-
Články
Top novinky
Reklama- Vzdělávání
- Časopisy
Top články
Nové číslo
- Témata
Top novinky
Reklama- Videa
- Podcasty
Nové podcasty
Reklama- Kariéra
Doporučené pozice
Reklama- Praxe
Top novinky
ReklamaEffective Blocking of the Enhancer Requires Cooperation between Two Main Mechanisms Suggested for the Insulator Function
Chromatin insulators block the action of transcriptional enhancers when interposed between an enhancer and a promoter. In this study, we examined the role of chromatin loops formed by two unrelated insulators, gypsy and Fab-7, in their enhancer-blocking activity. To test for this activity, we selected the white reporter gene that is activated by the eye-specific enhancer. The results showed that one copy of the gypsy or Fab-7 insulator failed to block the eye enhancer in most of genomic sites, whereas a chromatin loop formed by two gypsy insulators flanking either the eye enhancer or the reporter completely blocked white stimulation by the enhancer. However, strong enhancer blocking was achieved due not only to chromatin loop formation but also to the direct interaction of the gypsy insulator with the eye enhancer, which was confirmed by the 3C assay. In particular, it was observed that Mod(mdg4)-67.2, a component of the gypsy insulator, interacted with the Zeste protein, which is critical for the eye enhancer–white promoter communication. These results suggest that efficient enhancer blocking depends on the combination of two factors: chromatin loop formation by paired insulators, which generates physical constraints for enhancer–promoter communication, and the direct interaction of proteins recruited to an insulator and to the enhancer–promoter pair.
Published in the journal: . PLoS Genet 9(7): e32767. doi:10.1371/journal.pgen.1003606
Category: Research Article
doi: https://doi.org/10.1371/journal.pgen.1003606Summary
Chromatin insulators block the action of transcriptional enhancers when interposed between an enhancer and a promoter. In this study, we examined the role of chromatin loops formed by two unrelated insulators, gypsy and Fab-7, in their enhancer-blocking activity. To test for this activity, we selected the white reporter gene that is activated by the eye-specific enhancer. The results showed that one copy of the gypsy or Fab-7 insulator failed to block the eye enhancer in most of genomic sites, whereas a chromatin loop formed by two gypsy insulators flanking either the eye enhancer or the reporter completely blocked white stimulation by the enhancer. However, strong enhancer blocking was achieved due not only to chromatin loop formation but also to the direct interaction of the gypsy insulator with the eye enhancer, which was confirmed by the 3C assay. In particular, it was observed that Mod(mdg4)-67.2, a component of the gypsy insulator, interacted with the Zeste protein, which is critical for the eye enhancer–white promoter communication. These results suggest that efficient enhancer blocking depends on the combination of two factors: chromatin loop formation by paired insulators, which generates physical constraints for enhancer–promoter communication, and the direct interaction of proteins recruited to an insulator and to the enhancer–promoter pair.
Introduction
The complexity of regulatory systems in higher eukaryotes, featuring many distantly located enhancers that nonetheless properly activate the target, has promoted the hypothesis that the action of enhancers should be restricted by elements called insulators. Initially, insulators were regarded as genomic regulatory elements (nucleoprotein complexes) that have two characteristic properties: they can block the action of an enhancer on a promoter when interposed between them and can protect the transgenes they flank from chromosomal position effects (for reviews, see [1]–[7]). However, recent results of studies on insulators in transgenic Drosophila lines [8]–[12], genome-wide identification of biding sites for insulator proteins by ChIP-on-ChIP and ChIP-seq [13]–[17], analysis of locus architecture by different variants of chromosome conformation capture technology [18]–[19], and genome-wide analysis of interaction between CTCF sites by paired-end tag (PET) approach, ChIA-PET [20], and Hi-C technique [21], [22] suggest that insulators are mainly involved in organization of long-distance specific interactions between remote genome regions such as enhancers and promoters, different promoters, or multiple regulatory elements.
Well-characterized insulators in Drosophila include the scs and scs' sequences from the 87A heat shock locus [23], [24]; the Mcp, Fab-7 and Fab-8 insulators from the Abd-B regulatory region [25]–[29]; the SF1 insulator from the Antennapedia complex [30]; the IdefixU3 insulator [31]; the Wari insulator located at the 3′ side of the white gene [10]; and the insulator sequences associated with the Su(Hw) protein [32]–[36]. Today, there are two basic models explaining how insulators block the activity of enhancers [1], [2], [4], [6], [37]. The decoy model suggests that the insulator complex binds to an enhancer or a promoter complex to neutralize it or traps its vital component(s). The alternative model suggests that the interaction between insulators results in the formation of chromatin loops that constrain interaction between an enhancer and a promoter located on the opposite sides of the insulator. The latter model is indirectly supported by the ability of the insulators to specifically interact over large distances [11], [12], [20]–[22], [38]–[40]. However, there are only a few pieces of indirect experimental evidence supporting the model that a loop formed by interacting insulators is essential for enhancer blocking [41]–[46].
Bondarenko et al. (2003) used a bacterial enhancer–promoter pair and a pair of lac operators (lacO) that mimicked eukaryotic insulators [42], [43]. It was shown in an in vitro transcription assay that the enhancer action was blocked when the interacting lacO copies formed two closed loops, one with the enhancer and the other with the promoter. This finding suggests that if DNA looping alone is sufficient to suppress the enhancer activity in an in vitro model system, it may as well play an important role in eukaryotic cells. Ameres et al. (2005) examined the expression of a reporter gene in HeLa cells transfected with the plasmid in which the SV40 enhancer was placed downstream of the reporter gene [41]. The SV40 enhancer was flanked by two boxes, each consisting of seven repeats of the tetR element. When the chimeric protein consisting of the tetR protein and a dimerization domain bound to tetR elements, the dimerized proteins formed a 344-bp chromatin loop containing the SV40 enhancer. As a result, this enhancer was blocked, with consequent reduction of the reporter gene expression, which suggested a role for the loop in preventing the interaction between the SV40 enhancer and the promoter. However, alternative mechanisms of SV40 enhancer blocking cannot be excluded. For example, the small chromatin loop may interfere with proper binding of transcription factors to the enhancer. In the study by Hou et al. (2008), a CTCF-dependent insulator ectopically inserted between the beta-globin locus control region (LCR) and downstream genes was found to function as an enhancer blocker and form an aberrant loop with the endogenous CTCF region located upstream of the LCR [46]. However, these authors did not perform experiments to show that the ectopic insulator could not block LCR in the absence of the loop formation with the endogenous insulator and, therefore, failed to obtain direct evidence for the role of chromatin loop formation in enhancer blocking.
For this reason, we established a Drosophila transgenic model system in order to test whether isolation of either an enhancer or the reporter white gene in a loop formed by gypsy or Fab-7 insulators can block the enhancer–promoter communication.
The gypsy insulator, the strongest and the best studied in Drosophila, contains 12 consecutive degenerate direct repeats of the binding motif for the zinc-finger protein Su(Hw), which is indispensable for the insulator function [32], [33]. The Su(Hw) protein also associates with hundreds of non-gypsy regions that do not contain clustered Su(Hw)-binding sites, with the vast majority of them carrying a single copy of the corresponding sequence [13], [14], [36]. Su(Hw) interacts with three other components of the gypsy insulator, Mod(mdg4)-67.2 [47], [48], CP190 [49], and E(y)2 [50]. Based on the results of genetic interactions, it has been suggested that Mod(mdg4)-67.2 and CP190 are essential for the enhancer-blocking activity of Su(Hw) insulators. Mod(mdg4)-67.2 interacts with Su(Hw) through its carboxy-terminal domain [47], [48], [51]. The BTB domain is located at the N-terminus of Mod(mdg4)-67.2 and mediates homo-multimerization [52]. There are many evidences of functional distant interactions between the gypsy insulators [9], [11], [53], which were recently confirmed by the 3C method [54].
Among insulators found in the regulatory region of Abd-B, the best characterized is Fab-7 located between the iab-6 and iab-7 cis-regulatory domains [55]. Mutations that inactivate Fab-7 lead to the fusion of the iab-6 and iab-7 domains, and this disrupts the specification of PS11 [56]–[58]. Previously we found that interaction between paired two copies of the Fab-7 insulator can support long-distance enhancer-promoter interactions [59]. It was also found that Fab-7 insulators, similar to gypsy insulators [11], can support interactions across several megabases [12].
In previous studies [9], [28], [33], [45], [59], [60]–[62], the activities of gypsy and Fab-7 insulators were mainly tested in transgenic lines with the yellow and white genes as reporters that allowed changes in gene expression to be assayed by simple phenotypic analysis. It was found that one copy of the gypsy insulator completely blocked the communication between the yellow enhancers and the promoter [33], [60], [62]. Genetic studies on transgenic lines carrying different mutations in the yellow promoter suggested that the gypsy insulator directly interacted with the yellow promoter [63]. Placing the yellow enhancers in a 10-kb chromatin loop formed by the gypsy insulators led to neutralization of enhancer blocking [62], providing evidence against the role of a chromatin loop formed by the insulators in enhancer blocking.
Therefore, we used the white model system to test the role of the chromatin loop in the enhancer-blocking activity of the gypsy and Fab-7 insulators. Recently we found that the white gene contains the insulator, named Wari, located downstream of the polyadenylation signal [10]. This insulator can interact equally well with another copy of Wari and with unrelated Su(Hw) insulators [9], [10]. It was shown that the interaction between the Wari and gypsy insulators strongly improves enhancer blocking. To test whether a single copy of gypsy or Fab-7 insulator can block the eye enhancer, we deleted the Wari insulator from the white gene.
We found that one copy of the gypsy or Fab-7 insulator failed to affect the eye enhancer activity in most of the transgenic lines tested. At the same time, the insertion of two gypsy insulators on both sides of the eye enhancer or the white gene completely blocked the enhancer–promoter communication in all transgenic lines. In contrast, flanking the eye enhancer by Fab-7 insulators only slightly contributed to the blocking of enhancer–promoter communication. Such a difference in the ability to block the eye enhancer between the pairs of gypsy and Fab-7 insulators is explained by the finding that the gypsy insulator can directly interact with the enhancer. In particular, Mod(mdg4)-67.2 interacts with Zeste and can interfere with its activity in supporting enhancer–promoter communication at the white gene.
Results
The gypsy insulator fails to block the eye enhancer in more than 70% of transgenic lines
To establish a model system for testing the role of a chromatin loop in insulation, we used the white reporter gene that is stimulated by a tissue-specific enhancer in the eyes. The level of eye pigmentation is a sensitive indicator of the amount of white transcription. To test the enhancer-blocking activity of one copy of the gypsy insulator in different genomic positions, we deleted the Wari insulator from the white gene (WΔ). The gypsy insulator flanked by lox sites was inserted between the eye enhancer flanked by frt sites and the white gene (Figure 1A). Parentheses in construct designations and short downward arrows in the schemes indicate the elements flanked by lox or frt sites for in vivo excision by crossing, as outlined in Materials and Methods. Such excisions are denoted by “Δ” in the primary (expression) data. Comparing eye phenotypes in the transgenic lines before and after deletion of either the eye enhancer or the gypsy insulator allowed estimation of their contribution to white expression. Since it was shown [64] that the eye enhancer can initiate transcription in the direct orientation, we inserted the eye enhancer in either direct (Ee) or reverse orientation (EeR). In Figure 1, we combined the results obtained with transgenic lines carrying both constructs, because they displayed a similar range of phenotypes.
Fig. 1. Testing the enhancer-blocking activity of the gypsy insulator in one copy. 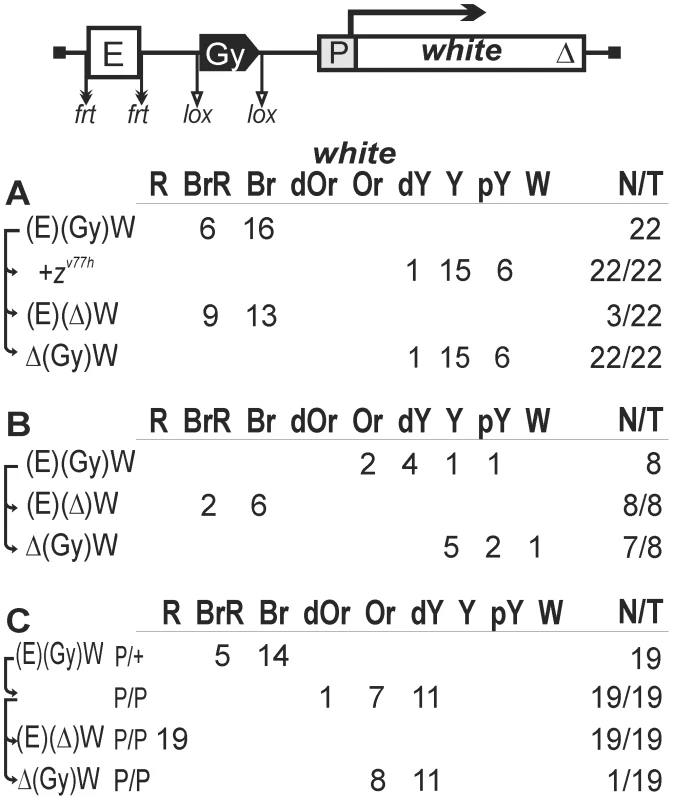
Transgenic lines were grouped into those in which the gypsy insulator displayed (A) weak or (B) strong enhancer-blocking activity. (C) Homozygous transgenic lines in which the gypsy insulator displayed a weak enhancer-blocking activity. In the reductive scheme of the transgenic construct used in the assay, the white gene is shown as white box with an arrow indicating the direction of transcription; the triangle indicates deletion of the Wari insulator located at the 3′ end of the white gene; downward arrows indicate target sites for Flp recombinase (frt) or Cre recombinase (lox); the same sites in construct names are denoted by parentheses; the eye enhancer (E) is shown as white rectangle; the direction of the gypsy insulator (Gy) is indicated by the apex of the pentagon. The numbers of transgenic lines with different levels of white pigmentation in the eyes are indicated. Arrows indicate the excision of an element to produce the derivative transgenic lines. Wild-type white expression determined the bright red eye color (R); in the absence of white expression, the eyes were white (W). Intermediate levels of pigmentation, with the eye color ranging from pale yellow (pY), through yellow (Y), dark yellow (dY), orange (Or), dark orange (dOr), and brown (Br) to brownish red (BrR), reflect the increasing levels of white expression. N is the number of lines in which flies acquired a new eye color phenotype by deletion (Δ) of the specified DNA fragment; T is the total number of lines examined for each particular construct. zv77h, a null-mutation of the zeste gene. In 22 out of 34 transgenic lines, males heterozygous for the construct had high levels of eye pigmentation (from brown to brown-red) that decreased significantly after deletion of the eye enhancer (Figures 1A, 2A). These results suggest that the eye enhancer can stimulate white expression across the gypsy insulator. Moreover, deletion of the gypsy insulator slightly changed eye pigmentation in only 3 out of 22 tested lines, indicating that gypsy failed to block the eye enhancer. To test for the functional role of Zeste in white stimulation by the eye enhancer, we crossed the transgenic lines into the background of zv77h, a null mutation of the zeste gene [65]. As a result, we observed that zv77h strongly reduced eye pigmentation to the same level as did the deletion of the eye enhancer (Figure 1A), indicating that Zeste is critical for the eye enhancer activity. After deletion of the gypsy insulator, zv77h still reduced white expression in transgenic lines (Figure S1A). However, zv77h did not influence white phenotypes in transgenic lines carrying derivative constructs with the deleted eye enhancer. These results suggest that Zeste is essential for the eye enhancer activity even when it is located close to the white promoter.
