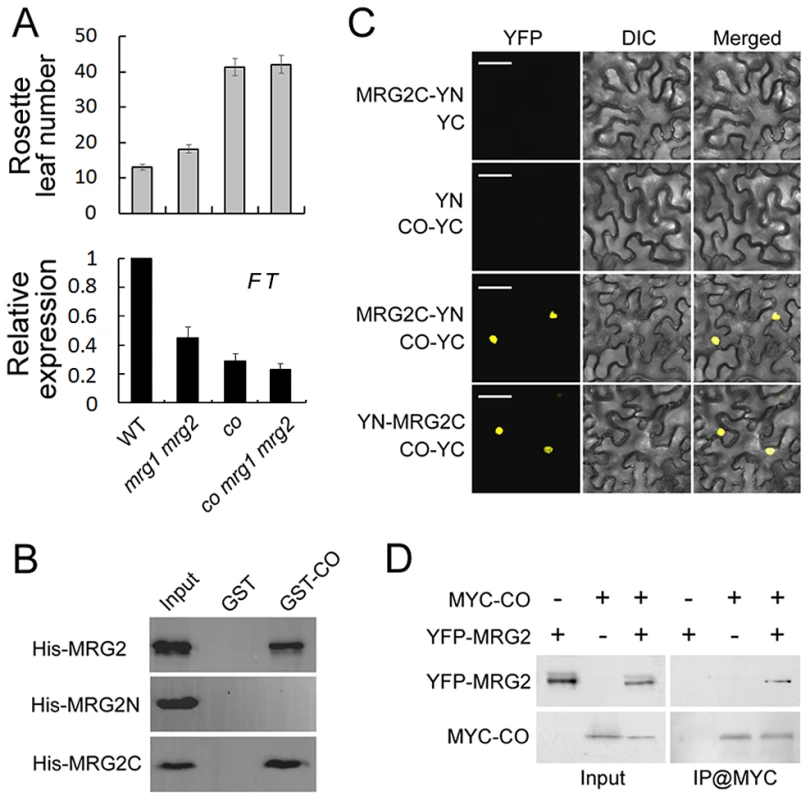
A. Top panel, flowering time of WT, mrg1 mrg2, co and co mrg1 mrg2, as measured by rosette leaf number at bolting, in plants grown under LD conditions. The mean value from 20 plants is shown. Error bars represent standard deviations. Bottom panel, relative expression of FT in indicated genotypes at ZT16. Values are normalized to ACTIN2. Error bars show standard deviation from three biological replicates. B. Interaction of purified His-tagged MRG2 and GST-fused CO from E. coli by pull-down assays. C. BiFC analysis of the interaction between MRG2 and CO in tobacco leaf cells. Bar = 50 µm. D. Co-IP detection of MRG2 and CO interaction in planta. Total protein extracts from plants expressing YFP-MRG2 alone, MYC-CO alone, or both YFP-MRG2 and MYC-CO were immunoprecipitated with anti-MYC affinity beads and the resulting fractions analyzed by western blot using anti-GFP antibodies (top panel) or HRP-conjugated anti-MYC monoclonal antibodies (bottom panel). The facts that MRG1/2 and CO are both positive regulators of FT expression, that they both express mainly in the vasculature of leaves, and that they bind to the overlapped regions of FT promoter ([41] and this study) drive us to test whether MRG1/2 might interact with CO physically. To test this hypothesis, we examined the protein-protein interaction between MRG2 and CO. In an in vitro pull-down assay, we found that GST-fused CO but not GST alone could pull-down MRG2 tagged with multiple His (histidine) residues (Figure 5B). To determine which domain in MRG2 was required for this interaction, we performed a truncation analysis and found that the MRG domain (MRG2C) but not the chromodomain (MRG2N) of MRG2 contributed to binding to CO (Figure 5B). To verify the interaction observed in the pull-down assay, an in vivo binding assay was performed using bimolecular fluorescence complementation (BiFC) analysis. When tobacco leaves were infiltrated with Agrobacterium cells carrying appropriate constructs, MRG2/MRG2C and CO proteins tagged with split YFP interacted in vivo to reconstitute the whole YFP protein with fluorescent signal; therefore only the interaction of MRG2C and CO is shown in Figure 5C. Further support for a physical interaction between MRG2 and CO in planta came from a co-immunoprecipitation (CoIP) experiment, in which YFP-MRG2 was detected in the MYC-CO immunoprecipitated fraction from transgenic Arabidopsis expressing both MYC-CO and YFP-MRG2 (Figure 5D).
To explore the role of CO and MRG1/2 protein-protein interaction at the FT promoter, we examined MRG2 binding at FT upon loss of CO function by a ChIP assay using the anti-MRG2 antibody in the co-1 mutant. Loss of CO function resulted in declined MRG2 binding to the FT promoter (Figure 6A) without changes of the MRG1/2 transcription levels (Figure S5A) and H3K4me3/H3K36me3 levels in FT promoter (Figure S5B), indicating that CO is required for normal levels of MRG2 binding to the FT chromatin, and supporting the genetic result that the MRG1/2 function in promoting flowering depends on CO (Figure 5A). Conversely, to test whether MRG1/2 proteins affect the level of CO at the FT promoter, 35S::MYC-CO/mrg1 mrg2 plants were used for a ChIP analysis of the MYC-CO protein with anti-MYC antibody. As observed previously [41], CO peaks near the FT transcription start site where the CORE sequence is located (Figure 6B). In addition, upstream of this CO peak, there is a relatively low but detectable enrichment of CO at the FT promoter (Figure 6B), where several canonical CCAAT boxes are embedded. Western blot analysis of total protein extracts revealed that CO protein accumulated at similar levels in wild-type and the mrg1 mrg2 double mutant (Figure 6C). Surprisingly, CO enrichment at the FT promoter decreased in mrg1 mrg2 (Figure 6B), implying that MRG1/2 proteins may interact with CO at the FT promoter, and affect the stable binding of CO at the locus. This result is also consistent with that the overexpression of CO failed to rescue the mrg1 mrg2 double mutant late-flowering phenotype (Figure 3B). Taken together, both our biochemical and genetic analyses demonstrate that MRG2 and CO physically interact and depend on each other for their stable association with the FT promoter chromatin to promote flowering.
