-
Články
Top novinky
Reklama- Vzdělávání
- Časopisy
Top články
Nové číslo
- Témata
Top novinky
Reklama- Videa
- Podcasty
Nové podcasty
Reklama- Kariéra
Doporučené pozice
Reklama- Praxe
Top novinky
ReklamaThe RCC1 Family Protein TCF1 Regulates Freezing Tolerance and Cold Acclimation through Modulating Lignin Biosynthesis
Cold acclimation is a well-known adaptive process through which plants can dramatically increase their tolerance to freezing temperature. Modifications of cell wall have been recognized as a key characteristic during plant acclimation to low temperature. However, the molecular mechanism responsible for such cellular adaptation still remains a mystery. Here, we report an unexpected regulatory role of TCF1 on lignin content during cold acclimation in Arabidopsis. TCF1 is specifically induced by cold and is required for chromatin based gene regulation of cold responsive genes such as BCB (a GAP) that regulates lignin genes. Further evidence shows that reduction in lignin dramatically increases plant freezing tolerance, while lignin maintenance required for cold acclimation is regulated by TCF-mediated signaling. Thus, our study has revealed, for the first time, lignin remodeling as a key function of cold acclimation and freezing tolerance. The findings provide the first direct molecular evidence that freezing tolerance is directly related to cell wall properties during cold acclimation and extra/intercellular freezing upon and freezing/thawing process.
Published in the journal: . PLoS Genet 11(9): e32767. doi:10.1371/journal.pgen.1005471
Category: Research Article
doi: https://doi.org/10.1371/journal.pgen.1005471Summary
Cold acclimation is a well-known adaptive process through which plants can dramatically increase their tolerance to freezing temperature. Modifications of cell wall have been recognized as a key characteristic during plant acclimation to low temperature. However, the molecular mechanism responsible for such cellular adaptation still remains a mystery. Here, we report an unexpected regulatory role of TCF1 on lignin content during cold acclimation in Arabidopsis. TCF1 is specifically induced by cold and is required for chromatin based gene regulation of cold responsive genes such as BCB (a GAP) that regulates lignin genes. Further evidence shows that reduction in lignin dramatically increases plant freezing tolerance, while lignin maintenance required for cold acclimation is regulated by TCF-mediated signaling. Thus, our study has revealed, for the first time, lignin remodeling as a key function of cold acclimation and freezing tolerance. The findings provide the first direct molecular evidence that freezing tolerance is directly related to cell wall properties during cold acclimation and extra/intercellular freezing upon and freezing/thawing process.
Introduction
Freezing temperature is an important environmental factor that determines the natural geographical distribution of plants and limits crop productivity [1]. Sudden exposure to freezing temperature causes intracellular freezing, membrane damage and cell death [1–3]. To better survive freezing low temperature, plants have evolved coping mechanisms through initiating cold acclimation when the temperature gradually drops lower in autumn in nature. Many signal transduction cascades are involved in this physiological adaptation process. In Arabidopsis, expression profiling of cold-treated plants revealed that up to 20% of genes in the genome are regulated by cold. Characterization of a group of cold-regulated (COR) genes which are highly induced by cold stress using forward and reverse genetics has led to identification of a key CBF (C-repeat binding factor, also known as dehydration-responsive element-binding protein 1 or DREB1) signaling pathway. CBF transcription factors (CBF1, CBF2, CBF3) can activate expression of the COR genes by binding to cis-elements in their promoters and induce cold acclimation and freezing tolerance [4–6]. Several regulators of CBF genes have been identified, such as Inducer of CBF expression 1 (ICE1), calmodulin binding transcription activator 3 (CAMTA3), MYB15 and Ethylene Insensitive 3 (EIN3) [7–10]. Most recently, it has been shown that OPEN STOMATA 1 (OST1), a central component in ABA signaling pathway, plays a crucial role in plant response to cold. OST1 is induced by cold and cold-activated OST1 can interact and phosphorylate ICE1 to enhance the stability of ICE1, resulting in increased plant tolerance to freezing [11]. However, multiple studies have reported that the CBF signaling pathway is not the sole mechanism modulating plant cold acclimation and cold tolerance, because only 12% of the cold responsive genes are regulated by CBF transcription factors [12]. The prominent example is HOS15, which regulates freezing tolerance through modification of histone acetylation [13,14]. In addition, SFR2 was found to modulate freezing tolerance through lipid remodeling of the outer chloroplast membrane [15]. Most recently, it was found that AtHAP5A modulates freezing stress resistance in Arabidopsis independent of the CBF pathway [16]. Through these signaling pathways, a wide variety of antifreeze/stress-related proteins and compounds are accumulated to minimize intracellular ice formation, to increase tolerance to dehydration caused by water outflow, and to maintain cell membrane stability and integrity that is considered central to the ability of plants to survive freezing [2,15,17–19].
The plant cell wall is the extracellular matrix consisting of cellulose, hemicellulose and lignin. It plays essential roles in plant growth and adaptive responses to adverse environmental conditions [20–22]. The cell wall integrity (CWI) and structures are dynamically regulated during plant development and are capable of being remodeled in response to various environmental stresses [23–26]. Fine-tuning regulation of the proportions and the amounts of each matrix component within the cell wall determines its nature and functions. Remarkably, deposition of lignin, phenylpropanoid polymer, which is highly hydrophobic in the cell wall, determines cell wall stiffness and permeability to water [27–29]. In yeast, the CWI signaling pathway plays a vital role in adjusting the cell wall thickness and composition to environmental cues, in particular freezing temperature and osmotic stress [30,31]. In plants, similar processes are employed for controlling cell wall integrity and performance during development, drought and defense [32], but the precise mechanisms remain unclear.
Previous studies have shown that the expression of genes related to cell wall biosynthesis and remodeling is dramatically altered under cold treatment [33]. Using cryo-scanning electron microscopy (cryo-SEM), several studies have revealed that both cell membrane and cell wall properties play equally important roles in cold acclimation and freezing tolerance [18]. Most strikingly, cell wall thickness and rigidity have been linked to dynamic water heterogeneities during cold acclimation and extra-/inter - or intracellular freezing upon freezing/thawing process. Variations in cell wall rigidity and composition in different types of plant tissues and cells (e.g. xylem, phloem, living fibers and mesophyll cells) showed altered intracellular freezing, tension-induced cavitation and cell viability during freezing/thawing [34–36]. Thus, resistance to freezing temperatures is dependent on the capacity for water outflow from the cells during cold acclimation and freezing and water reabsorption during thawing, on the capacity to accommodate growth of ice crystals in extra-/intercellular spaces, and on the ability of cell wall elasticity to respond to cellular shrinkage.