Fig. 2. Eye phenotypes in flies from the transgenic line with the gypsy insulator in one copy. 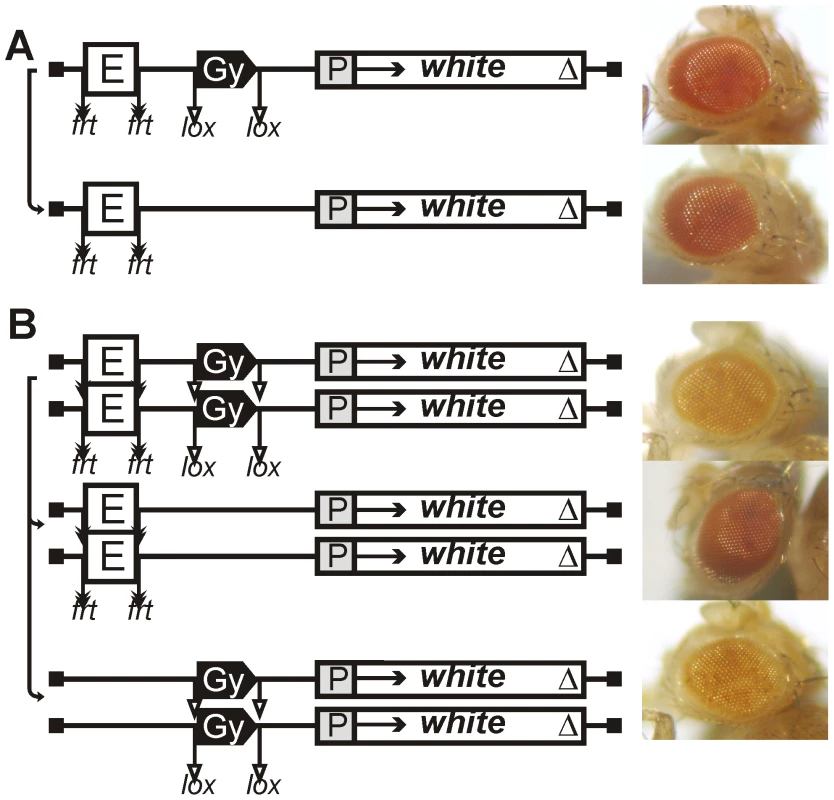
(A) heterozygous (P/+) or (B) homozygous (P/P) for the construct and in flies after deletion of either the eye enhancer or the gypsy insulator. Other designations are as in Figure 1. Four transgenic lines had yellow eyes that did not change in color after deletion of the gypsy insulator, indicating that the eye enhancer was inactive in these lines (data not shown). In the remaining eight lines (Figure 1B), the deletion of gypsy led to change in eye color from yellow to brown, suggesting that this insulator could effectively block the enhancer–promoter communication. Thus, the gypsy insulator proved to block the eye enhancer in only 8 out of 30 transgenic lines (27%).
Next, we examined eye color in flies of 19 homozygous transgenic lines in which the gypsy insulator failed to block the eye enhancer (Figure 1C). Unexpectedly, we found that flies carrying the homozygous transgene had lighter eyes than flies with the heterozygous transgene (Figures 1C, 2B). The deletion of the eye enhancer did not change eye pigmentation in lines homozygous for the transgene, indicating that the gypsy insulator completely blocked the eye enhancer in these lines.
Taken together, these results suggest that one copy of the gypsy insulator failed to block the eye enhancer–white promoter communication in more than 70% of the transgenic lines. However, pairing between the gypsy insulators located on homologous chromosomes restricted the eye enhancer activity.
Fab-7 has a weak enhancer-blocking activity in most genomic sites of transgene insertion
As a second model insulator, we selected the well-described Fab-7 insulator that supports specific long-distance interactions [12]. As shown in previous experiments with transgenic lines carrying constructs with the 1.2-kb Fab-7 insulator inserted between the eye enhancer and the white promoter, flies in approximately half of these lines had relatively light eyes indicating effective blocking of the eye enhancer by the Fab-7 insulator [25], [61]. However, it was not proved that the eye enhancer was functional in these lines, since the Wari insulator located at the 3′ end of the white gene could also improve the activity of Fab-7.
For these reasons, we again tested the enhancer-blocking ability of one Fab-7 copy in the construct where it was flanked by lox sites and inserted between the eye enhancer (flanked by frt sites) and the white gene (Figure 3). In 14 transgenic lines, flies had extensive eye pigmentation ranging from dark orange to brown (Figure 3A). Deletion of the Fab-7 insulator resulted in a slight enhancement of pigmentation in 9 out of 14 transgenic lines, suggesting that Fab-7 functioned as a weak enhancer blocker in these transgenic lines. As in the case of transgenic lines carrying the gypsy insulator, the deletion of the eye enhancer and crossing into the zv77h mutant background reduced eye pigmentation in flies to the same extent (Figure 3A, S1B), suggesting a key role for Zeste in the eye enhancer activity and its ability to bypass the Fab-7 insulator.
Fig. 3. The enhancer-blocking activity of the Fab-7 insulator in one copy. 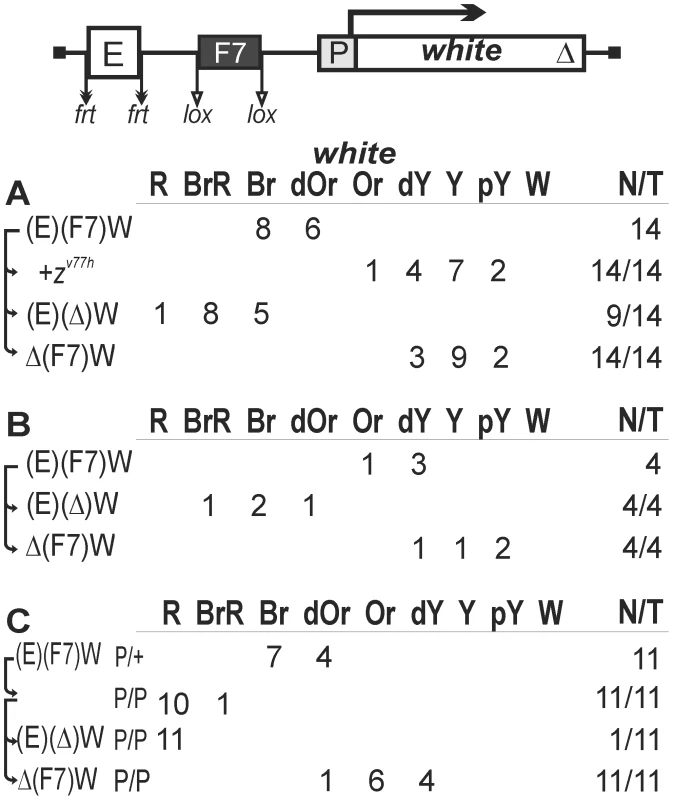
Transgenic lines were grouped into those in which the Fab-7 insulator (F7, black rectangle) displayed (A) weak or (B) strong enhancer-blocking activity. (C) Homozygous transgenic lines in which the Fab-7 insulator displayed a weak enhancer-blocking activity. Other designations are as in Figure 1. Flies of the remaining 10 transgenic lines had yellow eyes. When the Fab-7 insulator was deleted, eye pigmentation was restored in only four lines (Figure 3B), suggesting that the eye enhancer in other six lines was inactive (data not shown). Thus, the Fab-7 insulator could effectively block the eye enhancer in only 4 out of 18 transgenic lines (22%) carrying the transgene with the functional eye enhancer.
We then examined eye pigmentation in transgenic lines homozygous for the construct (Figure 3C) in which the Fab-7 insulator displayed a weak enhancer-blocking activity. In all these lines, flies had darker eye color, compared to the lines heterozygous for the construct, suggesting that the Fab-7 insulator failed to effectively block the eye enhancer when the construct was in the homozygous state.
Taken together, these results indicate that the Fab-7 insulator can only weakly affect the activity of the eye enhancer in most of genomic positions. Both Fab-7 and gypsy insulators can effectively block the eye enhancer in approximately one-fourth of transgenic lines. In contrast to the gypsy insulator, the pairing of two Fab-7 insulators located on homologous chromosomes failed to improve the eye enhancer blocking.
Role of pairing between two identical insulators, gypsy or Fab-7, flanking the eye enhancer in blocking its activity
Since one copy of the gypsy or Fab-7 insulator in most insertion sites of the transgenes failed to block the eye enhancer, we decided to test if the eye enhancer placed between two insulators would improve the efficiency of blocking. For this purpose, we made constructs where the eye enhancer was flanked by a pair of gypsy insulators (Figure 4A) or Fab-7 insulators (Figure 4B) inserted in opposite orientations. In both cases, the insulator located upstream of the eye enhancer was flanked by lox sites, which allowed us to assess its role in blocking the eye enhancer.
Fig. 4. Testing functional interactions between gypsy and Fab-7 insulators. 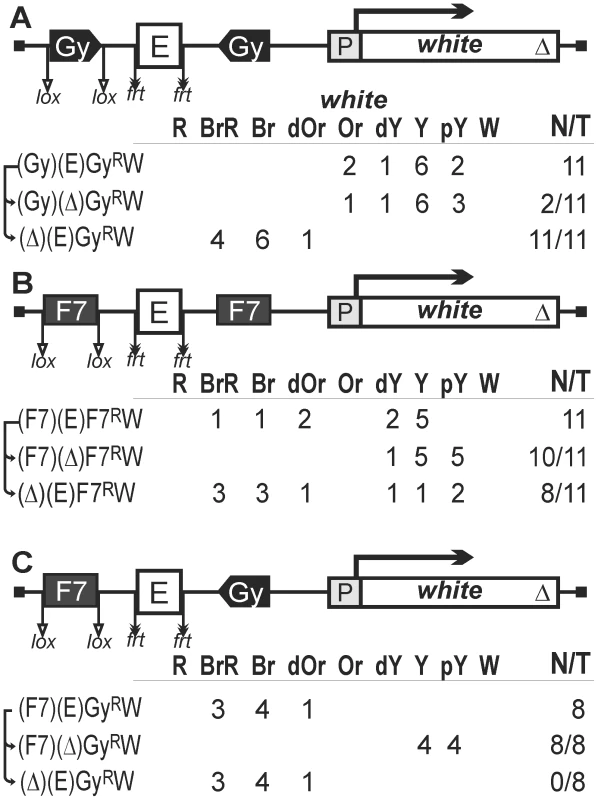
(A) Two gypsy insulators flank the eye enhancer. (B) Two Fab-7 insulators flank the eye enhancer. (C) One Fab-7 and one gypsy insulators flank the eye enhancer. Superscript “R” indicates that the corresponding element is inserted in the reverse orientation relative to the white gene in the construct. Other designations are as in Figure 1. In all 11 transgenic lines carrying the construct with two gypsy insulators (Figure 4A), flies had eye pigmentation in the range from pale yellow to orange, indicating that the eye enhancer activity was strongly suppressed. Deletion of the eye enhancer resulted in a slight reduction of pigmentation in only 2 out of 11 lines, providing evidence that the eye enhancer was strongly blocked in all these lines. Deletion of the upstream insulator restored eye pigmentation in 10 transgenic lines (Figure 4A), while subsequent deletion of the eye enhancer reduced it to the initial level (data not shown). These results showed that two copies of the gypsy insulator flanking the eye enhancer completely blocked its activity.
In transgenic lines carrying the construct with two Fab-7 insulators (Figure 4B), we observed a wide range of eye phenotypes, from brown-red to yellow. Deletion of the eye enhancer resulted in reduction of pigmentation in 10 out of 11 transgenic lines, showing that two Fab-7 insulators failed to effectively block the eye enhancer. However, the deletion of the upstream insulator provided for slight intensification of eye pigmentation in 8 out of 11 transgenic lines, suggesting that the interaction between the Fab-7 insulators could contribute to enhancer blocking.
To test whether two different insulators can cooperate in blocking the eye enhancer, we made the construct that contained one Fab-7 insulator flanked by lox sites inserted upstream of the eye enhancer and one gypsy insulator placed between the eye enhancer flanked by frt sites and the promoter (Figure 4C). We obtained eight transgenic lines that displayed high levels of eye pigmentation. Deletion of the eye enhancer strongly reduced eye pigmentation, while deletion of the Fab-7 insulator did not have any effect on eye color in any of the lines tested. This is evidence that the Fab-7 and gypsy insulators do not functionally interact in blocking the eye enhancer.
Enhancer-blocking activity depends on the position of the eye enhancer in the insulator loop and on the relative orientation of interacting gypsy insulators
In transgenic lines described in Figure 4, the eye enhancer was tightly flanked by the gypsy insulators. Therefore, the putative insulator loop was probably quite small, and chromatin could be wound into a “tight knot” with consequent conformational and/or steric hindrances to the enhancer function. To test whether an increase in the distance between the gypsy insulators flanking the eye enhancer can restore enhancer–promoter communication at the white gene, we inserted the frt-flanked eye enhancer in the center of a 4.3-kb fragment bordered by the gypsy insulators in either the opposite (Figure 5A) or the same orientation (Figure 5B). The upstream gypsy insulator was flanked by lox sites.