Fig. 6. MRG2 and CO enhance each other's binding to the FT promoter. 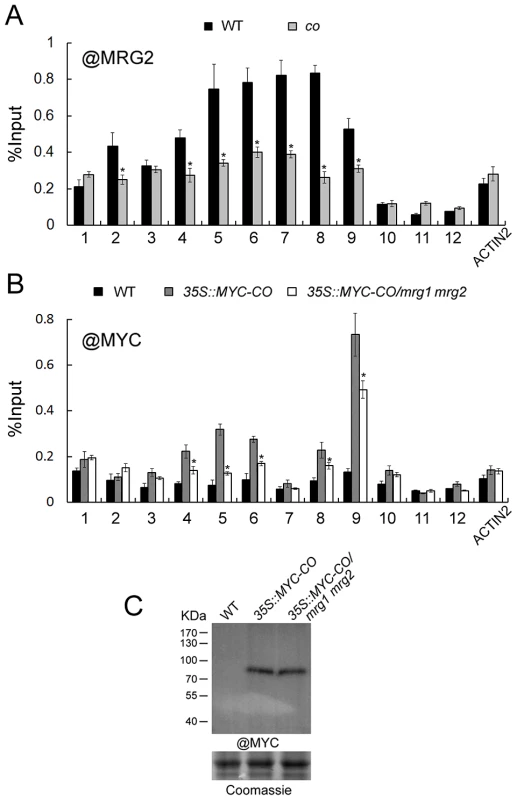
A. Enrichment of MRG2 at the FT promoter at ZT16 in wild-type and co mutant at ZT16. Error bars show standard deviation from three biological replicates. Asterisks indicate statistically significant differences between co and the wild-type (P<0.01). B. Enrichment of MYC-CO at the FT promoter at ZT16 in indicated genotypes at ZT16. Error bars show standard deviation from three biological replicates. Asterisks indicate statistically significant differences between 35S::MYC-CO/mrg1 mrg2 plants and 35S::MYC-CO plants (P<0.01). C. Protein levels of MYC-tagged CO in indicated genotypes at ZT16. Co-crystal structures of MRG2 chromodomain and methylated H3 peptides reveal the residues for recognition of H3K4me3 or H3K36me3
To obtain the information regarding the specific MRG2 residues that are important for recognition of the histone marks H3K4me3 and H3K36me3, we determined the co-crystal structure of MRG2 and the methylated H3 peptides. To identify the suitable length of MRG2 chromodomain-containing fragments for crystallization, limited proteolysis was performed to remove unstructured flexible regions, because these tend to be difficult to crystalize. The proteolyzed samples were analyzed using mass spectrometry (MS) analysis to determine the protein sequence, resulting in the identification of a region containing the chromodomain with residues 53–123 of MRG2 as suitable for crystallization. MRG2 complexes with the H3K4me3 and H3K36me3 peptides were obtained, but the complexes with the H3K27me3 and H3K9me3 peptides could not form crystals under the same conditions, consistent with the ITC results (Figure S2). The crystals of MRG2 with H3K4me3 or H3K36me3 were diffracted to 1.68 and 1.65 Å, respectively (Table S1). The structures were solved by the molecular replacement method using the chromodomain of human MRG15 (PDB entry 2F5K) as template. In both structures, the MRG2 chromodomain contains mainly four beta-strands. However, the predicted alpha-helix located at the C-terminus is missing in the electron density map (Figure S1A and 7A). For the complexes of the MRG2 chromodomain with the H3K4me3/H3K36me3 peptides, the recognition modes are almost the same; therefore, only the complex of the MRG2 chromodomain with H3K36me3 peptide is shown in Figure 7A. The tri-methylated lysine projects into the aromatic cage formed by the His62, Tyr67, Tyr87, Trp90, and Trp94 residues of the MRG2 chromodomain (Figure 7A). The co-crystal structures clearly indicate that the chromodomain of MRG2 can directly bind to tri-methylated H3K4 and H3K36, and provide strong evidence that the aromatic cage of the MRG2 chromodomain and the associated residues are essential for the binding, the same as other chromodomain proteins [29].
Fig. 7. MRG1/2 binding to histone marks is required for their function in regulating flowering. 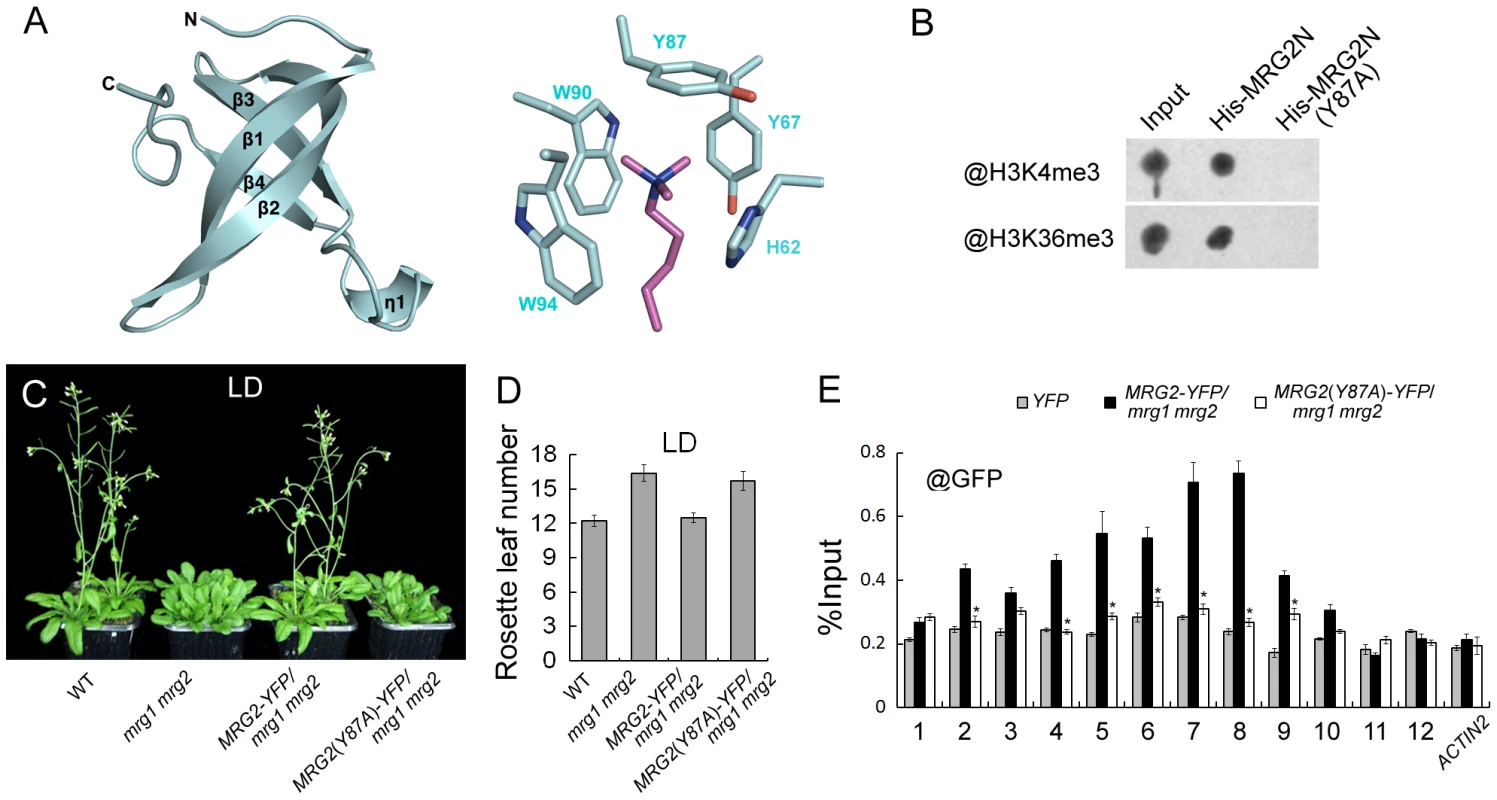
A. Structure of the MRG2 chromodomain in complex with H3K36me3. The overall structure of the MRG2 chromodomain consisting of four β strands is shown in the left panel. The tri-methylated lysine sticking in the aromatic cage was surrounded by five conserved residues (right panel). Tri-methylated lysine is shown in purple; residues forming the aromatic cage are shown in cyan. B. Binding assays of His-tagged chromodomains of MRG2 (His-MRG2N) and a Y87A substitution in MRG2 chromodomain (His-MRG2N(Y87A)) with H3K4me3 and H3K36me3. C. Phenotypes of wild-type (WT), mrg1 mrg2, PMRG2::MRG2-YFP/mrg1 mrg2 (MRG2-YFP/mrg1 mrg2), and PMRG2::MRG2(Y87A)-YFP/mrg1 mrg2 (MRG2(Y87A)-YFP/mrg1 mrg2) plants grown under long-day photoperiods (LD; 16 h light: 8 h dark), noting that the mrg1 mrg2 double mutants have bolted. D. Flowering time of indicated genotypes, as measured by rosette leaf number at bolting, in plants grown under LD conditions. The mean value from 20 plants is shown. Error bars represent standard deviations. E. ChIP analysis using @GFP at FT chromatin in indicated genotypes at ZT16. Error bars show standard deviation from three biological replicates. Asterisks indicate statistically significant differences between MRG2(Y87A)-YFP/mrg1 mrg2 plants and MRG2-YFP/mrg1 mrg2 plants (P<0.01). MRG1/2 binding to histone marks is required for their biological function in vivo
According to the co-crystal structures described above, we specifically replaced Tyr87 with Ala. Consistent with the structure, the Tyr87Ala (Y87A) mutation in MRG2 disrupted its association with H3K4me3 and H3K36me3 peptides (Figure 7B), supporting the idea that the hydrophobic pocket formed by the aromatic residues is essential for the binding, as with other chromodomain proteins [29].