Investigation of the roles of the cell wall in cold acclimation and freezing/thawing has been limited to correlation of cellular changes in various plant species or different types of tissues and cells using cryo-SEM; mechanistic analysis of the regulatory genes and signaling cascades underlying cell wall mediated water movement and freezing tolerance is lacking. Lignin is a major component of the plant secondary cell wall, and the amounts of lignin are altered after cold treatment in various species [37,38]. In the past decades, some genes that regulate lignin biosynthesis have been identified [39–43]. Among them, Phenylalanine ammonia-lyase 1–4 (PAL; EC 4.3.1.5) encoding the enzymes that catalyze the first step in the phenylpropanoid pathway regulate biosynthesis of lignin and secondary metablites (e.g. flavonoids and salicylic acid) in Arabidopsis thaliana [39,44–48]. Arabidopsis thaliana blue copper binding gene (BCB) is another positive regulator of lignin synthesis, and AtBCB overexpression substantially increases lignin content in Arabidopsis roots [49]. It has been shown that PAL1-PAL4 and BCB genes are responsive to a variety of environmental stimuli, including pathogen infection, wounding, nutrient depletion, UV irradiation, and extreme temperature, etc. [49,50], suggesting their roles in plant stress resistance. However, it remains unknown how these genes mediate plant responses to biotic and abiotic stresses.
Here we report that Tolerant to Chilling and Freezing 1 (TCF1), a gene encoding a RCC1 family protein, is required for chromatin based gene regulation of cold responsive genes in a CBF-independent pathway. Importantly, we reveal that lignin content in leaves is directly related to freezing tolerance, and that TCF1 plays a critical role in the adjustment of lignin accumulation through modulation of expression of BCB and downstream effectors PAL1/3/4 genes during cold acclimation and freezing tolerance in Arabidopsis.
Results
Identification and Characterization of the TCF1 Gene
To identify the genetic loci that regulate specifically plant cold acclimation and freezing tolerance through chromatin condensation and remodeling, we examined cold responses of the genes encoding Regulator of Chromatin Condensation 1 (RCC1) family proteins from AtGenExpress Data [51]. The gene At3g55580 which is specifically responsive to cold was identified (S1A Fig), and designated Tolerant to Chilling and Freezing1 (TCF1) based on the phenotypes of its mutant. RT-PCR analysis and GUS assay of TCF1pro::GUS lines validated induction of TCF1 expression in response to cold but not to osmotic stress or ABA (Fig 1A–1D).
Fig. 1. Analysis of TCF1 expression and nuclear localization of the TCF1 protein. 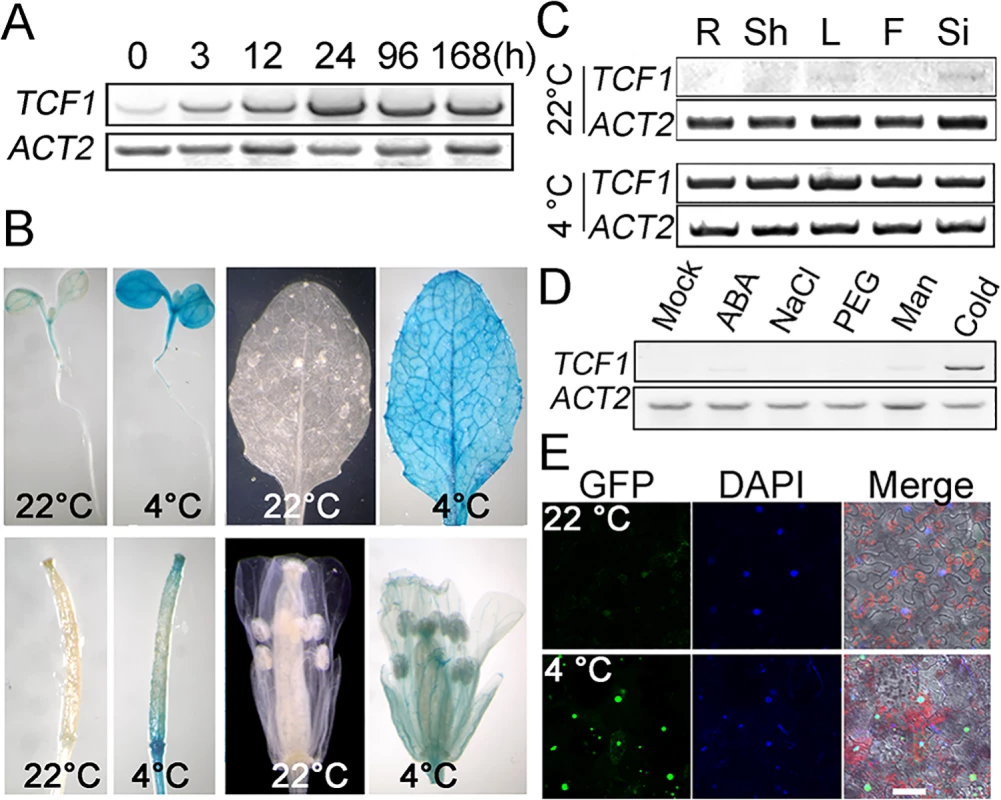
(A) Three-week-old Col-0 (WT) plants were subjected to low temperature (4°C) and the samples were harvested at the indicated time points for semi-quantitative RT-PCR analysis of TCF1 transcripts. ACT2 (At3g18780) was used as a loading control. (B) GUS staining of transgenic plants expressing TCF1pro::GUS under normal temperature or treated at 4°C for 7 days. (C) Semi-quantitative RT-PCR for TCF1 in different tissues with or without cold treatments for 7 days. R, root; Sh, shoot; L, leaves; F, flowers; Si, siliques. (D) Semi-quantitative RT-PCR analysis for TCF1 of three-week-old Col-0 plants treated with 100 μM ABA, 400 mM mannitol, 20% PEG6000, 300 mM NaCl for 3 h and 4°C Cold treatment for 24 h. (E) Localization of fluorescence in tcf1-1 plants expressing a TCF1pro::GFP-TCF1 fusion at 22°C (Upper) and 4°C for 7 days (Bottom). GFP: GFP-TCF1 fusion protein (tcf1-1TCF1-3), DAPI: DAPI staining, Merge: Merger of GFP and DAPI channels (Scale bars, 20 μm). The TCF1 gene encodes a protein containing six predicted tandem RCC1 repeats that shows similarity to RCC1 in yeast and human [52,53] (S1B and S1C Fig). To determine whether TCF1 is localized in the nucleus like RCC1 [54,55], we made translational fusions with GFP and expressed them in tcf1-1 plants using the native promoters. Examination of independent transgenic lines revealed that GFP-TCF1 fluorescence was present in the nucleus (Fig 1E), and the level of the fusion protein GFP-TCF1 was also induced by cold (Fig 1E).