Fig. 5. The role of gypsy orientation and distance between gypsy insulators in blocking the eye enhancer. 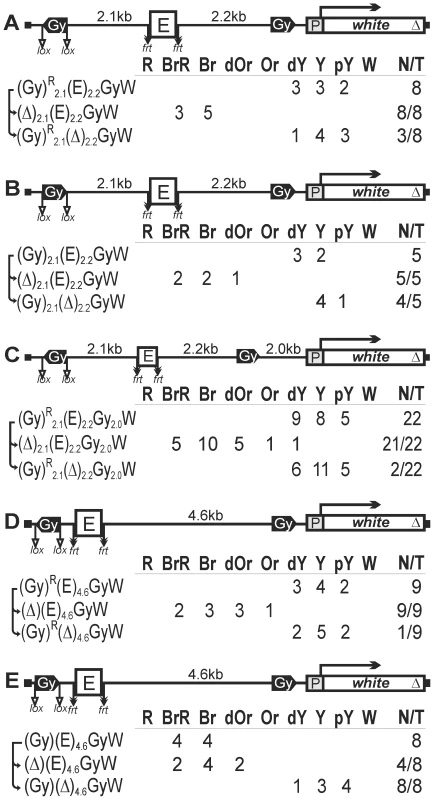
The eye enhancer is inserted in the center of a 4.3-kb domain bordered by the gypsy insulators located (A) in the opposite or (B) in the same orientation. (C) Insertion of additional 2-kb DNA fragment between the 4.3-kb domain bordered by the gypsy insulators and the white promoter. (D, E) The eye enhancer is inserted in a 5.5-kb loop formed by the gypsy insulators located (D) in the opposite or (E) in the same orientation. Designations are as in Figures 1 and 4. In both series of transgenic lines, flies had eye pigmentation in a dark-yellow to pale-yellow range. The deletion of the eye enhancer slightly reduced eye pigmentation in 7 out of 13 transgenic lines. The deletion of the upstream gypsy insulator restored eye pigmentation in all 13 transgenic lines, indicating that one copy of the gypsy insulator failed to block the eye enhancer. These results showed that a 5.2-kb loop (4.3-kb DNA fragment and 0.9-kb frt-flanked eye enhancer) formed by the gypsy insulators allowed blocking of the eye enhancer located in the center of the loop. Importantly, the enhancer blocking did not depend on the relative orientation of the gypsy insulators.
In all the above constructs, the white promoter was located in close proximity to one of the insulators that formed the chromatin loop around the eye enhancer. In such configuration of the regulatory elements, the promoter might be unable to interact with the eye enhancer due to steric hindrances. To test for the role of distance between the white promoter and the insulator loop in this process, we modified the construct shown in Figure 5A by inserting an additional 2-kb DNA fragment between the gypsy insulator and the white promoter (Figure 5C). Once again, the interacting gypsy insulators effectively blocked the eye enhancer in most of transgenic lines, while one copy of the insulator had no enhancer-blocking activity.
In the next series of experiments, we tried to test whether the relative orientation of the gypsy insulators is important for the enhancer blocking when the eye enhancer is located in close proximity to the insulator inside the loop. For this purpose, we inserted the frt-flanked eye enhancer in close proximity to the upstream gypsy insulator. As a result, the eye enhancer was inside the 5.5-kb loop formed by the insulators located in either the opposite (Figure 5D) or the same orientation (Figure 5E).
In lines carrying the transgene with gypsy insulators in opposite orientations, flies had eye pigmentation ranging from dark yellow to pale yellow, which remained unchanged after the deletion of the eye enhancer. Thus, in such configuration of regulatory elements, gypsy insulators completely blocked the eye enhancer. The deletion of the proximal gypsy insulator restored the enhancer activity, suggesting that the interaction between the insulators was critical for blocking the eye enhancer. Interestingly, when the insulators were in the same orientation, flies had brown eyes and the deletion of the enhancer strongly reduced eye pigmentation, which was indicative of a role for the enhancer in stimulating of the white expression. The deletion of the proximal insulator also slightly reduced eye pigmentation in half of the transgenic lines. Thus, the gypsy insulators located in the same orientation allow the interaction between the eye enhancer located within the loop and the white promoter located outside the loop.
Addition of the gypsy insulator downstream of the white gene improves the enhancer-blocking activity of the upstream insulator and potentiates promoter activity
Next, we tested if placing gypsy insulators on both sides of the white gene would also lead to the improvement of enhancer-blocking activity. In two constructs, one frt-flanked gypsy insulator was inserted downstream of the white gene, and the other, lox-flanked gypsy insulator was inserted between the eye enhancer and the promoter in either the opposite (Figure 6A) or the same orientation (Figure 6B). As a result, the eye enhancer was located upstream of the chromatin domain formed by two gypsy insulators bordering the white gene.
Fig. 6. Testing the activities of gypsy insulators flanking the white gene. 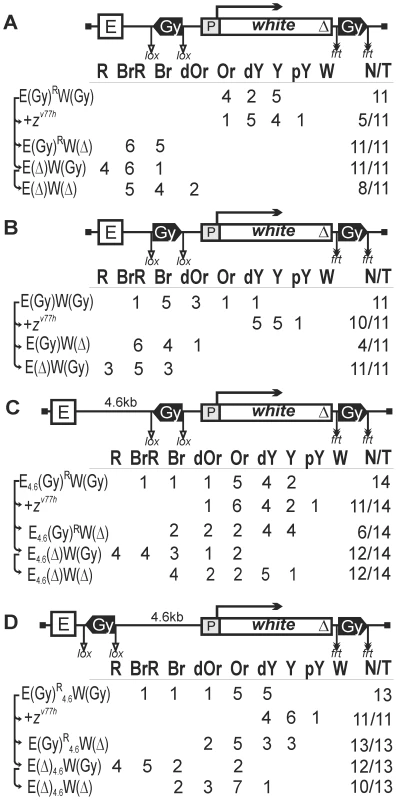
The gypsy insulators flanking the white gene are inserted either (A) in the opposite or (B) in the same orientation. (C) Insertion of the 4.6-kb DNA fragment between the eye enhancer and the white gene domain flanked by the gypsy insulators. (D) Insertion of the 4.6-kb DNA fragment between the gypsy insulator and the white promoter. Designations are as in Figures 1 and 4. When the gypsy insulators were in opposite orientations, the activity of the eye enhancer was almost completely blocked (Figure 6A): flies in 11 transgenic lines had eye pigmentation in the range from orange to yellow. As Zeste is essential for the eye enhancer activity, we regarded the eye phenotype in the zv77h background as resulting from deletion of the eye enhancer. Crossing transgenic lines into the mutant zv77h background only weakly diminished eye pigmentation in 5 out of 11 lines (Figure 6A), confirming that the eye enhancer was inactive. At the same time, the deletion of the downstream gypsy insulator restored white expression, suggesting that the interaction between the gypsy insulators is critical for enhancer blocking.
When the gypsy insulators were inserted in the same orientation (Figure 6B), flies displayed higher levels of eye pigmentation. Crossing the transgenes into the zv77h background significantly reduced eye pigmentation in all test lines, indicating that the eye enhancer was partially active. Therefore, when the gypsy insulators had the same orientation, their enhancer-blocking potential was reduced.
To test if the distance between the chromatin domain formed by insulators bordering the white gene and the eye enhancer is important for insulation, we inserted a 4.6-kb DNA fragment between the eye enhancer and the proximal gypsy insulator (Figure 6C). The gypsy insulators were placed in opposite orientations. In 14 transgenic lines, flies displayed a wide range of eye colors, from brown-red to yellow. The zv77h mutation reduced eye pigmentation in 11 out of 14 lines; i.e., the eye enhancer could stimulate transcription in most of the lines. Thus, an increase in the distance between the eye enhancer and the chromatin domain formed by the gypsy insulators diminished the insulating effect of the loop.
Next, we inserted the 4.6-kb fragment between the gypsy insulator and the white promoter (Figure 6D) so that the eye enhancer was near the upstream insulator. As a result, the white promoter was in the center of the chromatin domain formed by the gypsy insulators inserted in opposite orientations. Once again, we found that the eye enhancer was partially active in all transgenic lines, indicating that the distance between the enhancer or promoter and the gypsy insulator is important for blocking activity.
Comparisons of eye phenotypes in all derivative transgenic lines before and after deletion of the gypsy insulator located on the 3′ side of the white gene (Figures 6A, 6C, 6D) showed that the downstream gypsy insulator effectively stimulated white expression. Thus, the gypsy insulator placed at the end of the white gene can potentiate the white promoter activity.
Mod(mdg4)-67.2 is required for blocking of the eye enhancer by paired gypsy insulators
According to FlyBase data, the su(Hw) gene is weakly expressed in the eyes of adult flies. Therefore, the level of the Su(Hw) protein is also low, which may account for the inability of a single copy of the gypsy insulator to block the eye enhancer. To test this possibility, we produced three transgenic lines carrying the su(Hw) gene under control of the hsp70 promoter (Figure S2A). The elevated level of Su(Hw) had no effect on eye pigmentation in transgenic lines with either one or two gypsy insulators (Figures 7, S3) suggesting that the concentration of this protein is not critical for eye enhancer blocking by the gypsy insulator. Further stimulation of Su(Hw) expression by heat shock (Figure 7, S3) also had no effect on eye pigmentation in any of transgenic lines. Thus, overexpression of Su(Hw) failed to provide for eye enhancer blocking by one copy of the gypsy insulator.
Fig. 7. The role of Su(Hw) and Mod(mdg4)-67.2 proteins in the enhancer blocking by the gypsy insulator. 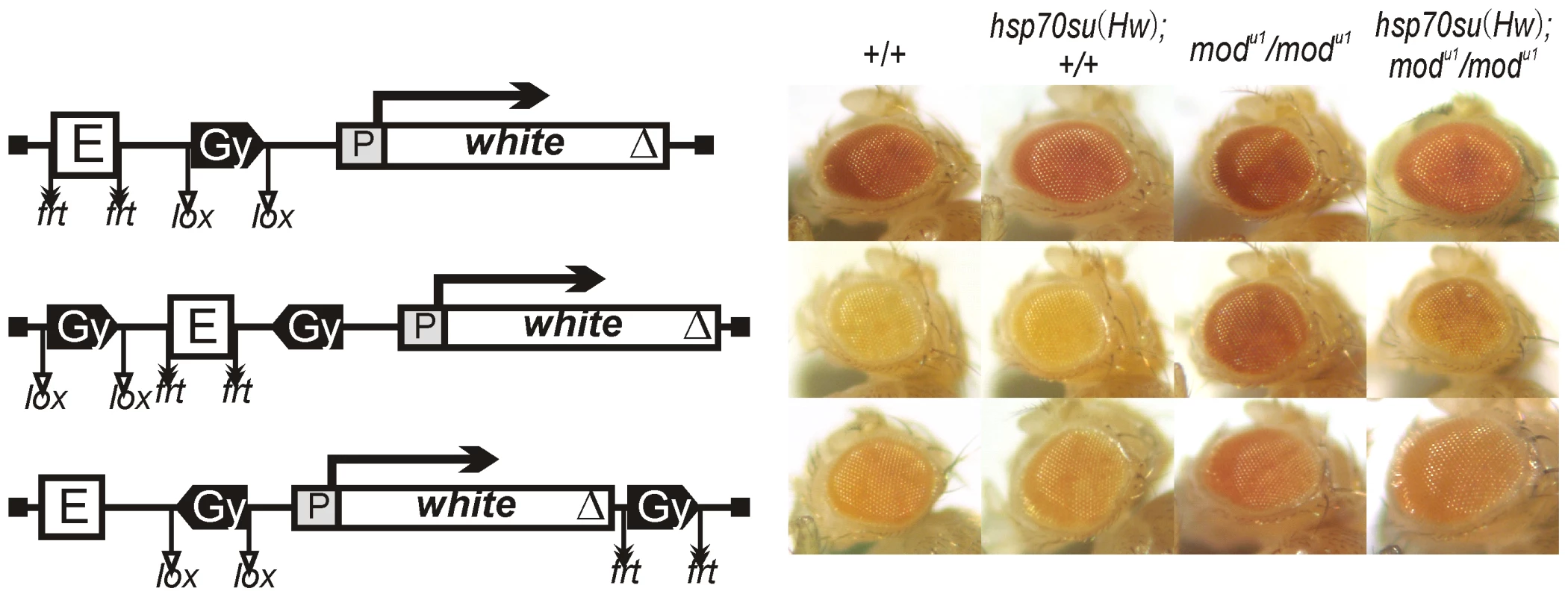
Photographs show eye phenotypes in flies from the transgenic lines carrying either of the constructs in the wild-type (+/+) or the mod(mdg4)u1 mutant (mod) background. Expression of Su(Hw) in transgenic lines carrying the hsp70su(Hw) construct was induced by heat shock. Other designations are as in Figure 1. Next, we examined the role of Su(Hw) and Mod(mdg4)-67.2 proteins in blocking the eye enhancer by two copies of the gypsy insulator. To test Su(Hw), we used the su(Hw)v/su(Hw)2 combination of mutations that significantly reduced the amount of the Su(Hw) protein [66], [67]. In the su(Hw)v/su(Hw)2 background, eye pigmentation was restored to the same level as after deletion of the gypsy insulator, indicating that Su(Hw) is critical for insulation (data not shown).
In the mod(mdg4)u1 and mod(mdg4)T6 mutations, the truncated Mod(mdg4)-67.2 with deleted C-terminal domain partially lost its functional activity [47], [68]. Both mutations significantly but not completely restored eye pigmentation in the transgenic lines carrying two copies of the gypsy insulator (Figures 7, S3), suggesting a role for Mod(mdg4)-67.2 in blocking the eye enhancer.
To further test the role of the insulator proteins, we examined the effect of the mod(mdg4)u1 mutation in combination with overexpression of the Su(Hw) protein (Figure 7, S3). The hsp70su(Hw) transgene did not affect white expression in the mod(mgd4) mutant background in any of transgenic lines, suggesting that a moderate increase in the amount of Su(Hw) is insufficient for counterbalancing Mod(mdg4)-67.2 inactivation. However, strong overexpression of Su(Hw) after induction by heat shock proved to partially restore enhancer blocking by paired gypsy insulators suppressed by the mod(mdg4)u1 and mod(mdg4)T6 mutations (Figure 7, S3).
Previously it was found that, in the mod(mdg4) mutant background, the gypsy insulator directly repressed the yellow promoter in pupae [68] and the white promoter in embryos [69]. Therefore, overexpression of Su(Hw) in the mod(mdg4) mutant background could possibly lead to direct repression of the white promoter. However, induction of Su(Hw) expression by heat shock had no effect on eye pigmentation in flies carrying one copy of the gypsy insulator in the mod(mdg4) mutant background (Figure 7). Thus, a high level of Su(Hw) did not induce direct repression of the white promoter. These results suggest that Mod(mdg4)-67.2 is required for blocking the eye enhancer by paired gypsy insulator and that overexpression of Su(Hw) can partially compensate for inactivation of Mod(mdg4)-67.2 in the mod(mdg4) mutations.