To further test whether the Tyr87 residue is important for in vivo MRG2 function, we constructed PMRG2::MRG2-YFP and PMRG2::MRG2(Y87A)-YFP fusions and introduced them into the mrg1 mrg2 double mutant. As expected, the late-flowering phenotype of the double mutant could be fully rescued by the PMRG2::MRG2-YFP transgene (Figure 7C and 7D). On the other hand, the PMRG2::MRG2(Y87A)-YFP transgene could not rescue the mrg1 mrg2 mutant phenotype (Figure 7C and 7D), suggesting that the capability of the MRG2 protein to bind the methylated histones is essential for its biological function in vivo.
We then used the PMRG2::MRG2-YFP/mrg1 mrg2 and PMRG2::MRG2(Y87A)-YFP/mrg1 mrg2 transgenic plants with similar mRNA and protein expression levels of MRG2-YFP and MRG2(Y87A)-YFP (Figure S6) for ChIP assays using a specific antibody against GFP. At the FT promoter, the MRG2-YFP proteins in the PMRG2::MRG2-YFP/mrg1 mrg2 plants showed a similar enrichment pattern to the endogenous MRG2 protein (Figure 7E and 4B). However, in the PMRG2::MRG2(Y87A)-YFP/mrg1 mrg2 plants (Figure 7E), in which the mutated MRG2 protein failed to bind to the tri-methylated H3K4 or H3K36 (Figure 7B), the enrichment of MRG2(Y87A)-YFP proteins to FT were obviously decreased, showing that the association of MRG2 with FT promoter depends on its H3K4me3/H3K36me3 binding activity, thus providing a direct link between the biochemical activity of MRG1/2 and their in vivo biological functions.
Discussion
The photoperiodic regulation of flowering is widely observed among flowering plants. In Arabidopsis, this pathway requires the key regulator CO and its target gene FT. Although some chromatin modifiers have been reported to be involved in FT regulation, little is known regarding direct interaction between chromatin effectors and the CO-FT pathway. In this work, we present Arabidopsis histone mark readers MRG1/2 as novel chromatin effectors that interact with both the CO protein and the FT promoter, thus providing a chromatin regulatory mechanism linking the transcription factor CO and the H3K4/36-methylation readers MRG1/2 in FT activation to promote plant flowering under long-day photoperiods.
Firstly, via their chromodomain, MRG1/2 proteins act as readers that recognize methylated H3K4 and H3K36. Unlike EAF3, which bind to H3K36me3/2 and only very weakly to H3K4me3/2 [29], our in vitro binding and crystal structure data revealed that the chromodomain of MRG2 has a preference to interact with tri-methylated forms of both H3K4 and H3K36. The ChIP assays revealed that MRG2 binds to the FT promoter at positions where the tri-methylated H3K4 is enriched (Figure 4B). Loss of ATX1 function resulted in a decrease of MRG2 enrichment at the FT promoter without change of MRG1/2 expression levels (Figure 4C). Unlike H3K4me3 distribution, which associated with MRG2 binding pattern, we found that H3K36me3 was homogeneously distributed along FT promoter (Figure 4B). The sdg8 mutant exhibited slightly decreased levels of H3K4me3, and significant reductions of H3K36me3 and MRG2 enrichment in most FT chromatin regions also with no change of MRG1/2 expression (Figure 4C). Given that the slightly reduced H3K4me3 level in sdg8 might not be sufficient for a severe decrease of MRG binding ability, H3K36me3 is very likely to contribute in the MRG binding in vivo. The co mutant again showed declined MRG2 binding to the FT promoter (Figure 6A) without changes of the MRG1/2 transcription levels (Figure S5A). Therefore, we propose that H3K4me3/H3K36me3 and transcription factor CO may play together to specify the MRG2 enrichment in FT chromatin in planta.
The binding capability of the MRG1/2 proteins with H3K4me3/H3K36me3 is essential for their biological function in planta because the Y87A substitution in the chromodomain of MRG2, which lost the H3K4me3/H3K36me3 binding capacity, failed to rescue the mrg1 mrg2 double mutant late-flowering phenotype (Figure 7). However, kinetic analysis in vitro revealed that MRG2 binding affinities with H3K4me3 and H3K36me3 are fairly weak (Figure 1B). Therefore, MRG2 chromodomain itself may have difficulty in remaining associated with methylated histone tails without the assistance of other factors. Here, participation of another domain, namely the MRG domain, provides extra binding affinity and specificity to exert biological function. We demonstrated that MRG2 interacts with CO via the MRG domain, and that this interaction is required for stable binding of MRG2 to H3K4me3 and H3K36me3 at the FT promoter, as loss of CO function results in reduction of MRG2 enrichment at the FT locus.