TCF1 Interacts with Histones and Has Negligible GEF Activity in Vitro
RCC1 is a guanine nucleotide exchange factor (GEF) for the small GTP-binding protein Ran. RCC1 is constitutively localized in the nucleus, binds to chromatin, and generates a Ran-GTP/Ran-GDP gradient across the nuclear envelope that is required both to drive nucleo-cytoplasmic transport and to regulate processes associated with progression of the cell cycle and mitosis [54,55]. We then ask whether TCF1 has the similar roles of RCC1. To address whether TCF1 interacts physically with chromatin, an in vitro assay was performed. The fusion protein GST-TCF1 expressed in E. coli bound strongly to a histone agarose column (Fig 2A). To further investigate which histone TCF1 interacts preferentially with, in vitro translated Myc-tagged TCF1 was pre-incubated with each kind of purified core histone in 20-fold excess followed by incubation with histone-agarose. As shown in Fig 2B, H3 and H4 were the histones that can compete effectively to diminish the binding of Myc-TCF1 to histone-agarose, suggesting high affinity binding between TCF1 and histones H3 and H4. Further yeast-two-hybrid results confirmed that TCF1 can indeed interact with the specific histone H4 (HFO2, at5g59690), but not other tested histone H3 and H4s (HTR9, At5g10400; HFO4, At1g07820) (Fig 2C). To exam whether TCF1 has GEF activity, an in vitro assay was performed. The results showed that TCF1 exhibited less than 1% of the GEF activity with human Ran as substrate of that measured for RCC1 (Fig 2D). Also expression of TCF1 did not complement the phenotype of the yeast prp20 mutant lacking yeast RCC1 (S2 Fig). The results indicate that TCF1 is not the ortholog of RCC1 in Arabidopsis, but it does associate with chromatin via its interaction with histones.
Fig. 2. GEF activity assay of TCF1 and analysis of TCF1-histone interactions. 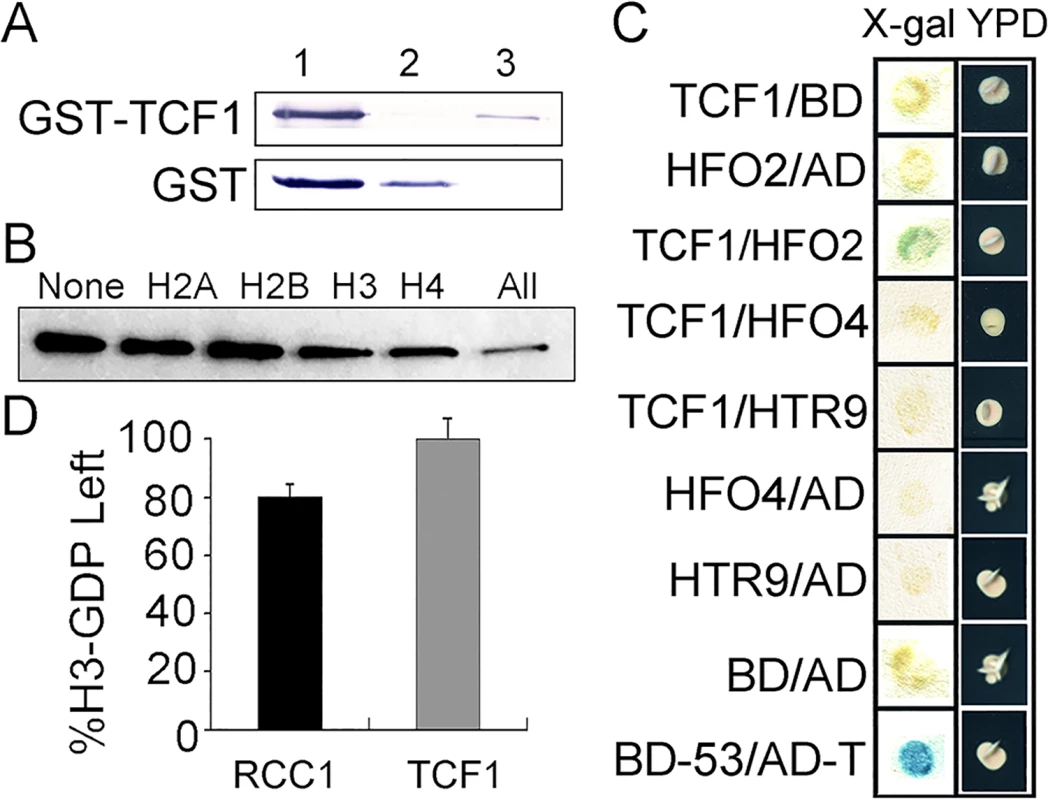
(A) Binding of E. coli-expressed GST-TCF1 to a calf thymus histone-agarose column. Western blots with anti-GST antibody showing GST-TCF1 (77 kDa) or control GST (26 kDa). Lane 1, protein applied to the columns; lane 2, unbound material that flowed through; lane 3, protein bound after a 5 min incubation, eluted with 0.3 M NaCl. (B) 0.3 μg of in vitro translated Myc-TCF1 was incubated with 20 μg of the appropriate histone (20-fold excess) for 30 min at room temperature. The samples were then incubated with histone-agarose overnight at 4°C. Myc-TCF1 binding to histone-agarose was analyzed by Western blot. The different histones used as competitors are indicated at the top of each lane. ‘All’ indicates the mixture of histones and ‘None’ indicates the control without competitors. (C) Yeast co-transformant strains carrying both TCF1 and vector control (TCF1/BD), histone H3/H4 and vector control (HFO2/AD, HFO4/AD and HTR9/AD), TCF1 and H3/H4 (TCF1/HFO2,TCF1/HFO4 and TCF1/HTR9), negative control (BD/AD) and positive control (BD-53/AD-T) were streaked onto selective media. Activation of the lacZ reporter gene is indicated by the formation of blue or blue-green colonies on plates containing X-Gal (left), the growth state of yeast co-transformant strains in YPD medium is shown on the right (right). (D) Ran-GEF activity of E. coli-expressed GST-RCC1 and GST-TCF1 is shown as the percentage of [3H]GDP remaining at the end of the GEF assay. TCF1 Mutant Plants Are Tolerant to Freezing
To verify the function of TCF1 in cold and/or freezing response, tcf1-1 and tcf1-2 were analyzed (Fig 3A and 3B). tcf1-1 with T-DNA insertion at the first exon of TCF1 showed no TCF1 transcript in response to cold treatment (Fig 3B), but tcf1-2 having a T-DNA insertion in the 3’-UTR region of TCF1 (Fig 3A) exhibited similar TCF1 expression to that of the wild-type under cold treatment (Fig 3B). Thus, the phenotypic analysis of tcf1-1 was shown thereafter. All of the F1 plants from tcf1-1×wild-type cross were resistant on MS medium containing 5 mg/L bialaphos. The F2 progeny of the selfed F1 plants segregated in a 3 : 1 ratio (From 3130 plants, 2322 conferring resistant to bialaphos compared with 808 plants showing sensitive phenotype, x2 = 1.11 < 3.841; x2 test with one degree of freedom). Analysis of the bialaphos resistance revealed the presence of a single functional T-DNA that is inserted in the genome of the tcf1-1 mutant. The results indicated that the tcf1-1 mutation is recessive in a single nuclear gene.