Interestingly, a similar result was obtained with the ct6 mutation, a classical model for testing the activity of the gypsy insulator. In the ct6 allele, a gypsy element is inserted close to and completely blocks a wing margin enhancer located about 85 kb upstream of the cut promoter [47], producing a cut wing phenotype (Figure S2B). The mod(mdg4)u1 mutation almost completely suppressed the ct6 mutant phenotype, suggesting that Mod(mdg4)-67.2 is essential for blocking the wing enhancer. Overexpression of Su(Hw) in the mod(mdg4) mutant background partially rescued the blocking of the wing enhancer, resulting in an intermediate cut wing phenotype. Thus, a high level of Su(Hw) can partially counterbalance the negative influence of the mod(mdg4)u1 mutation on insulation. This is also supported by the previous observation that overexpression of Su(Hw) partially restored y2 expression in bristles that was repressed in the mod(mdg4)u1 background [70].
Finally, we tested whether Mod(mdg)-67.2 is essential for the ability of the distal gypsy insulator located on the 3′ side of the gene to stimulate white expression. We examined six transgenic lines (described in Figure 6) carrying only the distal gypsy insulator and the eye enhancer located either close to the white promoter (Figure S4 A) or at a distance of 4 kb from it (Figure S4 B, C). The results showed that deletion of the gypsy insulator reduced white expression in all cases, while its reduction in mod(mdg4)u1 mutants was relatively weak. Thus, Mod(mdg4)-67.2 proved to be essential but not critical for the ability of the gypsy insulator to stimulate white expression from a distance.
The gypsy insulator directly interacts with the eye enhancer in vivo
The ability of the gypsy insulator located on the 3′ side of the white gene to stimulate its expression suggested that the insulator may directly interact with the white regulatory regions. To test this possibility, we used the 3C assay to examine long-distance interactions in pupae from a transgenic line homozygous for the construct shown in Figure 6D. In this transgene (Figure 8A), the eye enhancer was isolated by the proximal gypsy insulator located at 4.6 kb from the white promoter, while the distal gypsy insulator was inserted downstream of the white gene. We examined the original transgenic line and two derivatives carrying either the distal gypsy insulator or no insulators. We also tested the role of the mod(mdg4)u1 mutation in the interaction between the distal gypsy insulator and the regulatory regions of the white gene.
Fig. 8. The gypsy insulators directly interact with the enhancer and promoter of the white gene. 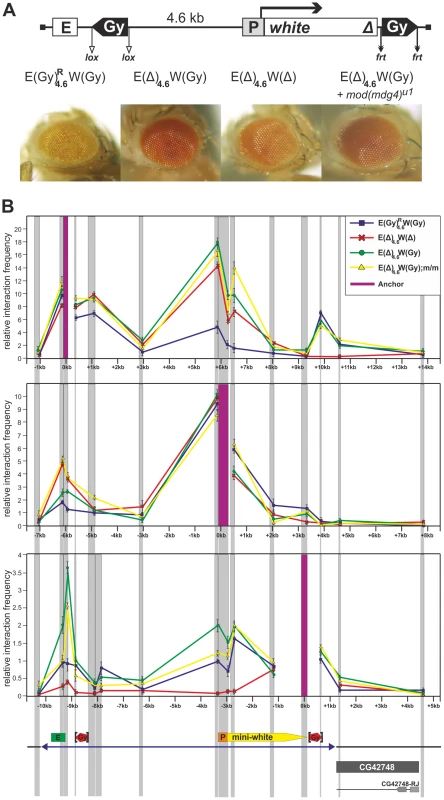
(A) Eye phenotypes in flies from the transgenic line heterozygous (P/+) for the construct and in flies after deletion of either the proximal gypsy insulator, or both insulators, or introduction of the mod(mdg4)u1 mutation. (B) The 3C profile of the E(Gy)R4.5W(Gy) line and its derivatives analyzed at the pupa stage for an anchor fragment located in the enhancer or promoter region or downstream of the distal gypsy insulator. The transgene and the surrounding genomic region are drawn to scale. Relative interaction frequencies between DpnII restriction fragments selected for this analysis (gray vertical bands) and anchor regions (purple vertical bands) are shown for the construct (blue-square line), and its derivatives with deletion of proximal gypsy (green-circle line), proximal gypsy at the mod(mdg4)u1 background (yellow-triangle line), and both gypsy copies (red-cross line). Samples were normalized by qPCR relative to an undigested locus. Error bars show standard deviations from the means of three independent experiments. Transgene DNA is shown as blue line at the bottom of figure, and the boundaries of the transgenes are indicated by blue triangles. The white gene, eye enhancer, and gypsy insulator are represented, respectively, by yellow and green boxes and red pentagon. The lox and frt sites allowing the excision of insulators are indicated by vertical black brackets. Other designations are as in Figure 1. Using anchors at the eye enhancer, the white promoter, and the distal gypsy insulator, we observed strong interaction between the gypsy insulators (Figure 8B). The interaction between the eye enhancer and the promoter was reduced, correlating with the low level of white expression in the transgenic line carrying two copies of the gypsy insulator. After deletion of the proximal gypsy copy, the enhancer-promoter interaction was considerably increased, and this was accompanied by stimulation of white expression by the enhancer in the derivative transgenic line. Thus, the interaction between two gypsy insulators partially blocked the eye enhancer.
When the distal insulator alone was present in the transgene, this insulator interacted with the promoter and the eye enhancer. The mod(mdg4)u1 mutation did not significantly affect these interactions. In the absence of distal gypsy insulators, no interactions were found between the regulatory regions and the 3′ side of the white gene, confirming that the gypsy insulator is essential for the observed contacts. In correlation with the observed interactions, deletion of the distal insulator, but not introduction of the mod(mdg4)u1 mutation, strongly reduced white expression. These results support the model that the gypsy insulator directly interacts with the white enhancer/promoter and stimulates its basal activity.
To further verify the ability of the gypsy insulator to interact with the white regulatory elements, we used chromatin immunoprecipitation (ChIP) assay to analyze binding of the insulator proteins Su(Hw), E(y)2, CP190, and Mod(mdg4)-67.2 to several sites in the promoter, enhancer and insulator regions in pupae from the same transgenic line and its derivatives (Figure 9). In the derivative transgenic line carrying the construct without gypsy insulators, the insulator proteins were not detected on the enhancer and promoter of the white gene. The recruitment of these proteins to the transgene only by the gypsy insulator allowed us to test interaction of the gypsy insulator with the white regulatory regions. It could be expected that the insulator proteins would be detected on the enhancer or/and promoter if these regulatory elements directly interacted with the insulator. The insulator proteins proved to bind to the insulators and the eye enhancer in the transgenic line carrying two copies of the gypsy insulator, but no binding took place after both copies were deleted. Therefore, pairing of the gypsy insulators did not preclude their interaction with the eye enhancer. When the proximal insulator alone was deleted, we still detected the insulator proteins binding to the eye enhancer (Figure 9). Taken together, these results strongly suggest that one or two copies of the gypsy insulator can directly interact with the eye enhancer.
Fig. 9. Binding of insulator proteins to the eye enhancer, promoter, white gene and gypsy insulators in the transgenic construct. 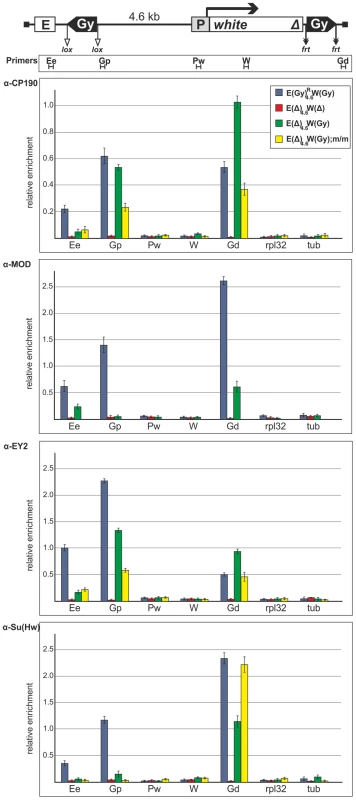
Chromatin was isolated from transgenic flies carrying the construct or its derivatives described in Figure 8 was treated with antibodies against CP190, Mod(mdg4)-67.2 (designated as MOD), E(y)2, and Su(Hw). The results of ChIP are presented as a percentage of input DNA normalized relative to the endogenous positive binding site for insulator proteins from the 62D region. Relative locations of primers for ChIP are indicated under the construct scheme. Proximal and distal gypsy insulators are designated as Gp and Gd correspondingly. The rpl32 and tubulin (tub) coding regions were used as controls devoid of binding sites for the test proteins. Error bars indicate standard deviations of quadruplicate PCR measurements. Other designations are as in Figure 1. In the mod(mdg4)u1 background (Figure 9), the E(y)2 and CP190 proteins continued to bind to the eye enhancer region, confirming the 3C results that the gypsy insulator interacts with the eye enhancer in the absence of Mod(mdg4)-67.2 protein. This finding correlates with data on the ability of the gypsy insulator to stimulate transcription in the mod(mdg4)u1 background.
Interaction of Mod(mdg4)-67.2 with Zeste may be involved in blocking of the eye enhancer
As shown previously, Zeste has binding sites in the enhancer and promoter regions of the white gene [64], and these sites in the promoter region are essential for the insulator bypass by the eye enhancer [71].
Using the ChIP assay, we analyzed the binding of Zeste to the eye enhancer and the promoter of the white gene in pupae from the transgenic line homozygous for the construct and compared its binding to the white promoter before and after deletion of the eye enhancer (Figure 10A). As expected, Zeste bound to the eye enhancer and the white promoter in the initial line, but only traces of this protein were detected on the white promoter in the derivative transgenic line with the deleted eye enhancer. Thus, Zeste was found to be recruited to the white promoter only in the presence of the eye enhancer. In agreement with this finding, inactivation of Zeste in the zv77h mutants did not affect white expression in absence of the eye enhancer (Figure S1). Zeste was not detected on the white promoter in the transgenic line carrying the gypsy insulator between the eye enhancer and the promoter, in agreement with data on the blocking of the eye enhancer by one copy of the gypsy insulator in transgenic lines homozygous for the construct (Figure 10A).
Fig. 10. Binding of Zeste to the white enhancer, promoter, and coding region in the transgenic constructs. 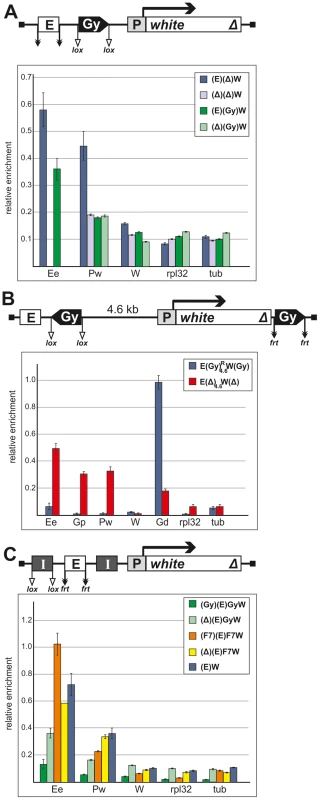
Results of immunoprecipitation experiments with chromatin isolated from transgenic flies and treated with anti-Zeste antibodies. (A) The results of ChIP (percentages of input DNA normalized relative to the endogenous positive binding site for Zeste from the Ubx promoter region) of specified chromatin regions with antibodies to Zeste in the transgenic construct in the presence or absence of gypsy insulator (Gy) in one copy. (B) The results of ChIP with antibodies to Zeste in the derivatives of the transgenic construct described in Figure 8 in the presence or absence of the gypsy insulator (Gy) in one copy. (C) Comparison of the results of ChIP with anti-Zeste antibodies in the presence of two copies of either gypsy or Fab-7 insulators (I, black boxes). Designations: E (the eye enhancer), P (promoter) and W (coding region) of the white gene. The rpl32 and tubulin (tub) coding regions were used as controls devoid of Zeste binding sites. Error bars indicate standard deviations of quadruplicate PCR measurements. Other designations are as in Figure 1 and 9. Next, we examined the binding of Zeste to the transgenic line and its derivatives described in Figures 8 and 9. In the presence of two gypsy insulators, the enrichment of enhancer and promoter sequences upon ChIP with anti-Zeste antibodies was considerably reduced (Figure 10B). However, we unexpectedly observed strong Zeste binding to the distal insulator. Similar results were obtained with two additional transgenic lines in which the white gene was flanked by the gypsy insulators (Figure S5). For both transgenic lines, we observed strong enrichment of sequences related to the distal gypsy insulator upon ChIP with anti-Zeste antibodies. At the same time, binding of Zeste to the promoter and the enhancer was considerably reduced.
After deletion of the proximal gypsy insulator (Figure 10B), the level of Zeste at the white enhancer and promoter was still low, but this protein was detected at the reference sequence near the eye enhancer. After deletion of both gypsy insulator, Zeste was again detected by ChIP on the eye enhancer and the promoter. To confirm these results, we analyzed the enrichment of the enhancer and promoter regions by ChIP with anti-Zeste antibodies in several additional transgenic lines. The enrichment of enhancer sequences was considerably reduced in all transgenic lines carrying one or two copies of the gypsy insulator near the eye enhancer (Figures 10A, 10B, S5), but it returned to the reference level after the insulators were deleted.
In experiments with pupae carrying the transgene with the eye enhancer flanked by two Fab-7 insulators, the enrichment of the enhancer sequences upon ChIP with anti-Zeste antibodies was even higher than in experiments with the control transgenic line carrying only the eye enhancer or the derivative transgenic line with the deleted upstream Fab-7 insulator (Figure 10C). Thus, proteins bound to the Fab-7 insulator did not interfere with Zeste.
The plausible explanation of these results is that the proteins bound to the gypsy insulators interacted with Zeste and partially masked the epitopes recognized by the antibodies in ChIP. As a result, Zeste was not detected at the eye enhancer and promoter but was found at the distal gypsy insulator.