On the other hand, although the effect of mrg1 mrg2 double mutant on flowering time in LDs is comparably mild, MRG binding to FT is important for CO-dependent FT activation because in mrg1 mrg2 background, overexpression of MYC-CO (Figure 3B) or FLAG-CO (Figure S3) failed to induce FT activation, and the transgenic plants still displayed late-flowering phenotype comparing with the wild type plants. Furthermore, the requirement of MRG proteins for CO overexpression is in a dosage-dependent manner (Figure 3B). As to the mild late flowering phenotype of mrg1 mrg2 double mutant, we speculate that there might be redundant factors to help CO to induce FT transcription, for in mrg1 mrg2 double mutant, FT expression is down-regulated but not abolished. Overexpression of MRG2 by introducing 35S::MRG2 into the wild-type plants do not affect their flowering time, indicating that the increased MRG2 is not sufficient to induce early flowering and the endogenous MRG proteins are enough. Now we do not know the reason why the deletion of MRGs repressed the effect of CO overexpression, and one possibility may be that without MRG proteins, an more effective combination of CO at FT chromatin probably could not be established to induce a major change in FT activation. CO protein is stabilized towards the end of LDs, and its abundance declines rapidly in the dark [4], [42]. MRG1/2 do not seem to affect the stability of CO proteins since they accumulate at similar levels in both wild-type and the mrg1 mrg2 double mutant at ZT16 (Figure 6C). Therefore, we propose that MRG1/2 are critical for stabilizing CO recruitment at the FT locus. This reinforcement mechanism may resemble the role of H3K4me3 binding by TAF3 in the TFIID complex, in which TAF3 acts as a transcriptional coactivator of the basal transcription factor TFIID in a PHD finger-dependent manner [43].
In summary, our findings strongly support a model in which MRG1/2 proteins interact with CO to activate FT transcription. CO directly binds to the FT promoter, enhancing the recruitment of MRG1/2 proteins to the FT locus. In addition, MRG1/2 proteins bind to chromatin that contains tri-methylated H3K4 and H3K36 via their chromodomains, the bound MRG1/2 in turn stabilize the binding of CO and ultimately controls the activation of FT transcription. Therefore, MRG1/2 act as a novel type of chromatin modulators, linking H3K4/H3K36 methylations and CO in FT activation in the photoperiodic flowering regulation in plants.
How FT expression is modulated after CO and MRG1/2 binding to trigger correct flowering transition remains to be investigated. Evidence from yeast and animals shows that EAF3 and MRG15 are present in both histone acetyltransferase (HAT) and deacetylase (HDAC) complexes, and are involved in the regulation of chromatin structure [32], [44], [45]. It is therefore possible that transcriptional activation of FT might be due to the recruitment of subsequent chromatin effectors, such as the HAT complex. Another important issue is how FT expression is rhythmically regulated. Light signaling regulates CO protein stability and acts to generate daily rhythms in CO abundance [4], [42]. We propose that the association of CO with MRG1/2 may help its re-association with the FT promoter as MRG1/2 might be more stably bound to FT due to the overall stability of H3K4me3 and H3K36me3. Gu et al. revealed a periodic histone deacetylation mechanism for dampening FT mRNA expression at dusk, thereby modulating day length-dependent FT expression [15]. Further exploration will help to clarify the chromatin mechanism involved in the photoperiodic regulation of flowering time control.
Materials and Methods
Dot-blot binding assay
For the dot-blot binding assay, MRG1N (1–321, AT4G37280) and MRG2N (1–369, AT1G02740) cDNA were amplified by primers MRG1-1/MRG1-2 and MRG2-1/MRG2-2, respectively, and cloned into a pET-30a vector (Novagen, www.novagen.com). Site mutation was generated using a Takara MutanBEST Kit (http://www.takara-bio.com) with primers MRG2-3/MRG2-4. The in vitro binding assay of the chromodomain containing proteins with the histone peptides were performed as described previously [46]. Methylated H3K4 (H3K4me1/2/3, residues 1–21), H3K9 (H3K9me1/2/3, residues 1–21), H3K27 (H3K27me1/2/3, residues 21–44), and H3K36 (H3K36me1/2/3, residues 21–44) peptides were synthesized by Scilight Biotechnology (http://www.scilight-peptide.com). Anti-monomethyl-H3K4 (07-436), anti-dimethyl-H3K4 (07-030), anti-trimethyl-H3K4 (07-473), anti-trimethyl-H3K9 (07-442), and anti-trimethyl-H3K27 (07-449) were purchased from Millipore (http://www.millipore.com), and anti-monomethyl-H3K36 (ab9048), anti-dimethyl-H3K36 (ab9049), and anti-trimethyl-H3K36 (ab9050) were purchased from Abcam (http://www.abcam.com).
Isothermal titration calorimetry (ITC)
ITC experiments were carried out at 25°C on a MicroCal iTC200 (GE Healthcare, www.gelifesciences.com). Protein and peptide were kept in an identical buffer of 50 mM Tris pH 8.0, 100 mM NaCl. The sample cell was filled with a 0.2–0.4 mM solution of protein, and peptide (2–4 mM) was added sequentially in 2 µl aliquots (total of 20 injections) at 2.5 min intervals. Binding isotherms were analyzed by fitting data into the one-site model using the ITC data analysis module Origin 7.0.
Plant materials and growth conditions
All Arabidopsis alleles were derived from the Columbia ecotype. mrg1 and mrg2 alleles, corresponding to SALK_057762 and SK28487 T-DNA insertion lines respectively, were obtained from the Arabidopsis Biological Resource Center (ABRC, http://www.arabidopsis.org) and the Saskatoon collection [36] (http://aafc-aac.usask.ca/FST/). The mrg1 mrg2 double mutant was created in our laboratory by genetic crossing. The mutants sdg8-2, atx1-2, and co-1 have been previously described [39], [40], [47], as have the 35S::FT and 35S::MYC-CO lines [38], [48]. Higher order combinations of mutants were produced by genetic crossing. In vitro plant culture was performed on agar-solidified MS medium M0255 (Duchefa, http://www.duchefa.com) supplemented with 1% sucrose and 0.9% agar. The studied photoperiods were 16 h light and 8 h dark for long-day (LD), and 8 h light and 16 h dark for short-day (SD).
Complementation of mutant plants and histochemical GUS activity assay
For GUS staining, MRG1 and MRG2 genomic sequences were amplified by primers MRG1-3/MRG1-4 and MRG2-5/MRG2-6, respectively. They were fused to the GUS genomic sequence, and then cloned into pCAMBIA1300 (CAMBIA, http://www.cambia.org). The resulting constructs of PMRG1::MRG1-GUS and PMRG2::MRG2-GUS were used to transform the mrg1 mrg2 plants. GUS activity was assayed by incubating plant tissues in GUS staining buffer [49] for 3–6 hours at 37°C. Plant material was cleared in 70% ethanol, and observed directly under a dissecting microscope (MZ10F, Leica, Germany).
Gene expression analysis
Total RNA was prepared from plant tissues using TRI Reagent according to the manufacturer's instructions (Invitrogen, http://www.invitrogen.com). Reverse transcription was performed using standard procedures with Improm-II reverse transcriptase (Promega, http://www.promega.com). PCR amplification from the cDNA template was performed using gene-specific primers (see Table S2). ACTIN2 was used as a reference gene to normalize the data.