Fig. 3. tcf1-1 plants are tolerant to freezing treatment. 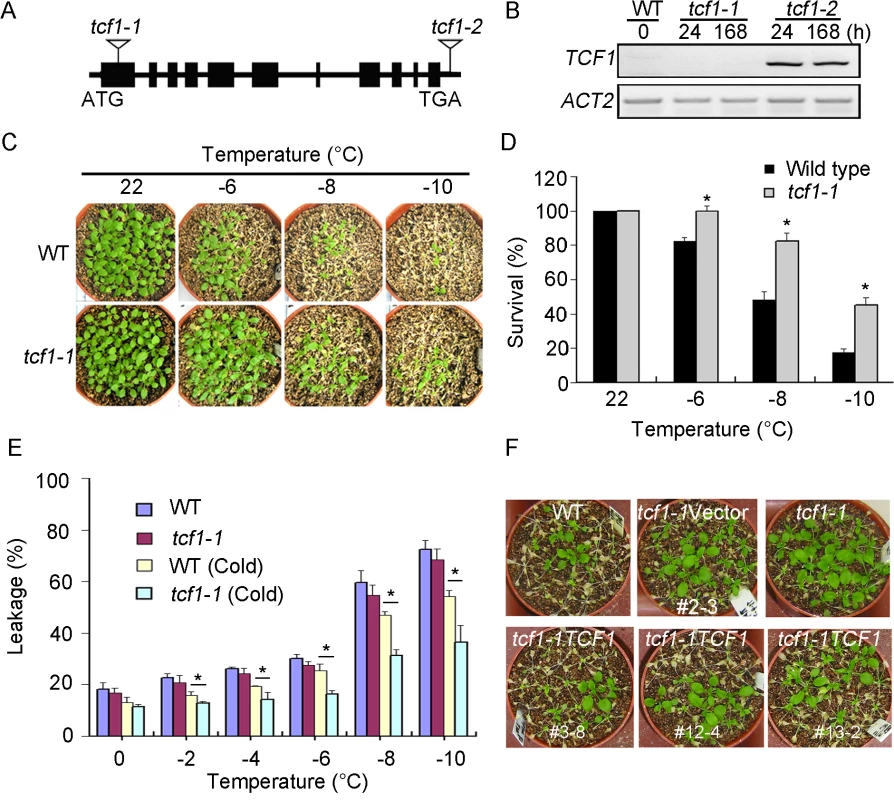
(A) Schematic presentation of the TCF1 gene structure and T-DNA insertions in the TCF1 gene (arrowheads). The closed rectangles represent exons and lines between the exons denote introns. (B) tcf1-1 is a null mutation of TCF1. The levels of TCF1 transcripts were determined by RT-PCR using 3-week-old tcf1-1 seedlings subjected to low temperature (4°C) for the indicated time periods (h); the ACT2 gene was used as loading control. (C) Tolerance of 3-week-old tcf1-1 and wild-type (WT) plants at the indicated temperatures below freezing under long-day photoperiod with cold acclimation for 7 days. The pictures were taken 7 days after treatments. (D) Quantification of survival rate of the treated plants in (C), (*, P < 0.05, t-test). (E) Leakage of electrolytes in tcf1-1 and WT plants treated at indicated temperatures below freezing. WT (cold) and tcf1-1 (cold): 3-week-old plants were cold-acclimated (4°C for 7 day), WT and tcf1-1: both plants were grown under normal conditions. Error bars are standard deviation (n = 8), (*, P < 0.05, t-test). (F) Tolerance of freezing treatments (-8°C for 2 h) of control and transgenic plants, which were cold-acclimated at 4°C for 7 days before the treatment. The plants included WT, tcf1-1, representative homozygous lines of tcf1-1 transformed with an empty vector pEZR(K)LC (tcf1-1Vector-2 (#2–3)) or TCF1 gene (tcf1-1TCF1-3 (#3–8), tcf1-1TCF1-12 (#12–4) and tcf1-1TCF1-13 (#13–2)). To evaluate the effect of the tcf1-1 mutation on freezing tolerance, we performed the whole-plant freezing test. Without cold acclimation, tcf1-1 shows a slightly higher survival rate than wild-type, but there was no significant difference between wild-type and tcf1-1 (S3A Fig). When the plants were acclimated at 4°C for 7 days, 45.2% of the tcf1-1 plants survived freezing temperature as low as -10°C, but only 17.4% of the wild-type plants survived the treatment (Fig 3C and 3D). The electrolyte leakage assay confirmed the freezing tolerance of tcf1-1 plants under cold acclimation (Fig 3E). To verify the role of TCF1 in cold acclimation, we generated TCF1 RNA interference (TCF1-RNAi) lines and two TCF1-RNAi lines (TCF1-RNAi-2 and TCF1-RNAi-6) with reduced expression of TCF1 were used in freezing response assay (S3B Fig). Without cold acclimation, both TCF1-RNAi lines showed a similar survival rate to the wild-type (S3C Fig). When the plants were acclimated at 4°C for 7 days, these TCF1-RNAi lines also displayed significantly higher survival rates than the wild-type plants at -10°C (S3D and S3E Fig). The percentages of electrolyte leakage of TCF1-RNAi lines were also decreased under cold acclimation (S3F Fig), suggesting a role of TCF1 during cold acclimation and freezing tolerance in Arabidopsis.
To further determine that freezing tolerance of tcf1-1 was due to loss of function in TCF1, we expressed the GFP-CDS of TCF1 under the control of its native promoter in the tcf1-1 background (Fig 1E). Three independent transgenic lines (tcf1-1TCF1-3, tcf1-1TCF1-12 and tcf1-1TCF1-13) with increased levels of TCF1 showed a cold-sensitive phenotype compared with tcf1-1 mutant and had a similar response to the wild-type under freezing treatment with cold acclimation (Figs 3F and S4), thereby confirming that expression of TCF1 complemented the freezing tolerance phenotype of the mutant.
TCF1 Regulates Freezing Tolerance through a CBF Independent Pathway
Because TCF1 associates with chromatin, we questioned whether TCF1 regulates freezing tolerance by modulating expression of CBF1-3 and the targeted genes. However, the expression levels and patterns of CBF1-3 under cold treatment in tcf1-1 did not show any significant difference from that in the wild-type (Fig 4A–4C and S5A). No significant changes in transcript levels of TCF1 in cbf2 (S5B Fig) were detected under cold (Fig 4D). In addition, the expression of the CBF regulon genes such as COR15A and COR47 was also not changed in tcf1-1 under cold (Fig 4E and 4F). Notably, TCF1 was not in the list of the CBF1, CBF2 and CBF3 coregulated genes in the previous study [12]. These results indicate that TCF1 may influence cold/freezing tolerance through a mechanism different from the CBF-COR cascade.