To test this possibility, we examined the interaction of insulator proteins with Zeste in the yeast two-hybrid assay (Figure 11A). The results showed that Zeste interacted with Mod(mdg4)-67.2 but not with the Su(Hw) and CP190 proteins. It is noteworthy that both BTB and C-terminal domains of Mod(mdg4)-67.2 are essential for the interaction with Zeste. We also demonstrated co-immunoprecipitation between Zeste and Mod(mgd4)-67.2 in embryonic extracts (Figure 11B).
Fig. 11. Testing for the interaction between the Mod(mdg4)-67.2 and Zeste proteins. 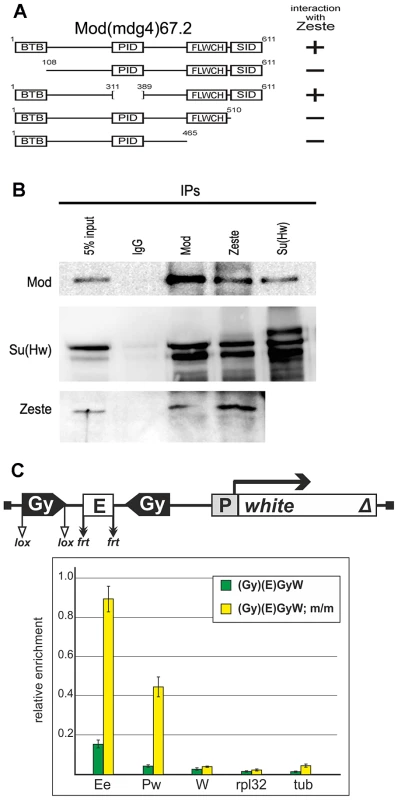
(A) Summary of interactions between Mod(mdg4)-67.2 and Zeste in the yeast two-hybrid assay. Schemes show the structure of the full-length Mod(mdg4)-67.2 and its deletion derivatives, indicating the main domains of this protein. Plus and minus signs refer to a relatively strong interaction and the absence of interaction, respectively. Different fragments of Mod(mdg4)-67.2 were individually fused to the C-terminus of the GAL4 activating domain and tested for the interaction with Zeste fused to the C terminus of GAL4 DNA binding domain. All Mod(mdg4) fragments were tested for the absence of interaction with GAL4 DNA binding domain alone. (B) Nuclear extracts from Drosophila S2 cells were immunoprecipitated with antibodies specific for the indicated proteins (with preimmune IgG used as a negative control), and the immunoprecipitates (IPs) were analyzed by Western blotting for the presence of Mod(mdg4)-67.2 (designated Mod), Su(Hw) and Zeste proteins. (C) The results of ChIP (percentage of input DNA normalized relative to the endogenous positive binding site for Zeste from the Ubx promoter region) of specified chromatin regions with antibodies to Zeste in the transgenic construct. Analysis was performed with wild-type and mod(mdg4)u1/mod(mdg4)u1 (m/m) pupae carrying the transgenic construct. Error bars indicate standard deviations of quadruplicate PCR measurements. Other designations are as in Figure 1 and 9. To confirm the role of Mod(mdg4)-67.2 in the interaction with Zeste, we performed ChIP experiments with pupae from two transgenic lines carrying a pair of gypsy insulators inserted on both sides of the eye enhancer in the mod(mdg4)u1 mutant background (Figure 11C). In both transgenic lines, partial inactivation of Mod(mdg4)-67.2 restored enrichment with the eye enhancer sequence upon ChIP with the anti-Zeste antibodies. At the same time, the mod(mdg4)u1 mutation did not affect Zeste expression in pupae (Figure S6). Taken together these results confirm the role of Mod(mdg4)-67.2–Zeste interaction in pairing of the eye enhancer with the gypsy insulator.
Finally, we used ChIP with anti-Zeste antibodies to test whether Mod(mdg4)-67.2 affected Zeste binding to the eye enhancer in transgenic lines carrying the gypsy insulator on the 3′ side of the white gene. The eye enhancer in these lines was located either close to the white promoter (Figure S7A) or 4.6 kb upstream of it (Figure S7B,C). We unexpectedly observed only a relatively weak enrichment of the enhancer sequence in the presence of the gypsy insulator, but its deletion or introduction of the mod(mdg4)u1 mutation restored the normal level its enrichment in all transgenic lines. These results provide additional evidence for the direct interaction between Mod(mdg4)-67.2 and Zeste and the long-distance interaction of the insulator complex formed on gypsy sequences with the regulatory elements of the white gene.
Discussion
In this study, we have examined two Drosophila insulators in the model system containing the eye enhancer and the white reporter gene lacking the endogenous Wari insulator that improves the enhancer-blocking activity of the gypsy insulator [10]. The results show that one copy of the gypsy or Fab-7 insulator fails to disrupt communication between the eye enhancer and the white promoter in the major part of transgenic lines, with the eye enhancer blocking being effective in only 22–28% of these lines. A plausible explanation to such a position-dependent enhancer blocking activity of the insulators is that there is only a minor part of genomic sites where an endogenous insulator and a transgenic insulator can form a loop that results in isolation of the eye enhancer. Alternatively, the strength of insulation depends on the functional activity of the eye enhancer, which depends on the site of transgene insertion [71].
We demonstrated the role of a putative chromatin loop formed by the gypsy insulators in blocking the eye enhancer. In general, two gypsy insulators flanking either the eye enhancer or the white gene effectively block the enhancer–promoter communication. However, two Fab-7 insulators fail to effectively block the eye enhancer activity, which is unlikely to be explained by their inability to form a loop around the eye enhancer. As shown previously, the interaction between the Fab-7 insulators can support long-distance enhancer–promoter communication [59] and higher-order organization of PcG targets in the nucleus [12]. Thus, the Fab-7 insulators should form a chromatin loop including the eye enhancer that fails to block the enhancer–promoter communication. Taken together, these results suggest that the chromatin loop formed by the interacting insulators is not sufficient for blocking enhancer–promoter communication coordinated by Zeste (Figure 12A). It appears that insulator complexes do not function as a neutral barrier and that specific interactions between insulator proteins and proteins bound either to an enhancer or to a promoter are essential for the effectiveness of blocking.
Fig. 12. Model describing the mechanisms of enhancer blocking by the gypsy insulator. 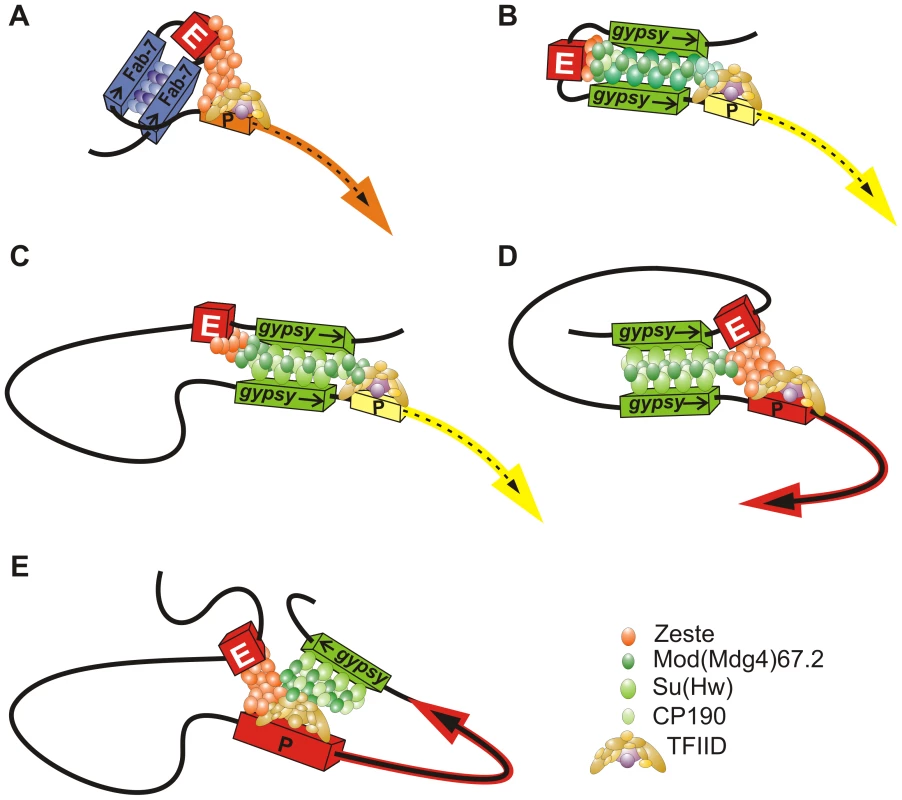
(A) The chromatin loop formed by the interacting insulators is not sufficient for effective blocking of the enhancer–promoter communication coordinated by Zeste. (B) The gypsy insulators block the eye enhancer by two different mechanisms: the chromatin loop formed by interacting gypsy insulators induces physical constrains on the enhancer–promoter communication; the interaction of Mod(mdg4)-67.2 with Zeste interferes with the enhancer-promoter communication. (C) The gypsy insulators located in opposite orientation block interaction between the eye enhancer located within the loop and white promoter located outside the loop; (D) The gypsy insulators located in the same orientation bring together the eye enhancer located within the loop and the white promoter located outside the loop. (E) The gypsy insulator located on the 3′ side of the white gene interacts with the enhancer and promoter. Here we have found that the proteins bound to the gypsy insulator effectively interact with the enhancer of the white gene. The gypsy insulator recruits Mod(mdg4)-67.2 that directly interacts with the Zeste protein. It appears that the BTB and C-terminal domains of Mod(mdg4)-67.2 are required for interaction with Zeste. Mod(mdg4)-67.2 can oligomerize through its BTB and second dimerization domain [51], [52], and this can help it to effectively interfere with the activity of Zeste, which also forms oligomers. Zeste is critical for the long-distance interaction between the white promoter and the eye enhancer [64], [71]. Thus, Mod(mdg4)-67.2 may interfere with the ability of Zeste to bring together remote regulatory elements. As the inactivation of Mod(mdg4)-67.2 does not completely disrupt the enhancer blocking, it seems likely that other insulator proteins also contribute to interactions with proteins bound to the eye enhancer and, therefore, may interfere with the enhancer–promoter communication.
The results presented above provide the basis for the model that two different mechanisms are involved in blocking the eye enhancer by the gypsy insulators: (1) a chromatin loop physically interferes with the ability of the protein complexes bound to the eye enhancer and promoter to interact with each other, and (2) direct interactions between insulator proteins and enhancer/promoter proteins interfere with the ability of an enhancer to properly stimulate a promoter. In particular, Mod(mdg4)-67.2 partially blocks the activity of Zeste via a direct protein–protein interaction (Figure 12B). These mechanisms function cooperatively, which ensures a strong blocking of the eye enhancer by the paired gypsy insulators. It seems likely that the protein complex bound to the Fab-7 insulator does not interfere with the activity of Zeste. As a result, a loop formed by the Fab-7 insulators only weakly affects communication between the eye enhancer and the white promoter.
As in the case of the insulator-mediated chromatin loop, the pairing of the gypsy insulators located on the homologous chromosomes may physically interfere with the enhancer–promoter communication. Thus, homologous pairing might be one of possible mechanisms contributing to the enhancer blocking activity of insulators. Supposedly, such mechanism may account for dosage compensation of some X-chromosomal genes that contain insulators between enhancers and promoters. Since paring between insulators can occur only in females, which have two X chromosomes, such insulators should block enhancers more effectively in females than in males.
Previously, we have used the Flp-recombination assay [53] to demonstrate that the pairing between gypsy insulators strongly depends on their relative orientation. According to our model, the orientation-dependent effect is explained by the involvement of at least two insulator-bound proteins in specific protein-protein interactions. Here we have found that when the eye enhancer is in the center of the loop, the relative orientation of the gypsy insulators is not critical for the efficient blocking of the eye enhancer. The opposite situation is observed when the eye enhancer is in close proximity to the upstream gypsy insulator (Figure 12C,D). In this configuration of the regulatory elements, the gypsy insulators located in the same orientation bring together the eye enhancer located within the loop and the white promoter located outside the loop. Therefore, the position of an enhancer relative to gypsy insulators within the loop appears to be critical for the functional outcome of loop formation.
The results of our study also support the model that insulators specifically interact with enhancers and promoters and potentiate their activity [72]–[74]. As shown previously, the endogenous Su(Hw)-binding region (1A2) placed at the 3′ end of the white gene in the transgenic line can interact with the promoter and stimulate its activity [74]. Here, we have found that the gypsy insulator located on the 3′ side of the white gene stimulates white expression by interacting with the enhancer (Figure 12D). The observed interaction between Mod(mdg4)-67.2 and Zeste is important but not critical for distant pairing of the eye enhancer and the gypsy insulator. Further study is required to identify additional transcription factors bound to the insulators, enhancers, and promoters that are involved in such interactions.
Materials and Methods
Drosophila strains, germline transformation, and genetic crosses
Flies were maintained at 25°C on standard medium. The construct, together with a P element containing defective inverted repeats (P25.7wc) that was used as a transposase source, were injected into yacw1118 preblastoderm embryos [75]. The resulting flies were crossed with yacw1118 flies, and the transgenic progeny were identified by their eye pigmentation under a Stemi 2000 stereomicroscope (Carl Zeiss, Germany). The transformed lines were tested for transposon integrity and copy number by Southern blot hybridization. Only single-copy transformants were included in the study.
The lines with DNA fragment excisions were obtained by crossing the transposon-bearing flies with the Flp (w1118; S2CyO, hsFLP, ISA/Sco; +) or Cre (y1w1; Cyo, P[w+, cre]/Sco; +) recombinase-expressing lines [76], [77]. The Cre recombinase induces 100% excisions in the next generation. A high level of Flp recombinase was produced by heat shock treatment (2 hours daily) during the first 3 days after hatching. All excisions were confirmed by PCR analysis.
To inactivate the Zeste protein, we used the null zv77h mutation (zv77h w67c23, Bloomington Stock Center #1385), which contains a 314-bp deletion including RNA leader sequences and the AUG initiation codon of zeste [65]. To inactivate Mod(mdg4)-67.2, we used the mod(mdg4)u1 and mod(mdg4)T6 mutations associated with the deletion of the C-terminal protein domain interacting with Su(Hw) [48], [68].
Generation of transgenic lines and introduction of zv77h, mod(mdg4) mutations, su(Hw)2/su(Hw)v mutations, and hsp70su(Hw) construct were described previously [51], [68], [71]. To express the su(Hw) gene regulated by the hsp70 promoter, flies carrying the construct were heat-shocked for 2 h every day during the period from the second instar larva to middle pupa stages.