Construction of MRG–YFP/YFP-MRG fusions and plant transformation
MRG2 genomic sequence, amplified by primers MRG2-5/MRG2-6 and fused with EYFP cDNA, was cloned into the pCAMBIA1300 vector. Site mutation was generated using a Takara MutanBEST Kit (http://www.takara-bio.com) with primers MRG2-7/MRG2-8. The resulting constructs PMRG2::MRG2-YFP and PMRG2::MRG2(Y87A)-YFP were used to transform the mrg1 mrg2 plants.
MRG2 cDNA, amplified by primers MRG2-1/MRG2-10 and fused with EYFP cDNA, was cloned into the pER8 vector [50], downstream to an estrogen inducible promoter, and the construct of pER8::YFP-MRG2 was transformed into the wild-type Arabidopsis.
Chromatin Immunoprecipitation (ChIP)
ChIP was performed as previously described [39] with minor modifications. After fixation, 14-day-old seedlings were ground crudely in liquid nitrogen. Low and high salt wash buffers were supplemented with 0.1% Triton X-100. Antibodies used in this study were anti-GFP (A-11122, Invitrogen, http://www.invitrogen.com), anti-MYC (11667149001, Roche, http://www.roche-applied-science.com), anti-trimethyl-H3K4 (07-473, Millipore, http://www.millipore.com), and anti-trimethyl-H3K36 (ab9050, Abcam, http://www.abcam.com). The polyclonal antibody against MRG2 was produced by Abmart (http://www.abmart.com.cn). Quantitative real-time PCR was performed with a kit from Takara (http://www.takara-bio.com) to determine the enrichment of DNA immunoprecipitated in the ChIP experiments, using gene-specific primers listed in Table S2. The efficiency values are the ratios determined by taking a fixed aliquot of the DNA extracted from the immunoprecipitated samples and the Input. Error bars show standard deviation from three paralleled biological replicates.
Pulldown assay
MRG2 and MRG2C (442–984) cDNA were amplified with primers MRG2-1/MRG2-10 and MRG2-9/MRG2-10, respectively, and then cloned into pET-30a vector (Novagen, www.novagen.com). The resulting constructs and pET-30a-MRG2N were used for purification of His-tagged MRG2, MRG2C, and MRG2N proteins. CO cDNA was amplified using primers CO-1/CO-2, and then cloned into pGEX-4T-1 vector for purification of GST-fused CO proteins. Pulldown experiments were performed according to a previously described protocol [51].
Bimolecular fluorescence complementation (BiFC) assay
BiFC was performed as described by Sun et al. [52]. For BiFC assays, MRG2C cDNA was amplified using primers MRG2-11/MRG2-12, and then cloned into pXY103 and pXY106 vectors [52]. CO cDNA was amplified by primers CO-3/CO-4, and then cloned into a pXY104 vector. Leaves of 4 - to 8-week-old Nicotiana benthamiana plants were co-infiltrated with Agrobacterium tumefaciens strain GV1301 carrying transgene constructs. Localization of BiFC fluorescence was observed 2–3 days after infiltration using a confocal laser scanning microscope (LSM 710, ZEISS, Germany).
Co-immunoprecipitation (Co-IP) assays
14-day-old seedlings expressing YFP-MRG2 (pER8::YFP-MRG2/WT), MYC-CO (35S::MYC-CO/WT), or both YFP-MRG2 and MYC-CO were grown on MS medium with 4 µm estrogen to induce the overexpression of YFP-MRG2, and a Co-IP assay was performed as described previously [21]. IP was performed using anti-MYC affinity gel (E6654, Sigma, http://www.sigmaaldrich.com). Immunoprecipitated proteins and the input fractions were separated on a 10% SDS-PAGE and detected by western blotting using anti-GFP antibodies (A-11122, Invitrogen, http://www.invitrogen.com) or HRP-conjugated anti-MYC monoclonal antibodies (11814150001, Roche, http://www.roche-applied-science.com).
Crystallization and structure determination
The cDNA encoding the chromodomain of Arabidopsis thaliana MRG2 (residues 41–123 and 53–123) were amplified by PCR, and cloned into a pET28-SMT3 vector [53]. Following purification and removal of the tag, target proteins were concentrated to 20 mg/ml for structural and biochemical studies. To optimize the construct for crystallization, limited proteolysis was performed, and samples treated with endoproteinase Glu-C gave a single band on SDS-PAGE; the digested fragment was identified as residues from 41 to 108 by Mass Spectrometry.
A solution of 12 mg/ml MRG2 chromodomain (residues 41–123) was incubated for 2 hours with H3K36me3 peptide and endoproteinase Glu-C at a 200∶400∶1 molar ratio before crystallization. MRG2 chromodomain (residues 53–123) was incubated with H3K4me3/H3K36me3 peptide in the same way, with the exception of the Glu-C treatment. H3K4me3 and H3K36me3 peptides used for crystallization were synthesized, and indicated as residues 1–9 and 31–41, respectively. Crystallization was performed using the hanging drop vapor diffusion method. Crystals were grown at 16°C by mixing 1 µl of the protein solution with 1 µl precipitant solution. Crystals of MRG2 (residues 41–108) in complex with H3K36me3 peptide were grown under conditions of 0.1 M HEPES pH 7.5, 12% PEG 6000, 5% MPD. Crystals of MRG2 (residue 53–123) in complex with H3K4me3/H3K36me3 were grown under conditions of 0.1 M MES pH 6.0, 27% PEG MME 5000, 0.2 M ammonium sulfate. Diffraction data were collected from flash-cooled crystals at 100K at SSRF (Shanghai Synchrotron Radiation Facility). The data was processed using HKL2000 [54]. Molecular-replacement solutions were generated using the Phaser in Phenix program and the crystal structure of MRG15 chromo domain (PDB entry code 2F5K) used as a search model. The initial models were built with COOT [55] and refined using Phenix [56]. The final refined structure was represented by Pymol (The PyMOL Molecular Graphics System, Version 1.4.1 Schrödinger, LLC). Coordinates have been deposited under PDB accession code 4PL6, 4PLI, and 4PLL.
Supporting Information
Zdroje
1. AmasinoR (2010) Seasonal and developmental timing of flowering. Plant J 61 : 1001–1013.
2. IetswaartR, WuZ, DeanC (2012) Flowering time control: another window to the connection between antisense RNA and chromatin. Trends Genet 28 : 445–453.
3. AndresF, CouplandG (2012) The genetic basis of flowering responses to seasonal cues. Nat Rev Genet 13 : 627–639.
4. TurckF, FornaraF, CouplandG (2008) Regulation and Identity of Florigen: FLOWERING LOCUS T Moves Center Stage. Annual Review of Plant Biology 59 : 573–594.
5. TiwariSB, ShenY, ChangHC, HouY, HarrisA, et al. (2010) The flowering time regulator CONSTANS is recruited to the FLOWERING LOCUS T promoter via a unique cis-element. New Phytol 187 : 57–66.
6. WenkelS, TurckF, SingerK, GissotL, Le GourrierecJ, et al. (2006) CONSTANS and the CCAAT box binding complex share a functionally important domain and interact to regulate flowering of Arabidopsis. Plant Cell 18 : 2971–2984.