Fig. 4. Gene expression analyses of the CBFs and the CBF regulon in the tcf1-1 and wild-type plants. 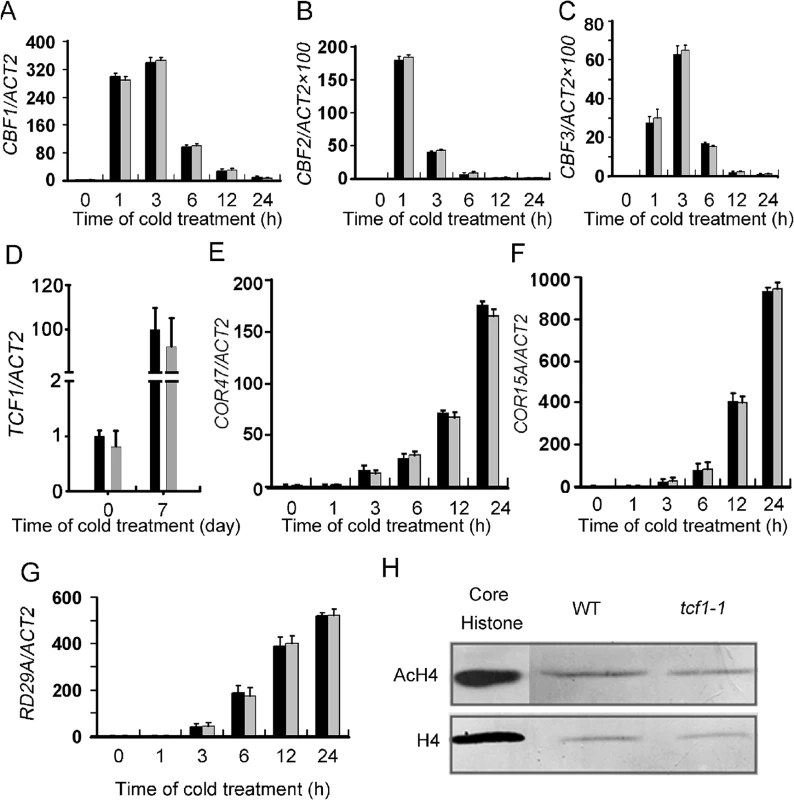
(A) to (C) The levels of CBF1, CBF2 and CBF3 transcripts, respectively, in tcf1-1 and wild-type plants. The plants were treated with cold (4°C) at the indicated time points. (D) The level of TCF1 transcripts in the loss-of-function cbf2 mutant plants grown under normal conditions or given a 4°C treatment for 7 day. Transcript levels of the ACT2 gene were used as a loading control. (E) to (G) Transcript levels of COR47 (E), COR15A (F) and RD29A (G) in tcf1-1 and wild-type plants. The plants were treated with cold (4°C) for the indicated times. (H) Western blot analysis with anti-acetylated H4 antibody (Top) revealed that tcf1-1 and WT plants have similar levels of acetylated H4. Histone H4 antibody for core histone H4 was used as the immunoblot control (Bottom). Because TCF1 reached the highest level of expression and loss of function in TCF1 enhanced freezing tolerance with a 7-day cold acclimation, the expression profiles of tcf1-1 and the wild-type grown under normal condition with a 7-day cold acclimation and without cold acclimation were compared by the GeneChip Array. Under normal conditions, only 20 genes up-regulated or down-regulated (> 1.5 fold) in tcf1-1 were identified and they are annotated to encode proteins with diverse cellular functions (S1 Table). Five genes that should be repressed during cold exposure were activated in tcf1-1, and 12 down-regulated genes are involved in diverse stress response processes. After cold acclimation, expression of 36 genes was varied (13 genes up-regulated and 23 down-regulated) in tcf1-1 (S2 Table). Among them, nineteen genes were cold responsive and are involved in different cell functions, but none of them appears to involve the CBF regulon. The highest and lowest two expression genes in microarray data under cold acclimation were validated by qRT-PCR (S6 Fig). The data supports that TCF1 functions in a novel pathway independent of the CBF-COR cascade.
TCF1 Does Not Affect Histone H4 Acetylation
HOS15 has been reported to function in a CBF-independent pathway to regulate cold acclimation and freezing tolerance [14]. HOS15 interacts with H4 and represses RD29A expression by facilitating H4 deacetylation through association with the RD29A promoter. To determine whether TCF1 functions in the same way as HOS15, RD29A expression was analyzed in tcf1-1 plants under cold treatments. However, there were no changes in RD29A transcript levels detected in tcf1-1 plants under stress treatment (Fig 4G). Importantly, in contrast to hos15 the level of nuclear tetra-acetylated histone H4 in tcf1-1 was similar to that in wild-type plants (Fig 4H). Together with the results of expression of RD29A, which is elevated in hos15 plants in response to cold treatment [14] but not in tcf1-1 (Fig 4G), we conclude that TCF1 functions differently from HOS15.
TCF1 Is Associated with Chromatin Containing the BCB Gene
To investigate whether TCF1 represses or activates the expression of target genes through direct interaction with chromatin, we performed chromatin immunoprecipitation (ChIP) assay using TCF1 complementational line (tcf1-1TCF1-3) that were cold-treated for 7 days. Seven genes (At1g23150, At1g69120, At1g75040, At5g50720, At5g10760, At5g20230 and At2g22500), which were responsive to cold in tcf1-1 in the microarray dataset were selected and three pairs of primers covering the entire genomic sequences of the candidate genes were designed. Using ChIP assay with an anti-GFP antibody, we found that among the tested genes, TCF1 was only associated with a chromatin fragment containing the coding region (BCBc1, CDS sequence +87 to +347) of the (BLUE-COPPER-BINDING PROTEIN) BCB gene (At5g20230), which is cold inducible (Fig 5A), whereas ACT2 whose expression level is not changed in the mutant was not immunoprecipitated (Fig 5B). The results suggest that the association of TCF1 with chromatin at the BCB locus mediates cold-induced and TCF1-regulated expression of BCB gene.
Fig. 5. Chromatin immunoprecipitation assay of chromatin loci associated with TCF1. 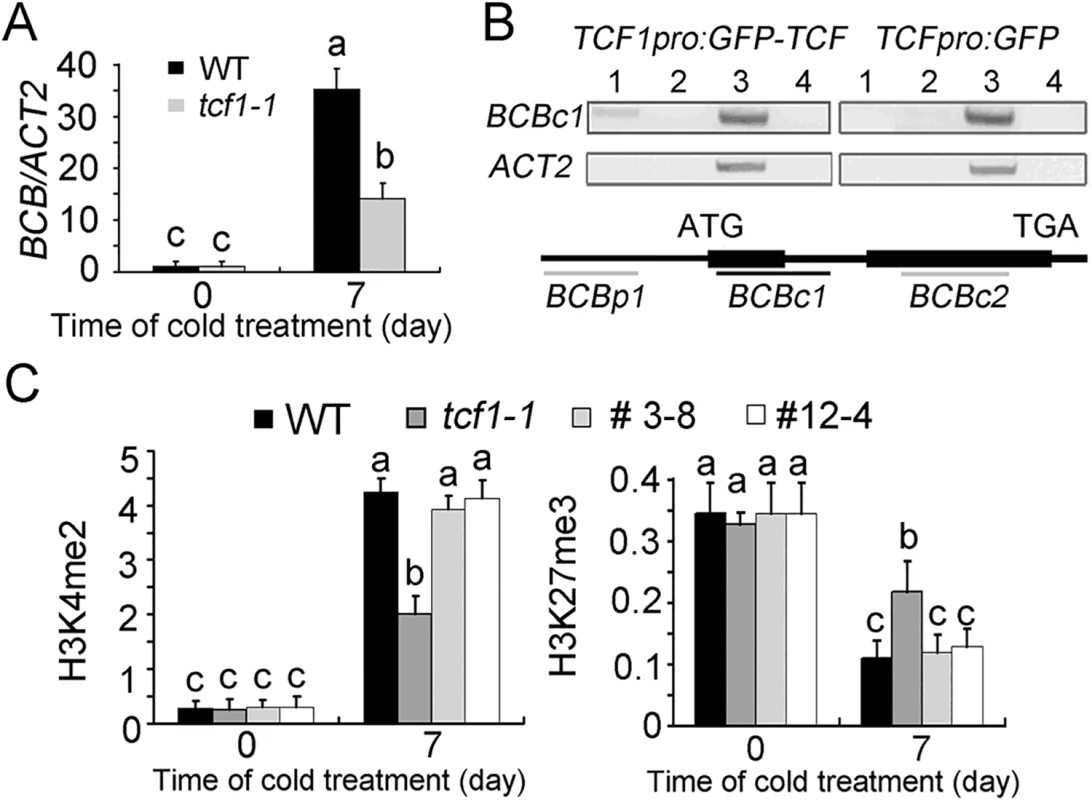