To estimate the levels of white expression, we visually determined the degree of pigmentation in the eyes (white) of 1 - to 3-day-old males developing at 25°C, with reference to standard color scales. All flies were scored independently by two observers. On the nine-grade scale for white, red (R) eyes corresponded to the wild type and white (W) eyes to the total loss of white expression; intermediate pigmentation levels, in order of decreasing gene expression, were brownish red (BrR), brown (Br), dark orange (dOr), orange (Or), dark yellow (dY), yellow (Y) and pale yellow (pY).
Plasmid construction
The constructs were based on the CaSpeR vector [78]. The pCaSpew15(+RI) plasmid was constructed by inserting an additional EcoRI site at +3190 of the mini-white gene (white) in the pCaSpew15 plasmid. The Wari insulator located on the 3′ side of the white gene was deleted from pCaSpew15(+RI) to produce plasmid pCaSpeRΔ700 (CΔ). The 777-bp white regulatory sequences containing the testis and eye enhancers from −967 to −1745 relative to the transcription start site (E) were cloned between two frt sites (frt(E)). Insulator fragments (I) were obtained by PCR amplification. The 340-bp fragment containing the Su(Hw)-binding region (Gy) was PCR amplified from the gypsy retrotransposon. The 0.858-kb Fab-7 fragment (F7) was cloned by PCR amplification between primers 5′-GATTTCAAGCTGTGTGGCGGGG-3′ and 5′-CGTGAGCGACCGAAACTC-3′. The products after sequencing were subcloned in pSK plasmid, or between lox (lox(I)) or frt sites (frt(I)).
(E)(I)W: The frt(E) fragment was inserted in front of the white promoter into the CΔ plasmid digested with XbaI ((E)W). The lox(F7) or lox(Gy) fragment was cloned in the middle of the 480-bp PvuI–PvuI fragment from lacZ cDNA digested with EcoRV. The resulting DNA fragment was inserted into the (E)W digested with BamHI. In the plasmid, the insulators were at −695 relative to the white transcription start site.
(I)(E)IRW: The lox(F7) or lox(Gy) fragment was cloned into the (E)W plasmid digested with PstI just upstream of the eye enhancer ((I)(E)W). The second insulator fragment was cloned in the middle of 480-bp lacZ spacer (Isp) and then was inserted into the (I)(E)W plasmid between the eye enhancer and promoter at −345 relative to the white transcription start site. As a result, the proximal insulator was located at 585 bp from the transcription start site.
E(Gy)W(Gy) and E(Gy)RW(Gy): The eye enhancer was cloned into the CΔ plasmid digested with XbaI (EW). The lox(Gy) fragment was cloned in two orientations in the EW plasmid between the eye enhancer and the promoter at −482 relative to the white transcription start site. The frt(Gy) fragment was cloned into the EW plasmid digested with EcoRI in direct orientation relative to the white gene.
E4,6(Gy)RW(Gy): The frt(Gy) fragment was cloned in EW digested with EcoRI (EW(Gy)). The 4.6-kb BamHI-BglII fragment of the yellow gene was cloned in the EW(Gy) plasmid digested with BamHI between the eye enhancer and the promoter (E4,6W(Gy)). The lox(Gy) fragment was cloned in reverse orientation in the E4,6W plasmid digested with BglII between the eye enhancer and the promoter at −482 relative to the white transcription start site.
E(Gy)R4,6W(Gy): This construct was made like E4, 6(Gy)RW(Gy), but the lox(Gy) fragment was cloned in reverse orientation in E4, 6W(Gy) restricted with BamHI between the white enhancer and promoter in position −5096 relative to the white transcription start site.
(Gy)E4,6(Gy)W and (Gy)RE 4,6(Gy)W: The 4.6-kb BamHI–BglII fragment of the yellow coding region was cloned in EW digested with BamHI upstream of the white promoter (E4,6W). The lox(Gy) fragment was cloned in both orientations into the E4,6W plasmid digested with PstI upstream of the eye enhancer. The frt(Gy) fragment was cloned in direct orientation in position −462 relative to the white transcription start site (considering one frt site).
(Gy)2,1(E)2,2GyW and (Gy)R2,1(E)2,2GyW: The 4.6-kb BamHI–BglII fragment of the yellow coding region was cloned in CΔ digested with BamHI in front of the white promoter (4,6W). The lacZ gene was cloned in pBluSK; frt(E) was cloned approximately in the center of the lacZ gene digested with Eco47III (1,4frt(E)2,1pSK). The KpnI–BamHI fragment was then cloned in the 4,6W plasmid digested with KpnI–BglII (1,8frt(E)2,4W). The Gysp fragment was inserted in the 1,4frt(E)2,4W plasmid digested with SmaI in front of the white promoter (1,8frt(E)2,2GyW). The lox(Gy) fragment was inserted into the 1,8frt(E) 2,2GyW plasmid digested with XbaI in both orientations.
(Gy)2,1(E)2,2Gy2,0W: The construct was made like (Gy)2,1(E)2,2GyW, but the 1460-bp EcoRI–BglII yellow fragment was inserted between proximal Gy and white promoter. As a result, the distance between the proximal gypsy insulator and white transcription start site was 2055 bp.
Chromatin immunoprecipitation
Chromatin was prepared from mid-late pupae. A 500-mg sample was ground in a mortar in liquid nitrogen and resuspended in 10 mL of buffer A (15 mM HEPES-KOH, pH 7.6; 60 mM KCl, 15 mM NaCl, 13 mM EDTA, 0.1 mM EGTA, 0.15 mM spermine, 0.5 mM spermidine, 0.5% NP-40, 0.5 mM DTT) supplemented with 0.5 mM PMSF and Complete (EDTA-free) Protease Inhibitor Cocktail V (Calbiochem, United States). The suspension was then homogenized in a Dounce homogenizer with pestle B and filtered through Nylon Cell Strainer (BD Biosciences, United States). The homogenate was transferred to 3 mL of buffer A with 10% sucrose (AS), and the nuclei were pelleted by centrifugation at 4 000 g, 4°C for 5 min. The pellet was resuspended in 5 mL of buffer A, homogenized again in a Dounce homogenizer, and transferred to 1.5 mL of buffer AS to collect the nuclei by centrifugation. The nuclear pellet was resuspended in wash buffer (15 mM HEPES-KOH, pH 7.6; 60 mM KCl, 15 mM NaCl, 1 mM EDTA, 0.1 mM EGTA, 0.1% NP-40, protease inhibitors) and cross-linked with 1% formaldehyde for 15 min at room temperature. Cross-linking was stopped by adding glycine to a final concentration of 125 mM. The nuclei were washed with three 10-mL portions of wash buffer and resuspended in 1.5 mL of nuclei lysis buffer (15 mM HEPES, pH 7.6; 140 mM NaCl, 1 mM EDTA, 0.1 mM EGTA, 1% Triton X-100, 0.5 mM DTT, 0.1% sodium deoxycholate, 0.1% SDS, protease inhibitors). The suspension was sonicated on ice with a Branson Sonifier 150 (Branson Instruments, United States) for 5×20 sec at 1-min intervals. Debris was removed by centrifugation at 14000 g, 4°C for 10 min, and chromatin was pre-cleared in Protein G agarose (Pierce, Unites States) blocked with BSA and salmon sperm DNA. Aliquots of such pre-cleared chromatin were used as the input samples. These samples were incubated overnight, at 4°C, with rat antibodies against Zeste (1∶100), Su(Hw) (1∶500), and CP190 (1∶500); rabbit antibodies against Mod(mdg4)-67.2 (1∶500) and Ey2 (1∶200); and nonspecific IgG purified from rat and rabbit preimmune sera. Chromatin–antibody complexes were collected using blocked protein G (for rat probes) or A (for rabbit probes) agarose at 4°C over 5 h. After several rounds of washing with lysis buffer (as such and with 500 mM NaCl), LiCl buffer (20 mM Tris-HCl, pH 8; 250 mM LiCl, 1 mM EDTA, 0.5% NP-40, 0.5% sodium deoxycholate, protease inhibitors), and TE buffer, the DNA was eluted with elution buffer (50 mM Tris-HCl, pH 8; 1 mM EDTA, 1% SDS), the cross-links were reversed, and the precipitated DNA was extracted by the phenol–chloroform method. The enrichment of specific DNA fragments was analyzed by real-time PCR, using a StepOne Plus Thermal Cycler (Applied Biosystems, United States). Relative enrichment was calculated as a percentage of the input normalized to a control positive genomic site (region 62D for Su(Hw), Cp190, Mod(mdg4)-67.2, and EY2 [36], and PRE from the Ubx gene for Zeste [79]. The primers used for PCR in ChIP experiments for genome fragments are shown in Table S1.
Chromosome conformation capture assay
The 3C assay was performed as described [54], with minimal modifications. The nuclear pellet prepared from 50-mg sample of pupae (see Chromatin Immunoprecipitation) was washed with wash buffer and resuspended in 5 mL of nucleus preparation (NP) buffer (15 mM HEPES, pH 7.6; 60 mM KCl, 15 mM NaCl, 4 mM MgCl2, 0.1% Triton X-100, 0.5 mM DTT, 1× Complete (EDTA-free) Protease Inhibitor Cocktail V (Calbiochem, United States), and 2% formaldehyde). The suspension was incubated with slow agitation for a total cross-linking time of 30 min at 25°C. Fixation was stopped by adding 2.5 M glycine to a concentration of 0.125 M, and the sample was cooled on ice for at least 5 min and centrifuged at 2500 g, 4°C for 5 min. The pellet was washed with two portions of cold NP buffer and one portion of cold 1.25× NEBuffer 3 (62.5 mM Tris-HCl, pH 8.0; 125 mM NaCl, 12.5 mM MgCl2, 1.25 mM DTT) (New England Biolabs, United States) and resuspended in 300 µL of 1.25× NEBuffer 3. The suspension was supplemented with 4.5 µL of 20% SDS and incubated at the thermoshaker at 37°C and 1400 rpm for 1 h; then 34 µL of 20% Triton X-100 was added, and the sample was incubated again at 37°C and 1400 rpm for 1 h. At this stage, a 30-µL aliquot of the sample was taken to be used as the undigested control. Thereafter, 1500 units of DpnII (New England Biolabs) was added, and the sample was incubated at 37°C and 1400 rpm for 10 h and then at 65°C and 1400 rpm for 20 min to inactivate DpnII. At this stage, a 30-µL aliquot of the sample was taken to be used as the digested control. Another 170-µL aliquot was cooled, mixed with 250 µL of 1.7× ligation buffer (1.7× T4 DNA ligase buffer with 100 units/mL of T4 DNA Ligase, New England Biolabs), and incubated with slow agitation at 25°C for 5 h. At this stage, a 75-µL aliquot of the sample was taken to be used as the ligation control. Cross-links were reversed overnight at 65°C and 1400 rpm. The sample was incubated with 4 µL of 10 mg/mL RNase A at 37°C and 1400 rpm for 1 h and then with 11 µL of 20 mg/mL Proteinase K at 56°C and 1400 rpm for 4 h. The sample was extracted with 10 mM Tris-HCl (pH 8.0) saturated phenol, 10 mM Tris-HCl (pH 8.0) saturated phenol/chloroform/isoamyl alcohol, and chloroform/isoamyl alcohol. At the each step, the mixture was centrifuged at 10 000 g and room temperature for 10 min. The extracted solution was mixed with 0.1 volume of 3 M AcNa (pH 5.2) containing 35 µg glycogen and 2 volumes of 96% ethanol and incubated overnight at −80°C. DNA was precipitated by centrifugation at 20 000 g for 90 min at 4°C. The DNA pellet was washed with 70% ethanol, air dried, and resuspended in 100 µL of 10 mM Tris-HCl (pH 7.5). Sample DNA concentrations were adjusted to 10 ng/µL with 10 mM Tris-HCl (pH 8.0). All control procedures and quantitative analyses were performed as described [80]. As a control template containing all ligation products in equal amounts, we used a BAC clone (BACR06H06, RPCI-98, Roswell Park Cancer Institute Drosophila melanogaster BAC library) covering the site of transgenic construct integration into the genome, which was mixed in equimolar amounts with plasmid DNA construct digested with DpnII (New England Biolabs) at a concentration of 10 U/µg DNA. Digested DNA was purified by phenol/chloroform extraction and ethanol precipitation as described above, ligated with T4 DNA ligase at 25°C for 5 h, and purified again in the same way.
Primers were designed so as to flank DpnII restriction sites in the transgenic construct. TaqMan Probes were designed with 5′FAM reporter dye and 3′BHG1 quencher dye. To normalize for the PCR efficiency of different primer pair/probe combinations, standard curves for each combination were generated using the BAC control template. Interaction frequencies were calculated on the basis of Ct values of each sample relative to the standard curve for the given primer pair/probe combination. The primers and probes used in the study are listed in Table S1.
All real-time PCR reactions were carried out in a StepOne Plus system (Applied Biosystems) according to the manufacturer's instructions, in four replications each. Amplification involved initial denaturation at 95°C for 15 min followed by 50 cycles of 95°C for 15 s and 60°C for 60 s. To compare interaction frequencies, the normalization procedure was performed: the amount of test ligation product was divided by the amount of reference product to give a “relative interaction frequency.” Loading adjustment was performed as described [81] to provide for normalization and subsequent comparison of probes from different transgenic flies.
Western blotting
A sample of 20 flies was ground with a homogenizer in extraction buffer (20 mM HEPES, pH 7.5; 100 mM KCl, 5% glycerol, 10 mM EDTA, 0.1% Triton X-100, 1 mM DTT, 0.5 mM PMSF, 20 mg/mL aprotinin, 5 mg/mL leupeptin, 5 mg/mL pepstatin A), 10 µL per fly). Debris was removed by centrifugation at 12 000 g, 4°C for 10 min, and the appropriate amount of 5× SDS loading was added directly to the homogenate. The sample was boiled for 5 min, centrifuged at 12 000 g for 5 min, and loaded onto SDS-PAGE.
Antibodies
Mouse anti-lamin antibody ADL67.10 (working dilution for Western blotting, 1∶1000) was from the Hybridoma Bank at the University of Iowa. Rabbit anti-Mod(mdg4)-67.2 antibody (1∶5000) was a gift from A. Golovnin. Rat antibodies against α-Su(Hw) (1∶1000), α-CP190 (1∶1000), α-Zeste (1∶200), and E(y)2 (1∶200) were raised in our laboratory and affinity purified. To this end, protein fragments 6× His-Su(Hw) (aa 1–150), CP190 (aa 308–1096), Zeste (aa 1–175) and EY2 were expressed in BL21 cells, affinity purified on Ni-NTA agarose (Invitrogen) according to the manufacturer's protocol, and injected into rats following the standard immunization procedure.