7. HeY (2012) Chromatin regulation of flowering. Trends in Plant Science 17 : 556–562.
8. JiangD, WangY, HeY (2008) Repression of FLOWERING LOCUS C and FLOWERING LOCUS T by the Arabidopsis Polycomb repressive complex 2 components. PLoS One 3: e3404.
9. FarronaS, ThorpeFL, EngelhornJ, AdrianJ, DongX, et al. (2011) Tissue-specific expression of FLOWERING LOCUS T in Arabidopsis is maintained independently of polycomb group protein repression. Plant Cell 23 : 3204–3214.
10. Lopez-VernazaM, YangS, MullerR, ThorpeF, de LeauE, et al. (2012) Antagonistic roles of SEPALLATA3, FT and FLC genes as targets of the polycomb group gene CURLY LEAF. PLoS One 7: e30715.
11. LuF, CuiX, ZhangS, JenuweinT, CaoX (2011) Arabidopsis REF6 is a histone H3 lysine 27 demethylase. Nat Genet 43 : 715–719.
12. JeongJH, SongHR, KoJH, JeongYM, KwonYE, et al. (2009) Repression of FLOWERING LOCUS T chromatin by functionally redundant histone H3 lysine 4 demethylases in Arabidopsis. PLoS One 4: e8033.
13. LuF, CuiX, ZhangS, LiuC, CaoX (2010) JMJ14 is an H3K4 demethylase regulating flowering time in Arabidopsis. Cell Res 20 : 387–390.
14. YangW, JiangD, JiangJ, HeY (2010) A plant-specific histone H3 lysine 4 demethylase represses the floral transition in Arabidopsis. Plant J 62 : 663–673.
15. GuX, WangY, HeY (2013) Photoperiodic Regulation of Flowering Time through Periodic Histone Deacetylation of the Florigen Gene FT. PLoS Biology 11: e1001649.
16. ShafiqS, BerrA, ShenW-H (2014) Combinatorial functions of diverse histone methylations inArabidopsis thalianaflowering time regulation. New Phytologist 201 : 312–322.
17. LiuC, LuF, CuiX, CaoX (2010) Histone Methylation in Higher Plants. Annual Review of Plant Biology 61 : 395–420.
18. BerrA, ShafiqS, ShenWH (2011) Histone modifications in transcriptional activation during plant development. Biochim Biophys Acta 1809 : 567–576.
19. de la Paz SanchezM, GutierrezC (2009) Arabidopsis ORC1 is a PHD-containing H3K4me3 effector that regulates transcription. Proc Natl Acad Sci U S A 106 : 2065–2070.
20. LeeWY, LeeD, ChungWI, KwonCS (2009) Arabidopsis ING and Alfin1-like protein families localize to the nucleus and bind to H3K4me3/2 via plant homeodomain fingers. Plant J 58 : 511–524.
21. MolitorAM, BuZ, YuY, ShenWH (2014) Arabidopsis AL PHD-PRC1 complexes promote seed germination through H3K4me3-to-H3K27me3 chromatin state switch in repression of seed developmental genes. PLoS Genet 10: e1004091.
22. JiangD, GuX, HeY (2009) Establishment of the winter-annual growth habit via FRIGIDA-mediated histone methylation at FLOWERING LOCUS C in Arabidopsis. Plant Cell 21 : 1733–1746.
23. HoppmannV, ThorstensenT, KristiansenPE, VeisethSV, RahmanMA, et al. (2011) The CW domain, a new histone recognition module in chromatin proteins. EMBO J 30 : 1939–1952.
24. HeF, UmeharaT, SaitoK, HaradaT, WatanabeS, et al. (2010) Structural insight into the zinc finger CW domain as a histone modification reader. Structure 18 : 1127–1139.
25. TurckF, RoudierF, FarronaS, Martin-MagnietteML, GuillaumeE, et al. (2007) Arabidopsis TFL2/LHP1 specifically associates with genes marked by trimethylation of histone H3 lysine 27. PLoS Genet 3: e86.
26. ZhangX, GermannS, BlusBJ, KhorasanizadehS, GaudinV, et al. (2007) The Arabidopsis LHP1 protein colocalizes with histone H3 Lys27 trimethylation. Nat Struct Mol Biol 14 : 869–871.
27. ZhangH, BishopB, RingenbergW, MuirWM, OgasJ (2012) The CHD3 remodeler PICKLE associates with genes enriched for trimethylation of histone H3 lysine 27. Plant Physiol 159 : 418–432.
28. HuY, LiuD, ZhongX, ZhangC, ZhangQ, et al. (2012) CHD3 protein recognizes and regulates methylated histone H3 lysines 4 and 27 over a subset of targets in the rice genome. Proc Natl Acad Sci U S A 109 : 5773–5778.
29. SunB, HongJ, ZhangP, DongX, ShenX, et al. (2008) Molecular Basis of the Interaction of Saccharomyces cerevisiae Eaf3 Chromo Domain with Methylated H3K36. Journal of Biological Chemistry 283 : 36504–36512.
30. ZhangP, DuJ, SunB, DongX, XuG, et al. (2006) Structure of human MRG15 chromo domain and its binding to Lys36-methylated histone H3. Nucleic Acids Research 34 : 6621–6628.
31. LarschanE, AlekseyenkoAA, GortchakovAA, PengS, LiB, et al. (2007) MSL Complex Is Attracted to Genes Marked by H3K36 Trimethylation Using a Sequence-Independent Mechanism. Molecular Cell 28 : 121–133.
32. EisenA, UtleyRT, NouraniA, AllardS, SchmidtP, et al. (2001) The yeast NuA4 and Drosophila MSL complexes contain homologous subunits important for transcription regulation. J Biol Chem 276 : 3484–3491.
33. ReidJL, MoqtaderiZ, StruhlK (2004) Eaf3 regulates the global pattern of histone acetylation in Saccharomyces cerevisiae. Mol Cell Biol 24 : 757–764.
34. GormanM, FrankeA, BakerBS (1995) Molecular characterization of the male-specific lethal-3 gene and investigations of the regulation of dosage compensation in Drosophila. Development 121 : 463–475.
35. BertramMJ, BerubeNG, Hang-SwansonX, RanQ, LeungJK, et al. (1999) Identification of a gene that reverses the immortal phenotype of a subset of cells and is a member of a novel family of transcription factor-like genes. Mol Cell Biol 19 : 1479–1485.
36. RobinsonSJ, TangLH, MooneyBA, McKaySJ, ClarkeWE, et al. (2009) An archived activation tagged population of Arabidopsis thaliana to facilitate forward genetics approaches. BMC Plant Biol 9 : 101.
37. Suarez-LopezP, WheatleyK, RobsonF, OnouchiH, ValverdeF, et al. (2001) CONSTANS mediates between the circadian clock and the control of flowering in Arabidopsis. Nature 410 : 1116–1120.
38. WiggePA, KimMC, JaegerKE, BuschW, SchmidM, et al. (2005) Integration of spatial and temporal information during floral induction in Arabidopsis. Science 309 : 1056–1059.