(A) The levels of BCB transcripts in tcf1-1 (Grey) and wild-type (Black) plants. The plants were treated with cold at 4°C for 7 days. Data represent means of three replicates ± SD. Different Letters (P < 0.05; One Way ANOVA) show significant difference from WT. (B) ChIP assay for the association of TCF1 with BCB loci compared to the control ACT2. Lane 1, DNA immunoprecipitated by using anti-GFP antibody; lane 2, no antibody control; lane 3, input DNA before immunoprecipitation; lane 4, PCR without added DNA. TCF1pro::GFP-TCF1 (Complemental line tcf1-1TCF1-3-8, Left) and TCF1pro::GFP (tcf1-1Vector-2-3, Right) transgenic plants treated at 4°C for 4 day were used in the experiments. Below: schematic presentation of the gene structures and the positions of the PCR fragments amplified from the ChIP products. Three fragments covering the genomic sequences of BCB (BCBp1, -664bp-301bp; BCBc2, 87bp-347bp; and BCBc3, 552bp-680bp) were used in the ChIP assay. The experiments were repeated three times; each gave similar data and results from one experiment are shown. (C) Levels of H3K4me2 and H3K27me3 by ChIP-qPCR analysis with WT, tcf1-1 and two TCF1 complementary transgenic lines (tcf1-1TCF1-3 and tcf1-1TCF1-12) plants normalized to ACT2 or AGAMOUS in BCBc1 fragment with and without a 7-day cold treatment; data represent means of three biological replicates ± SD. Means with the same letter are not significantly different at P < 0.05 by One Way ANOVA analysis. Histone Methylation May Be Involved in Cold Regulation of Transcription
To get a better understanding of the relationships between histone modifications and BCB gene expression, we analyzed histone modifications across the BCB locus in non-stressed and stressed wild-type, tcf1-1 and TCF1 complementational lines (Figs 5C and S7A). As histone H3K4me2 has been demonstrated to play widespread roles in activation of gene expression, we first analyzed the H3K4me2 status of the BCB gene. In the wild-type and TCF1 complementation lines, BCB was induced by cold treatment. Accordingly, the level of H3K4me2 increased in the transcribed region of BCB (BCBc1 fragment) after cold treatment (Figs 5C and S7A). Under normal condition, tcf1-1 had comparable level of H3K4me2 to that of the wild-type. When exposed to cold for 7 days, tcf1-1 also exhibited an increase in H3K4me2, but the level of H3K4me2 at the BCB locus was lower than that of the wild-type and TCF1 complementation lines (Figs 5C and S7A).
Trimethylation of histone H3 at lysine 27 (H3K27me3) is a histone mark associated with gene silencing. We then analyzed the status of H3K27me3 at the BCB locus (BCBc1 fragment) in non-stressed and stressed wild-type, tcf1-1 and tcf1-1TCF1 seedlings. In the non-stressed wild-type and tcf1-1TCF1 lines, the level of H3K27me3 was relatively high. However, the level of H3K27me3 was significantly decreased when the seedlings were treated at low temperature for 7 days (Figs 5C and S7A). In sharp contrast, loss of TCF1 function caused an opposite trend in H3K27me3. Under normal condition, the tcf1-1 mutant displayed a similar level of H3K27me3 compared with that of the wild-type and TCF1 complementational lines (Figs 5C and S7A). As expected, the level of H3K27me3 at the BCBc1 region of BCB was higher in mutant compared with wild-type when exposed to low temperature for 7 days. We also tested H3K36me3 and H3K9me2 (for gene repression), H3K14ac and H3K9ac (for gene activation) and AcH4 (global histone H4 tetra-acetylation at K5/K8/K12/K16, which is also associated with gene activation) levels in the BCBc1 fragment and found there were no significant differences between WT and tcf1-1 plants (S7B–S7E Fig). The results indicate that TCF1 may regulate freezing tolerance of plants through modulation of histone modification and subsequent expression of target genes.
Knock-down of the BCB Gene Results in Reduced Lignin Accumulation and Increased Cold Tolerance
To test whether BCB is a functional target of TCF1 in plant response to cold, we used artificial microRNA (amiRNA) method to generate the BCB knock-down transgenic plants. Two transgenic lines with similar reduction in BCB expression to tcf1-1 mutant (named amiR-BCB4-9 and amiR-BCB9-2) (S8 Fig) were used for phenotypic analysis. Three week-old seedlings of the BCB knock-down lines and the wild type control with cold acclimation were treated at -8°C and -10°C for 2 h and were then grown under normal conditions for 7 days. The results showed that the survival rates of the amiR-BCB transgenic lines were markedly increased compared with that of the wild type (Figs 6A and 6B and S9A and S9B). The electrolyte leakage assay revealed that knock-down of BCB significantly reduced the electrolyte leakage from the treated plant cells (Fig 6C), confirming that BCB gene plays an important role in freezing tolerance of Arabidopsis plants during cold acclimation. However, we noticed that the average survival rate for the amRNAi-BCB lines (about 35%) is significantly lower than the tcf1-1 (45.5%) under -10°C treatment (Figs 3D and 6B), indicating that in addition to BCB, alteration of other genes may also contribute to the freezing tolerance of tcf1-1.