RNA isolation and real-time PCR
Total RNA was isolated using the TRI reagent (Molecular Research Center, United States) according to the manufacturer's instructions. RNA was treated with two units of Turbo DNase I (Ambion) for 30 min at 37°C to eliminate genomic DNA. The synthesis of cDNA was performed using 2 µg of RNA, ArrayScript reverse transcriptase (Ambion) and oligo(dT) as a primer. The amounts of specific cDNA fragments were quantified by real-time PCR. At least three independent experiments with each primer set were performed for three independent RNA samples. Relative levels of mRNA expression were calculated in the linear amplification range by calibration to a standard curve of genomic DNA to account for differences in primer efficiencies. Individual expression values were normalized with reference to rpl32 mRNA.
Preparation of the nuclear extract and immunoprecipitation
The nuclear extracts were obtained from S2 cells, and the protein complexes were immunoprecipitated from the extracts. For this purpose, 1×108 S2 cells were washed twice in 10 mL of ice cold PBS, resuspended in 10 mL of ice cold IP-Sucrose buffer (10 mM Tris, pH 7.5; 10 mM NaCl, 10 mM MgCl2, 1 mM EDTA, 1 mM EGTA, 1 mM DTT, 250 mM sucrose, 0.5 mM PMSF) with Complete (EDTA-free) Protease Inhibitor Cocktail V (Calbiochem, United States), incubated on ice for 10 min, and homogenized with a Dounce loose pestle (20 strokes). The nuclei were then pelleted by centrifugation at 3000 g, 4°C for 10 min. The pellet was resuspended in 500 µL of ice cold IP-10 buffer (10 mM Tris, pH 7.5; 10 mM NaCl, 10 mM MgCl2, 1 mM EDTA, 1 mM EGTA, 1 mM DTT;,0.1% NP-40, 10% glycerol, 0.5 mM PMSF, and Complete Protease Inhibitor Cocktail V), homogenized with a Dounce tight pestle (20 strokes), and mixed with an equal volume of IP-850 buffer (10 mM Tris, pH 7.5; 850 mM NaCl, 10 mM MgCl2, 1 mM EDTA, 1 mM EGTA, 1 mM DTT, 0.1% NP-40, 10% glycerol, 0.5 mM PMSF, and Complete Protease Inhibitor Cocktail V). The suspension was incubated on ice for 10 minutes and then centrifuged at 20 000 rpm, 4°C, for 10 min. The supernatant fluid (the nuclear fraction) was collected for immunoprecipitation experiments.
Rat antibodies against α-Su(Hw) (1∶200) and α-Zeste (1∶100) were conjugated with Protein G agarose, and rabbit antibodies against α-Mod(mdg4)-67.2 (1∶500), with Protein A agarose beads (Pierce); in respective control experiments, rat or rabbit preimmune serum was used. An aliquot of an antibody was mixed with 30 µL of agarose beads equilibrated in IP buffer with 150 mM NaCl (IP-150) and incubated on a rotator at 4°C for 3 h. The beads were then washed with IP-150, blocked with 1% BSA for 30 min under the same conditions, and washed with two portions of IP-150. The nuclear extract was adjusted to 150 mM NaCl, and its 1 ml containing approximately 1 mg of total protein was mixed with 30 µL of “fresh” agarose beads equilibrated in IP-150 and incubated at 4°C for 1 h for pre-clearing the sample. The beads were pelleted, and the supernatant fluid was transferred to a new tube and mixed with antibody-conjugated beads. The samples were incubated on a rotary mixer at 4°C for 3 h, and the beads were washed with three portions of IP buffer with 300 mM NaCl, one portion of IP buffer with 500 mM NaCl, and one portion of IP buffer with 150 mM NaCl. After the last washing step, the beads were resuspended in SDS–PAGE loading buffer, boiled, and analyzed by Western blotting. Proteins were detected using the SuperSignal West Fempto substrate (Pierce).
Yeast two-hybrid assay
Yeast two-hybrid assay was carried out using yeast strain pJ69-4A, with plasmids and protocols from Clontech. For growth assays, plasmids were transformed into yeast strain pJ69-4A by the lithium acetate method, as described by the manufacturer, and plated on media without tryptophan and leucine. After 2 days of growth at 30°C, the cells were plated on selective media without tryptophan, leucine, histidine and adenine, and their growth was compared after 2–3 days. Liquid culture β-galactosidase assay was performed according to the yeast protocols handbook (Clontech). Each assay was repeated twice.
Supporting Information
Zdroje
1. Kuhn EJ, GeyerPK (2003) Genomic insulators: Connecting properties to mechanism. Curr Opin Cell Biol 15 : 259–265.
2. BrassetE, VauryC (2005) Insulators are fundamental components of the eukaryotic genomes. Heredity 94(6): 571–576.
3. ZhaoH, DeanA (2005) Organizing the genome: Enhancers and insulators. Biochem Cell Biol 83 : 516–524.
4. WallaceJA, FelsenfeldG (2007) We gather together: Insulators and genome organization. Curr Opin Genet Dev 17 : 400–407.
5. ValenzuelaL, KamakakaRT (2006) Chromatin insulators. Annu Rev Genet 40 : 107–138.
6. MaksimenkoOG, ChetverinaDA, GeorgievPG (2006) Insulators of higher eukaryotes: Properties, mechanisms of action, and role in transcription regulation. Russ J Genet 42 : 845–857.
7. BarkessG, WestAG (2012) Chromatin insulator elements: Establishing barriers to set heterochromatin boundaries. Epigenomics 4 : 67–80.
8. KyrchanovaO, ChetverinaD, MaksimenkoO, KullyevA, GeorgievP (2008) Orientation-dependent interaction between Drosophila insulators is a property of this class of regulatory elements. Nucleic Acids Res 36 : 7019–7028.
9. MaksimenkoO, GolovninA, GeorgievP (2008) Enhancer-promoter communication is regulated by insulator pairing in a Drosophila model bigenic locus. Mol Cell Biol 28 : 5469–5477.
10. ChetverinaD, SavitskayaE, MaksimenkoO, MelnikovaL, ZaytsevaO (2008) Red flag on the white reporter: A versatile insulator abuts the white gene in Drosophila and is omnipresent in mini-white constructs. Nucleic Acids Res 36 : 929–37.
11. KravchenkoE, SavitskayaE, KravchukO, ParshikovA, GeorgievP, et al. (2005) Pairing between gypsy insulators facilitates the enhancer action in trans throughout the Drosophila genome. Mol Cell Biol 25 : 9283–9291.
12. LiHB, MullerM, BahecharIA, KyrchanovaO, OhnoK, et al. (2011) Insulators, not Polycomb Response Elements, are required for long-distance interactions between Polycomb targets in Drosophila melanogaster. Mol Cell Biol 31 : 616–625.
13. AdryanB, WoerfelG, Birch-MachinI, GaoS, QuickM, et al. (2007) Genomic mapping of Suppressor of Hairy-wing binding sites in Drosophila. Genome Biol 8: R167.
14. NègreN, BrownCD, ShahPK, KheradpourP, MorrisonCA, et al. (2010) A comprehensive map of insulator elements for the Drosophila genome. PloS Genet 6: e1000814.
15. NègreN, BrownCD, MaL, BristowCA, MillerS, et al. (2011) A cis-regulatory map of the Drosophila genome. Nature 471 : 527–531.
16. BartkuhnM, StaubT, HeroldM, HerrmannM, RathkeC, et al. (2009) Active promoters and insulators are marked by the centrosomal protein 190. EMBO J 28 : 877–888.
17. RoyS, ErnstJ, KharchenkoPV, KheradpourP, NegreN, et al. (2010) Identification of functional elements and regulatory circuits by Drosophila modENCODE. Science 330 : 1787–1797.
18. KehayovaP, MonahanK, ChenW, ManiatisT (2011) Regulatory elements required for the activation and repression of the protocadherin-{alpha} gene cluster. Proc Natl Acad Sci USA 108 : 17195–17200.
19. LiuZ, ScannellDR, EisenMB, TjianR (2011) Control of embryonic stem cell lineage commitment by core promoter factor, TAF3. Cell 146 : 720–731.
20. HandokoL, XuH, LiG, NganCY, ChewE, et al. (2011) CTCF-mediated functional chromatin interactome in pluripotent cells. Nature Genet 43 : 630–638.
21. DixonJR, SelvarajS, YueF, KimA, LiY, et al. (2012) Topological domains in mammalian genomes identified by analysis of chromatin interactions. Nature 485 : 376–380.
22. SextonT, YaffeE, KenigsbergE, BantigniesF, LeblancB, et al. (2012) Three-dimensional folding and functional organization principles of the Drosophila genome. Cell 148 : 458–472.
23. KellumR, SchedlP (1991) A position-effect assay for boundaries of higher order chromosomal domains. Cell 64 : 941–950.
24. KellumR, SchedlP (1992) A group of scs elements function as domain boundaries in an enhancer-blocking assay. Mol Cell Biol 12 : 2424–2431.
25. HagstromK, MullerM, SchedlP (1996) Fab-7 functions as a chromatin domain boundary to ensure proper segment specification by the Drosophila bithorax complex. Genes Dev 10 : 3202–3215.
26. ZhouJ, BaroloS, SzymanskiP, LevineM (1996) The Fab-7 element of the bithorax complex attenuates enhancer-promoter interactions in the Drosophila embryo. Genes Dev 10 : 3195–3201.
27. BargesS, MihalyJ, GalloniM, HagstromK, MullerM, et al. (2000) The Fab-8 boundary defines the distal limit of the bithorax complex iab-7 domain and insulates iab-7 from initiation elements and a PRE in the adjacent iab-8 domain. Development 127 : 779–790.
28. SchweinsbergS, SchedlP (2004) Developmental modulation of Fab-7 boundary function. Development 131 : 4743–4749.
29. GruzdevaN, KyrchanovaO, ParshikovA, KullyevA, GeorgievP (2005) The Mcp element from the bithorax complex contains an insulator that is capable of pairwise interactions and can facilitate enhancer–promoter communication. Mol Cell Biol 25 : 3682–3689.
30. BelozerovVE, MajumderP, ShenP, CaiHN (2003) A novel boundary element may facilitate independent gene regulation in the Antennapedia complex of Drosophila. EMBO J 22 : 3113–3121.
31. ConteC, DastugueB, VauryC (2002) Coupling of enhancer and insulator properties identified in two retrotransposons modulates their mutagenic impact on nearby genes. Mol Cell Biol 22 : 1767–1777.
32. HoldridgeC, DorsettD (1991) Repression of hsp70 heat shock gene transcription by the suppressor of hairy-wing protein of Drosophila melanogaster. Mol Cell Biol 11 : 1894–1900.
33. GeyerPK, CorcesVG (1992) DNA position-specific repression of transcription by a Drosophila zinc finger protein. Genes Dev 6 : 1865–1873.
34. GolovninA, BiryukovaI, RomanovaO, SilichevaM, ParshikovA, et al. (2003) An endogenous Su(Hw) insulator separates the yellow gene from the Achaete-scute gene complex in Drosophila. Development 130 : 3249–3258.
35. ParnellTJ, VieringMM, SkjesolA, HelouC, KuhnEJ, et al. (2003) An endogenous suppressor of hairy-wing insulator separates regulatory domains in Drosophila. Proc Natl Acad Sci U S A 100 : 13436–13441.
36. ParnellTJ, KuhnEJ, GilmoreBL, HelouC, WoldMS, et al. (2006) Identification of genomic sites that bind the Drosophila suppressor of Hairy-wing insulator protein. Mol Cell Biol 26 : 5983–5993.
37. HeroldM, BartkuhnM, RenkawitzR (2012) CTCF: Insights into insulator function during development. Development 139 : 1045–1057.
38. CaiHN, ShenP (2001) Effects of cis arrangement of chromatin insulators on enhancer-blocking activity. Science 291 : 493–495.
39. MuravyovaE, GolovninA, GrachevaE, ParshikovA, BelenkayaT, et al. (2001) Loss of insulator activity by paired Su(Hw) chromatin insulators. Science 291 : 495–498.
40. BlantonJ, GasznerM, SchedlP (2003) Protein : protein interactions and the pairing of boundary elements in vivo. Genes Dev 17 : 664–675.
41. AmeresSL, DrueppelL, PfleidererK, SchmidtA, HillenW, et al. (2005) Inducible DNA-loop formation blocks transcriptional activation by an SV40 enhancer. EMBO J 24 : 358–367.
42. BondarenkoVA, LiuYV, JiangYI, StuditskyVM (2003) Communication over a large distance: Enhancers and insulators. Biochem Cell Biol 81 : 241–251.
43. BondarenkoVA, JiangYI, StuditskyVM (2003) Rationally designed insulator-like elements can block enhancer action in vitro. EMBO J 22 : 4728–4737.
44. MukhopadhyayS, SchedlP, StuditskyVM, SenguptaAM (2011) Theoretical analysis of the role of chromatin interactions in long-range action of enhancers and insulators. Proc Natl Acad Sci U S A 108 : 19919–19924.
45. GohlD, AokiT, BlantonJ, ShanowerG, KappesG, et al. (2011) Mechanism of chromosomal boundary action: Roadblock, sink, or loop? Genetics 187 : 731–748.
46. HouC, ZhaoH, TanimotoK, DeanA (2008) CTCF-dependent enhancer-blocking by alternative chromatin loop formation. Proc Natl Acad Sci U S A 105 : 20398–20403.
47. GauseM, MorcilloP, DorsettD (2001) Insulation of enhancer–promoter communication by a gypsy transposon insert in the Drosophila cut gene: Cooperation between suppressor of hairy-wing and modifier of mdg4 proteins. Mol Cell Biol 21 : 4807–4817.
48. GhoshD, GerasimovaTI, CorcesVG (2001) Interactions between the Su(Hw) and Mod(mdg4) proteins required for gypsy insulator function. EMBO J 20 : 2518–2527.
49. PaiC-Y, LeiEP, GhoshD, CorcesVG (2004) The centrosomal protein CP190 is a component of the gypsy chromatin insulator. Mol Cell 16 : 737–748.
50. KurshakovaM, MaksimenkoO, GolovninA, PulinaM, GeorgievaS, et al. (2007) Evolutionarily conserved E(y)2/Sus1 protein is essential for the barrier activity of Su(Hw)-dependent insulators in Drosophila. Mol Cell 27 : 332–338.