39. ZhaoZ, YuY, MeyerD, WuC, ShenW-H (2005) Prevention of early flowering by expression of FLOWERING LOCUS C requires methylation of histone H3 K36. Nature Cell Biology 7 : 1256–1260.
40. PienS, FleuryD, MylneJS, CrevillenP, InzeD, et al. (2008) ARABIDOPSIS TRITHORAX1 dynamically regulates FLOWERING LOCUS C activation via histone 3 lysine 4 trimethylation. Plant Cell 20 : 580–588.
41. SongYH, SmithRW, ToBJ, MillarAJ, ImaizumiT (2012) FKF1 Conveys Timing Information for CONSTANS Stabilization in Photoperiodic Flowering. Science 336 : 1045–1049.
42. ValverdeF (2004) Photoreceptor Regulation of CONSTANS Protein in Photoperiodic Flowering. Science 303 : 1003–1006.
43. VermeulenM, MulderKW, DenissovS, PijnappelWWMP, van SchaikFMA, et al. (2007) Selective Anchoring of TFIID to Nucleosomes by Trimethylation of Histone H3 Lysine 4. Cell 131 : 58–69.
44. CarrozzaMJ, LiB, FlorensL, SuganumaT, SwansonSK, et al. (2005) Histone H3 methylation by Set2 directs deacetylation of coding regions by Rpd3S to suppress spurious intragenic transcription. Cell 123 : 581–592.
45. PardoPS, LeungJK, LucchesiJC, Pereira-SmithOM (2002) MRG15, a novel chromodomain protein, is present in two distinct multiprotein complexes involved in transcriptional activation. J Biol Chem 277 : 50860–50866.
46. ZhangP, DuJ, SunB, DongX, XuG, et al. (2006) Structure of human MRG15 chromo domain and its binding to Lys36-methylated histone H3. Nucleic Acids Res 34 : 6621–6628.
47. LiD, LiuC, ShenL, WuY, ChenH, et al. (2008) A repressor complex governs the integration of flowering signals in Arabidopsis. Dev Cell 15 : 110–120.
48. ZuoZ, LiuH, LiuB, LiuX, LinC (2011) Blue light-dependent interaction of CRY2 with SPA1 regulates COP1 activity and floral initiation in Arabidopsis. Curr Biol 21 : 841–847.
49. YaoX, FengH, YuY, DongA, ShenW-H (2013) SDG2-Mediated H3K4 Methylation Is Required for Proper Arabidopsis Root Growth and Development. PLoS ONE 8: e56537.
50. ZuoJ, NiuQW, ChuaNH (2000) Technical advance: An estrogen receptor-based transactivator XVE mediates highly inducible gene expression in transgenic plants. Plant J 24 : 265–273.
51. LiZ, LiB, ShenW-H, HuangH, DongA (2012) TCP transcription factors interact with AS2 in the repression of class-I KNOX genes in Arabidopsis thaliana. The Plant Journal 71 : 99–107.
52. SunL, YangZT, SongZT, WangMJ, LuSJ, et al. (2013) The plant-specific transcription factor gene NAC103 is induced by bZIP60 through a new cis-regulatory element to modulate the unfolded protein response in Arabidopsis. Plant J 76 : 274–286.
53. LiuH, WangJY, HuangY, LiZ, GongW, et al. (2010) Structural basis for methylarginine-dependent recognition of Aubergine by Tudor. Genes Dev 24 : 1876–1881.
54. LeslieAG, PowellHR, WinterG, SvenssonO, SpruceD, et al. (2002) Automation of the collection and processing of X-ray diffraction data — a generic approach. Acta Crystallogr D Biol Crystallogr 58 : 1924–1928.
55. EmsleyP, LohkampB, ScottWG, CowtanK (2010) Features and development of Coot. Acta Crystallogr D Biol Crystallogr 66 : 486–501.
56. AdamsPD, AfoninePV, BunkocziG, ChenVB, DavisIW, et al. (2010) PHENIX: a comprehensive Python-based system for macromolecular structure solution. Acta Crystallogr D Biol Crystallogr 66 : 213–221.
Štítky
Genetika Reprodukční medicína
Článek An Evolutionarily Conserved Role for the Aryl Hydrocarbon Receptor in the Regulation of MovementČlánek Requirement for Drosophila SNMP1 for Rapid Activation and Termination of Pheromone-Induced ActivityČlánek Co-regulated Transcripts Associated to Cooperating eSNPs Define Bi-fan Motifs in Human Gene NetworksČlánek Identification of a Regulatory Variant That Binds FOXA1 and FOXA2 at the Type 2 Diabetes GWAS LocusČlánek tRNA Modifying Enzymes, NSUN2 and METTL1, Determine Sensitivity to 5-Fluorouracil in HeLa CellsČlánek Derlin-1 Regulates Mutant VCP-Linked Pathogenesis and Endoplasmic Reticulum Stress-Induced ApoptosisČlánek A Genetic Assay for Transcription Errors Reveals Multilayer Control of RNA Polymerase II FidelityČlánek The Proprotein Convertase KPC-1/Furin Controls Branching and Self-avoidance of Sensory Dendrites inČlánek Regulation of p53 and Rb Links the Alternative NF-κB Pathway to EZH2 Expression and Cell SenescenceČlánek BMPs Regulate Gene Expression in the Dorsal Neuroectoderm of and Vertebrates by Distinct MechanismsČlánek Unkempt Is Negatively Regulated by mTOR and Uncouples Neuronal Differentiation from Growth Control
Článek vyšel v časopisePLOS Genetics
Nejčtenější tento týden
2014 Číslo 9
-
Všechny články tohoto čísla
- Translational Regulation of the Post-Translational Circadian Mechanism
- An Evolutionarily Conserved Role for the Aryl Hydrocarbon Receptor in the Regulation of Movement
- Eliminating Both Canonical and Short-Patch Mismatch Repair in Suggests a New Meiotic Recombination Model
- Requirement for Drosophila SNMP1 for Rapid Activation and Termination of Pheromone-Induced Activity
- Co-regulated Transcripts Associated to Cooperating eSNPs Define Bi-fan Motifs in Human Gene Networks
- Targeted H3R26 Deimination Specifically Facilitates Estrogen Receptor Binding by Modifying Nucleosome Structure
- Role for Circadian Clock Genes in Seasonal Timing: Testing the Bünning Hypothesis
- The Tandem Repeats Enabling Reversible Switching between the Two Phases of β-Lactamase Substrate Spectrum
- The Association of the Vanin-1 N131S Variant with Blood Pressure Is Mediated by Endoplasmic Reticulum-Associated Degradation and Loss of Function
- Identification of a Regulatory Variant That Binds FOXA1 and FOXA2 at the Type 2 Diabetes GWAS Locus
- Regulation of Flowering by the Histone Mark Readers MRG1/2 via Interaction with CONSTANS to Modulate Expression
- The Actomyosin Machinery Is Required for Retinal Lumen Formation
- Plays a Conserved Role in Assembly of the Ciliary Motile Apparatus
- Hidden Diversity in Honey Bee Gut Symbionts Detected by Single-Cell Genomics
- Ribosome Rescue and Translation Termination at Non-Standard Stop Codons by ICT1 in Mammalian Mitochondria