Fig. 6. amiRNA-BCB plants are tolerant to freezing treatment. 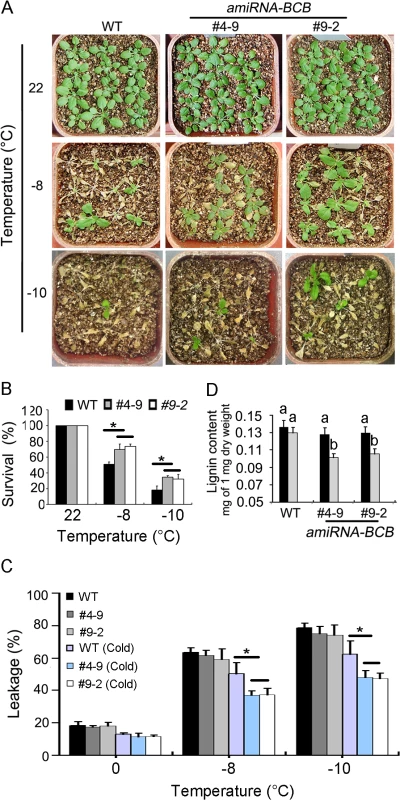
(A) Freezing treatment of 3-week-old amiRNA-BCB transgenic plants (#4–9 and #9–2) and wild-type (WT) plants at the indicated temperature below freezing under long-day photoperiod with cold acclimation. The pictures were taken 7 days after treatments. (B) Quantification of survival of the plants in (A). Error bars are standard deviation (n = 80–100), (*, P < 0.05, t-test). (C) Leakage of electrolytes in amiRNA-BCB transgenic plants and WT plants treated at indicated temperature below freezing. Error bars are standard deviation (n = 8). (*, P < 0.05, t-test). (D) Quantitative determination of lignin content from whole rosettes of three-week-old WT and amiRNA-BCB transgenic plants grown in soil with (Grey) or without (Black) cold treatment (4°C for 7 days). Twelve independent experiments were performed and the data are expressed as mean ± S.E. Means with the same letter are not significantly different at P < 0.05 by One Way ANOVA analysis. The immediate question is how BCB regulates plant freezing tolerance. Since a previous study has shown that overexpression of BCB results in increased lignin accumulation in Arabidopsis [49], we attempted to test whether BCB regulates plant freezing tolerance through modification of lignin content of plants. To this end, we analyzed the lignin levels of 3-week-old seedlings of the amiRBCB transgenic lines and the wild-type that were treated at 4°C for 7 days. As shown in Fig 6D, the lignin content of the wild type seedlings was not affected by cold treatment, by contrast, the lignin levels of the amiRBCB transgenic lines was significantly reduced compared with WT under cold treatment (Fig 6D). The results suggest that BCB is responsible for maintaining steady lignin content in Arabidopsis plants under cold stress conditions. Taken together, these results indicate that TCF1 may modulate freezing tolerance through a BCB-dependent mechanism that positively regulates lignin biosynthesis.
TCF1 and BCB Modulate PALs Genes Expression to Affect Lignin Accumulation under Cold Acclimation
To investigated whether the genes responsible for lignin biosynthesis showed any differential expression in tcf1 in response to cold treatment. The transcript levels of PAL genes, which encode isoforms of a key enzyme Phe ammonia lyase involved in lignin biosynthesis were analyzed. In wild-type seedlings, the transcript levels of PAL1 and PAL3 remained not changed after cold treatment, but the PAL2 and PAL4 gene was induced by cold treatment (Fig 7A and 7B). In the tcf1-1 mutant, expression of PAL1-4 was similar to that of wild type under normal condition, but PAL1, PAL3 and PAL4 exhibited significantly decreased transcript accumulation after cold treatment. PAL2 showed a similar level of transcript in the non-stressed and stressed tcf1-1 mutant to that of the wild-type (Fig 7A and 7B).
Fig. 7. Expression analysis of PAL genes in tcf1 and amiRNA-BCB mutants. 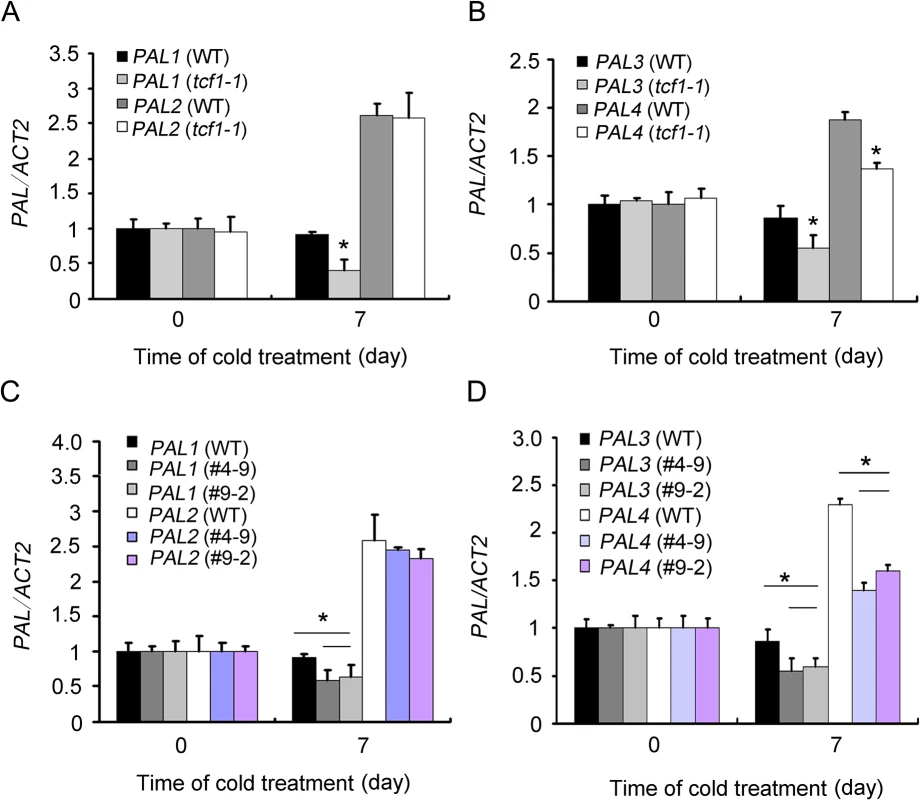
(A) to (D) Quantitative RT-PCR analysis of the expression of PALs in three-week-old WT, tcf1-1 and amiRNA-BCB plants grown in soil with or without cold treatment (4°C for 7 days). Three independent experiments were performed and the data are expressed as mean ±.SE. (*, t-test, P < 0.05). We also checked the PALs expression in amiRNA-BCB transgenic lines, we found that PAL2 showed a similar level of transcript in the non-stressed and stressed amiRNB-BCB mutant to that of the wild-type, but PAL1, PAL3 and PAL4 exhibited significantly decreased transcript accumulation after cold treatment in the amiRNB-BCB mutant (Fig 7C and 7D).These results suggest that TCF1 and BCB positively regulates lignin content in rosettes under low temperature and that this may be due to regulation of genes involved in lignin biosynthesis.
Reduced Lignin Content in tcf1-1 Results in Freezing Tolerance Phenotype
To test whether TCF1 regulates plant freezing tolerance through modulating lignin biosynthesis, we measured the lignin contents in tcf1 and wild-type plants with cold treatments. As expected, at the end of a 7-day cold treatment, the tcf1 rosettes accumulated a significantly lower level of lignin compared with the wild-type (Fig 8A), suggesting a role of TCF1 in lignin biosynthesis.