51. GolovninA, MazurA, KopantsevaM, KurshakovaM, GulakPV, et al. (2007) Integrity of the Mod(mdg4)-67.2 BTB domain is critical to insulator function in Drosophila. Mol Cell Biol 27 : 963–974.
52. BonchukA, DenisovS, GeorgievP, MaksimenkoO (2011) Drosophila BTB/Poz domains of “ttk group” can form multimers and selectively interact with each other. J Mol Biol 412 : 423–436.
53. KrivegaM, SavitskayaE, KrivegaI, KarakozovaM, ParshikovA, et al. (2010) Interaction between a pair of gypsy insulators or between gypsy and Wari insulators modulates Flp site-specific recombination in Drosophila melanogaster. Chromosoma 119 : 425–434.
54. CometI, SchuettengruberB, SextonT, CavalliG (2011) A chromatin insulator driving three-dimensional Polycomb response element (PRE) contacts and Polycomb association with the chromatin fiber. Proc Natl Acad Sci U S A 108 : 2294–2299.
55. MaedaRK, KarchF (2011) Gene expression in time and space: Additive vs. hierarchical organization of cis-regulatory regions. Curr Opin Genet Dev 21 : 187–193.
56. GyurkovicsH, GauszJ, KummerJ, KarchF (1990) A new homeotic mutation in the Drosophila bithorax complex removes a boundary separating two domains of regulation. EMBO J 9 : 2579–2585.
57. GalloniM, GyurkovicsH, SchedlP, KarchF (1993) The bluetail transposon: Evidence for independent cis-regulatory domains and domain boundaries in the bithorax complex. EMBO J 12 : 1087–1097.
58. KarchF, GalloniM, SiposL, GauszJ, GyurkovicsH, et al. (1994) Mcp and Fab-7: Molecular analysis of putative boundaries of cis-regulatory domains in the bithorax complex of Drosophila melanogaster. Nucleic Acids Res 22 : 3138–3146.
59. RodinS, KyrchanovaO, PomerantsevaE, ParshikovA, GeorgievP (2007) New properties of Drosophila Fab-7 insulator. Genetics 177 : 113–121.
60. KuhnEJ, VieringMM, RhodesKM, GeyerPK (2003) A test of insulator interactions in Drosophila. EMBO J 22 : 2463–2471.
61. SchweinsbergS, HagstromK, GohlD, SchedlP, KumarRP, et al. (2004) The enhancer-blocking activity of the Fab-7 boundary from the Drosophila bithorax complex requires GAGA-factor-binding sites. Genetics 168 : 1371–1384.
62. Savitskaya E, Melnikova L, Kostuchenko M, Kravchenko E, Pomerantseva E, et al. 2006. Study of long-distance functional interactions between Su(Hw) insulators that can regulate enhancer–promoter communication in Drosophila melanogaster. Mol Cell Biol 26 : 754–761.
63. MelnikovaL, KostuchenkoM, SilichevaM, GeorgievP (2008) Drosophila gypsy insulator and yellow enhancers regulate activity of yellow promoter through the same regulatory element. Chromosoma 117 : 137–145.
64. QianS, VarjavandB, PirrottaV (1992) Molecular analysis of the zeste-white interaction reveals a promoter-proximal element essential for distant enhancer-promoter communication. Genetics 131 : 79–90.
65. PirrottaV, ManetE, HardonE, BickelSE, BensonM (1987) Structure and sequence of the Drosophila zeste gene. EMBO J 6 : 791–799.
66. HarrisonDA, GdulaDA, CoyneRS, CorcesVG (1993) A leucine zipper domain of the suppressor of Hairy-wing protein mediates its repressive effect on enhancer function. Genes Dev 7 : 1966–1978.
67. KimJ, ShenB, RosenC, DorsettD (1996) The DNA-binding and enhancer-blocking domains of the Drosophila suppressor of Hairy-wing protein. Mol Cell Biol 16 : 3381–3392.
68. GeorgievP, KozycinaM (1996) Interaction between mutations in the suppressor of Hairy wing and modifier of mdg4 genes of Drosophila melanogaster affecting the phenotype of gypsy-induced mutations. Genetics 142 : 425–436.
69. CaiHN, LevineM (1997) The gypsy insulator can function as a promoter-specific silencer in the Drosophila embryo. EMBO J 16 : 1732–1741.
70. CapelsonM, CorcesVG (2005) The ubiquitin ligase dTopors directs the nuclear organization of a chromatin insulator. Mol Cell 20 : 105–116.
71. KostyuchenkoM, SavitskayaE, KoryaginaE, MelnikovaL, KarakozovaM, et al. (2009) Zeste can facilitate long-range enhancer–promoter communication and insulator bypass in Drosophila melanogaster. Chromosoma 118 : 665–674.
72. RaabJR, KamakakaRT (2010) Insulators and promoters: Closer than we think. Nature Rev Genet 11 : 439–446.
73. ChopraVS, CandeJ, HongJW, LevineM (2009) Stalled Hox promoters as chromosomal boundaries. Genes Dev 23 : 1505–1509.
74. ErokhinM, DavydovaA, KyrchanovaO, ParshikovA, GeorgievP, et al. (2011) Insulators form gene loops by interacting with promoters in Drosophila. Development 138 : 4097–4106.
75. KaressRE, RubinGM (1984) Analysis of P transposable element functions in Drosophila. Cell 38 : 135–146.
76. GolicKG, LindquistS (1989) The FLP recombinase of yeast catalyzes site-specific recombination in the Drosophila genome. Cell 59 : 499–509.
77. SiegalML, HartlDL (2000) Application of Cre/loxP in Drosophila: Site-specific recombination and transgene coplacement. Methods Mol Biol 136 : 487–495.
78. PirrottaV (1988) Vectors for P-mediated transformation in Drosophila. Biotechnology 10 : 437–456.
79. BensonM, PirrottaV (1987) The product of the Drosophila zeste gene binds to specific DNA sequences in white and Ubx. EMBO J 6 : 1387–1392.
80. HagègeH, KlousP, BraemC, SplinterE, DekkerJ, CathalaG, de LaatW, FornéT (2007) Quantitative analysis of chromosome conformation capture assays (3C-qPCR). Nature Protoc 2 : 1722–1733.
81. MoshkovichN, NishaP, BoylePJ, ThompsonBA, DaleRK, LeiEP (2011) RNAi-independent role for Argonaute2 in CTCF/CP190 chromatin insulator function. Genes Dev 25 : 1686–1701.
Štítky
Genetika Reprodukční medicína
Článek Independent Evolution of Transcriptional Inactivation on Sex Chromosomes in Birds and MammalsČlánek The bHLH Subgroup IIId Factors Negatively Regulate Jasmonate-Mediated Plant Defense and DevelopmentČlánek Selective Pressures to Maintain Attachment Site Specificity of Integrative and Conjugative ElementsČlánek Reassembly of Nucleosomes at the Promoter Initiates Resilencing Following Decitabine ExposureČlánek Hepatocyte Growth Factor Signaling in Intrapancreatic Ductal Cells Drives Pancreatic Morphogenesis
Článek vyšel v časopisePLOS Genetics
Nejčtenější tento týden
2013 Číslo 7
-
Všechny články tohoto čísla
- An Solution for Crossover Formation
- Genome-Wide Association Mapping in Dogs Enables Identification of the Homeobox Gene, , as a Genetic Component of Neural Tube Defects in Humans
- Independent Evolution of Transcriptional Inactivation on Sex Chromosomes in Birds and Mammals
- Stepwise Activation of the ATR Signaling Pathway upon Increasing Replication Stress Impacts Fragile Site Integrity
- Genomic Analysis of Natural Selection and Phenotypic Variation in High-Altitude Mongolians
- Modification of tRNA by Elongator Is Essential for Efficient Translation of Stress mRNAs
- Role of CTCF Protein in Regulating Locus Transcription
- Gene Set Signature of Reversal Reaction Type I in Leprosy Patients
- Mapping of PARK2 and PACRG Overlapping Regulatory Region Reveals LD Structure and Functional Variants in Association with Leprosy in Unrelated Indian Population Groups
- Is Required for Formation of the Genital Ridge in Mice
- Monopolin Subunit Csm1 Associates with MIND Complex to Establish Monopolar Attachment of Sister Kinetochores at Meiosis I
- Recombination Dynamics of a Human Y-Chromosomal Palindrome: Rapid GC-Biased Gene Conversion, Multi-kilobase Conversion Tracts, and Rare Inversions
- Mechanisms of Protein Sequence Divergence and Incompatibility
- Histone Methyltransferase DOT1L Drives Recovery of Gene Expression after a Genotoxic Attack
- Female Behaviour Drives Expression and Evolution of Gustatory Receptors in Butterflies
- Combinatorial Regulation of Meiotic Holliday Junction Resolution in by HIM-6 (BLM) Helicase, SLX-4, and the SLX-1, MUS-81 and XPF-1 Nucleases
- The bHLH Subgroup IIId Factors Negatively Regulate Jasmonate-Mediated Plant Defense and Development
- The Role of Interruptions in polyQ in the Pathology of SCA1
- Dietary Restriction Induced Longevity Is Mediated by Nuclear Receptor NHR-62 in
- Fine Time Course Expression Analysis Identifies Cascades of Activation and Repression and Maps a Putative Regulator of Mammalian Sex Determination
- Genome-scale Co-evolutionary Inference Identifies Functions and Clients of Bacterial Hsp90
- Oxidative Stress and Replication-Independent DNA Breakage Induced by Arsenic in
- A Moonlighting Enzyme Links Cell Size with Central Metabolism
- Budding Yeast Greatwall and Endosulfines Control Activity and Spatial Regulation of PP2A for Timely Mitotic Progression
- The Conserved Intronic Cleavage and Polyadenylation Site of CstF-77 Gene Imparts Control of 3′ End Processing Activity through Feedback Autoregulation and by U1 snRNP
- The BTB-zinc Finger Transcription Factor Abrupt Acts as an Epithelial Oncogene in through Maintaining a Progenitor-like Cell State
- The Cohesion Protein SOLO Associates with SMC1 and Is Required for Synapsis, Recombination, Homolog Bias and Cohesion and Pairing of Centromeres in Drosophila Meiosis
- The RNA-binding Proteins FMR1, Rasputin and Caprin Act Together with the UBA Protein Lingerer to Restrict Tissue Growth in
- Pattern Dynamics in Adaxial-Abaxial Specific Gene Expression Are Modulated by a Plastid Retrograde Signal during Leaf Development
- A Network of HMG-box Transcription Factors Regulates Sexual Cycle in the Fungus
- Bacterial Adaptation through Loss of Function
- ENU-induced Mutation in the DNA-binding Domain of KLF3 Reveals Important Roles for KLF3 in Cardiovascular Development and Function in Mice
- Interplay between Structure-Specific Endonucleases for Crossover Control during Meiosis
- FGF Signalling Regulates Chromatin Organisation during Neural Differentiation via Mechanisms that Can Be Uncoupled from Transcription
- The Arabidopsis RNA Binding Protein with K Homology Motifs, SHINY1, Interacts with the C-terminal Domain Phosphatase-like 1 (CPL1) to Repress Stress-Inducible Gene Expression
- Selective Pressures to Maintain Attachment Site Specificity of Integrative and Conjugative Elements
- The Conserved ADAMTS-like Protein Lonely heart Mediates Matrix Formation and Cardiac Tissue Integrity
- The cGMP-Dependent Protein Kinase EGL-4 Regulates Nociceptive Behavioral Sensitivity
- RBM5 Is a Male Germ Cell Splicing Factor and Is Required for Spermatid Differentiation and Male Fertility
- Disease-Related Growth Factor and Embryonic Signaling Pathways Modulate an Enhancer of Expression at the 6q23.2 Coronary Heart Disease Locus
- Yeast Pol4 Promotes Tel1-Regulated Chromosomal Translocations
- A Dual Role for SOX10 in the Maintenance of the Postnatal Melanocyte Lineage and the Differentiation of Melanocyte Stem Cell Progenitors
- SLC26A4 Targeted to the Endolymphatic Sac Rescues Hearing and Balance in Mutant Mice
- Odoriferous Defensive Stink Gland Transcriptome to Identify Novel Genes Necessary for Quinone Synthesis in the Red Flour Beetle,
- Prediction of Complex Human Traits Using the Genomic Best Linear Unbiased Predictor
- Gene × Physical Activity Interactions in Obesity: Combined Analysis of 111,421 Individuals of European Ancestry
- Reassembly of Nucleosomes at the Promoter Initiates Resilencing Following Decitabine Exposure
- Exquisite Light Sensitivity of Cryptochrome
- miR-133a Regulates Adipocyte Browning In Vivo
- Strabismus Promotes Recruitment and Degradation of Farnesylated Prickle in Planar Polarity Specification
- Hepatocyte Growth Factor Signaling in Intrapancreatic Ductal Cells Drives Pancreatic Morphogenesis
- Is a Potential Tumor Suppressor Gene Commonly Inactivated by Epigenetic Mechanisms in Colorectal Cancer
- Joint Molecule Resolution Requires the Redundant Activities of MUS-81 and XPF-1 during Meiosis
- The Mating Competence of Geographically Diverse Strains in Their Natural and Unnatural Sand Fly Vectors
- Defective Repair of Oxidative Base Lesions by the DNA Glycosylase Nth1 Associates with Multiple Telomere Defects
- Effective Blocking of the Enhancer Requires Cooperation between Two Main Mechanisms Suggested for the Insulator Function
- Trans-Ancestral Studies Fine Map the SLE-Susceptibility Locus
- PLOS Genetics
- Archiv čísel
- Aktuální číslo
- Informace o časopisu
Nejčtenější v tomto čísle- SLC26A4 Targeted to the Endolymphatic Sac Rescues Hearing and Balance in Mutant Mice
- Bacterial Adaptation through Loss of Function
- The Cohesion Protein SOLO Associates with SMC1 and Is Required for Synapsis, Recombination, Homolog Bias and Cohesion and Pairing of Centromeres in Drosophila Meiosis
- Gene × Physical Activity Interactions in Obesity: Combined Analysis of 111,421 Individuals of European Ancestry
Kurzy
Zvyšte si kvalifikaci online z pohodlí domova
Současné možnosti léčby obezity
nový kurzAutoři: MUDr. Martin Hrubý
Všechny kurzyPřihlášení#ADS_BOTTOM_SCRIPTS#Zapomenuté hesloZadejte e-mailovou adresu, se kterou jste vytvářel(a) účet, budou Vám na ni zaslány informace k nastavení nového hesla.
- Vzdělávání