- tRNA Modifying Enzymes, NSUN2 and METTL1, Determine Sensitivity to 5-Fluorouracil in HeLa Cells
- Causal Variation in Yeast Sporulation Tends to Reside in a Pathway Bottleneck
- Tissue-Specific RNA Expression Marks Distant-Acting Developmental Enhancers
- WC-1 Recruits SWI/SNF to Remodel and Initiate a Circadian Cycle
- Clonal Expansion of Early to Mid-Life Mitochondrial DNA Point Mutations Drives Mitochondrial Dysfunction during Human Ageing
- Methylation QTLs Are Associated with Coordinated Changes in Transcription Factor Binding, Histone Modifications, and Gene Expression Levels
- Differential Management of the Replication Terminus Regions of the Two Chromosomes during Cell Division
- Obesity-Linked Homologues and Establish Meal Frequency in
- Derlin-1 Regulates Mutant VCP-Linked Pathogenesis and Endoplasmic Reticulum Stress-Induced Apoptosis
- Stress-Induced Nuclear RNA Degradation Pathways Regulate Yeast Bromodomain Factor 2 to Promote Cell Survival
- The MAPK p38c Regulates Oxidative Stress and Lipid Homeostasis in the Intestine
- Widespread Genome Reorganization of an Obligate Virus Mutualist
- Trans-kingdom Cross-Talk: Small RNAs on the Move
- The Vip1 Inositol Polyphosphate Kinase Family Regulates Polarized Growth and Modulates the Microtubule Cytoskeleton in Fungi
- Myosin Vb Mediated Plasma Membrane Homeostasis Regulates Peridermal Cell Size and Maintains Tissue Homeostasis in the Zebrafish Epidermis
- GLD-4-Mediated Translational Activation Regulates the Size of the Proliferative Germ Cell Pool in the Adult Germ Line
- Genome Wide Association Studies Using a New Nonparametric Model Reveal the Genetic Architecture of 17 Agronomic Traits in an Enlarged Maize Association Panel
- Translational Regulation of the DOUBLETIME/CKIδ/ε Kinase by LARK Contributes to Circadian Period Modulation
- Positive Selection and Multiple Losses of the LINE-1-Derived Gene in Mammals Suggest a Dual Role in Genome Defense and Pluripotency
- Out of Balance: R-loops in Human Disease
- A Genetic Assay for Transcription Errors Reveals Multilayer Control of RNA Polymerase II Fidelity
- Altered Behavioral Performance and Live Imaging of Circuit-Specific Neural Deficiencies in a Zebrafish Model for Psychomotor Retardation
- Nipbl and Mediator Cooperatively Regulate Gene Expression to Control Limb Development
- Meta-analysis of Mutations in Autism Spectrum Disorders: A Gradient of Severity in Cognitive Impairments
- The Proprotein Convertase KPC-1/Furin Controls Branching and Self-avoidance of Sensory Dendrites in
- Hydroxymethylated Cytosines Are Associated with Elevated C to G Transversion Rates
- Memory and Fitness Optimization of Bacteria under Fluctuating Environments
- Regulation of p53 and Rb Links the Alternative NF-κB Pathway to EZH2 Expression and Cell Senescence
- Interspecific Tests of Allelism Reveal the Evolutionary Timing and Pattern of Accumulation of Reproductive Isolation Mutations
- PRO40 Is a Scaffold Protein of the Cell Wall Integrity Pathway, Linking the MAP Kinase Module to the Upstream Activator Protein Kinase C
- Low Levels of p53 Protein and Chromatin Silencing of p53 Target Genes Repress Apoptosis in Endocycling Cells
- SPDEF Inhibits Prostate Carcinogenesis by Disrupting a Positive Feedback Loop in Regulation of the Foxm1 Oncogene
- RRP6L1 and RRP6L2 Function in Silencing Regulation of Antisense RNA Synthesis
- BMPs Regulate Gene Expression in the Dorsal Neuroectoderm of and Vertebrates by Distinct Mechanisms
- Unkempt Is Negatively Regulated by mTOR and Uncouples Neuronal Differentiation from Growth Control
- Atkinesin-13A Modulates Cell-Wall Synthesis and Cell Expansion in via the THESEUS1 Pathway
- Dopamine Signaling Leads to Loss of Polycomb Repression and Aberrant Gene Activation in Experimental Parkinsonism
- Histone Methyltransferase MMSET/NSD2 Alters EZH2 Binding and Reprograms the Myeloma Epigenome through Global and Focal Changes in H3K36 and H3K27 Methylation
- Bipartite Recognition of DNA by TCF/Pangolin Is Remarkably Flexible and Contributes to Transcriptional Responsiveness and Tissue Specificity of Wingless Signaling
- The Olfactory Transcriptomes of Mice
- Muscular Dystrophy-Associated and Variants Disrupt Nuclear-Cytoskeletal Connections and Myonuclear Organization
- Interplay of dFOXO and Two ETS-Family Transcription Factors Determines Lifespan in
- Evidence for Widespread Positive and Negative Selection in Coding and Conserved Noncoding Regions of
- Genome-Wide Association Meta-analysis of Neuropathologic Features of Alzheimer's Disease and Related Dementias
- Rejuvenation of Meiotic Cohesion in Oocytes during Prophase I Is Required for Chiasma Maintenance and Accurate Chromosome Segregation
- Admixture in Latin America: Geographic Structure, Phenotypic Diversity and Self-Perception of Ancestry Based on 7,342 Individuals
- Local Effect of Enhancer of Zeste-Like Reveals Cooperation of Epigenetic and -Acting Determinants for Zygotic Genome Rearrangements
- Differential Responses to Wnt and PCP Disruption Predict Expression and Developmental Function of Conserved and Novel Genes in a Cnidarian
- PLOS Genetics
- Archiv čísel
- Aktuální číslo
- Informace o časopisu
Nejčtenější v tomto čísle- Admixture in Latin America: Geographic Structure, Phenotypic Diversity and Self-Perception of Ancestry Based on 7,342 Individuals
- Nipbl and Mediator Cooperatively Regulate Gene Expression to Control Limb Development
- Genome Wide Association Studies Using a New Nonparametric Model Reveal the Genetic Architecture of 17 Agronomic Traits in an Enlarged Maize Association Panel
- Histone Methyltransferase MMSET/NSD2 Alters EZH2 Binding and Reprograms the Myeloma Epigenome through Global and Focal Changes in H3K36 and H3K27 Methylation
Kurzy
Zvyšte si kvalifikaci online z pohodlí domova
Současné možnosti léčby obezity
nový kurzAutoři: MUDr. Martin Hrubý
Všechny kurzyPřihlášení#ADS_BOTTOM_SCRIPTS#Zapomenuté hesloZadejte e-mailovou adresu, se kterou jste vytvářel(a) účet, budou Vám na ni zaslány informace k nastavení nového hesla.
- Vzdělávání