Fig. 8. Reduced lignin content increased freezing tolerance. 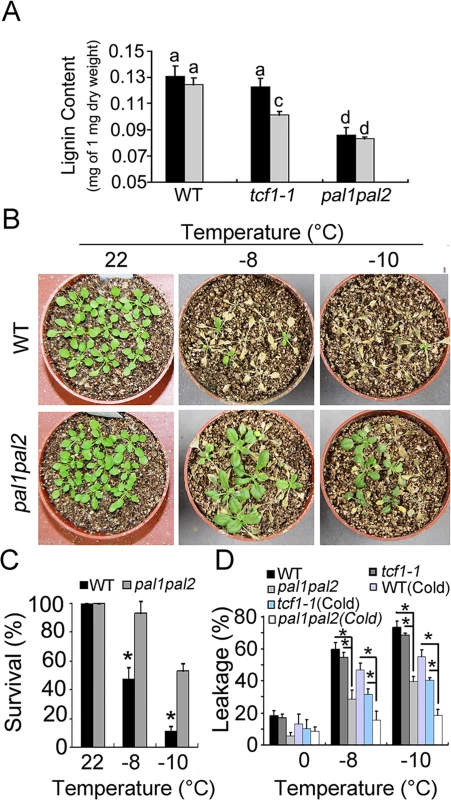
(A) Quantitative determination of lignin content from whole rosettes of three-week-old WT and tcf1-1 plants grown in soil with (Grey) or without (Black) cold treatment (4°C for 7 days). Twelve independent experiments were performed and the data are expressed as mean ±S.E. Means with the same letter are not significantly different at P < 0.05 by One Way ANOVA analysis. (B) Three week-old pal1pal2 and wild type (WT) plants with or without a 7-day cold treatment at 4°C were used for freezing treatments at indicated time point. The pictures were taken 7 days after treatments. (C) Quantification of survival of the wild type (Black) and pal1pal2 plants (Grey) in (B); Error bars represent standard deviation (n = 80–100). (*, t-test, P < 0.05). (D) Leakage of electrolytes in pal1pal2 and wild type plants treated (see experimental procedures) at indicated time point with and without cold acclimation. The experiments were repeated 3 times. Error bars are standard deviation (n = 6). (*, t-test, P < 0.05). As the loss of function in TCF1 causes reduced lignin content and increased freezing tolerance, we proposed that under freezing temperature, reduced lignin may protect plant cells from freezing stress. To test this possibility, we analyzed the freezing tolerance of the pal1pal2 (pal1-2/pal2-2) double mutant with or without cold acclimation. Under normal condition, the pal1pal2 rosettes contained the lowest level of lignin reduction compared with the wild-type and tcf1-1 mutant (Fig 8A). The substantial low level of lignin in the pal1pal2 double mutant observed in this study is consistent with previous reports [56]. Based on our hypothesis, the less lignin the plants accumulate, the higher freezing tolerance they have. Similar sized wild-type and pa11pal2 seedlings were subjected to freezing treatment with or without cold acclimation. As expected, the pal1pal2 mutant displayed the highest freezing tolerance (Fig 8B–8D and S10A and S10B). The stronger effect of pal1pal2 mutant on freezing tolerance than tcf1 and wild-type suggests that the reduced lignin level is correlated to plant freezing tolerance.
Discussion
The present study reports isolation and functional characterization of a novel nuclear protein TCF1 as a determinant of cold acclimation and freezing tolerance. We show that TCF1 specifically activated by cold associates with chromatin and regulates a specific set of genes that are involved in cold acclimation and adaptation to freezing temperature. Importantly, TCF1 regulates lignin accumulation during acclimation/freezing and affects plant freezing tolerance via a CBF independent pathway. Thus, our study not only identifies a novel TCF1-mediated signaling cascade that plays a key role in cold acclimation and freezing tolerance, but also reveals a critical role of cell wall remodeling, in particular lignin homeostasis in cell wall in protecting cells from freezing damage.
TCF1 specifically responded to cold stress via both transcript and protein accumulation (Fig 1), and tcf1-1 plants displayed specific enhancement of freezing tolerance after cold acclimation (Fig 3C and 3D). These results suggest that TCF1 is functions as a negative regulator in cold acclimation and freezing tolerance in Arabidopsis. Recently, extensive attention has been paid to the CBF signaling pathway, however, several studies have shown that the CBF signaling pathway is not the sole mechanism modulating plant cold acclimation and cold tolerance, because at least 28% of the cold-responsive genes were not regulated by the CBFs [12]. TCF1 may act independently of the CBF-COR signaling pathway, because expression of the CBF genes and CBF-regulated genes was unaltered in tcf1-1 (Fig 4A–4C, S1 and S2 Tables), and TCF1 expression remained unchanged in cbf2 (Fig 4D) and in the CBFs-overexpression plants [12]. The TCF1-mediated pathway is also distinct from HOS15, because in tcf1-1, cold induction of RD29A and the level of nuclear tetra-acetylated histone H4 were not affected (Fig 4G and 4H). Thus, we conclude that TCF1 regulates plant cold acclimation and tolerance through at least one additional regulatory pathway. Further genetic analysis will be necessary to determine the genetic relationship between TCF1 and CBFs under cold acclimation and freezing tolerance.
TCF1 belongs to a family of RCC1-like proteins in Arabidopsis. RCC1 functions as the GEF for the small G-protein Ran and is critical for maintaining the RanGTP/RanGDP gradient across nuclear envelope [57]. Our data show that unlike RCC1, TCF1 had very low Ran-GEF activity (Figs 2D and S2), indicating that TCF1 is not the ortholog of RCC1 in Arabidopsis. However, TCF1 shares several important features with UVR8, another RCC1 family protein: very low GEF activity, function in nucleus (Fig 1E) and histone/chromatin association (Fig 2A–2C), although UVR8 preferentially interacts with histone H2B [40,58]. UVR8 regulates plant responses specifically to UV-B through modifying expression of HY5 [40]. Thus, we hypothesized that TCF1 may regulate cold acclimation and freezing tolerance through a similar regulatory mechanism. Indeed, we found that TCF1 directly interacted with the coding regions of BCB (Fig 5B). Further we show that activity of BCB correlates with the enrichment of the positive mark H3K4me2 and reduction of the repressive mark H3K27me3 as cold acclimation is initiated (Figs 5C and S7A). Most importantly, TCF1 is required to modulate levels of both H3K4me2 and H3K27me3 at the BCB locus and regulate BCB transcription (Figs 5C and S7A). Therefore, H3K4me2 and H3K27me3 appear to synergistically regulate transcription activation of BCB, pointing to a critical role of active and repressive marks in cold acclimation.
It is clear that cold activates TCF1 to induce or repress a set of target genes through a chromatin based mechanism. Interestingly, BCB encodes a Blue Copper Binding protein, which is a glycosylphosphatidylinositol-anchored protein (GAP) targeted to the cell surface [59,60] and seems to be responsible for lignin accumulation and cell wall-based resistance to aluminum and bacteria [49,61]. Therefore, our data support the hypothesis that TCF1 regulates cold acclimation and freezing tolerance through modulating BCB to adjust lignin accumulation and consequent cell wall remodeling (Fig 6). We observed a TCF1 dependent reduction in BCB expression and lignin content in rosette leaves of tcf1-1 during cold acclimation (Fig 8A). Importantly, we found that expression of PAL1, PAL3 and PAL4 was reduced under cold in tcf1-1 and BCB knock-down transgenic lines (Fig 7), suggesting that the transcriptional activity of the genes in leaves is also influenced by TCF1 and BCB. Our data reveals that TCF1 directly influences BCB activity and affects PAL1, PAL3 and PAL4 expression and lignification, although it is unclear whether BCB directly regulates PAL genes and lignification or whether TCF1 mediated alteration of chromatin state during cold acclimation indirectly affects PALs expression. Therefore, our data define TCF1 as a key factor able to bind to chromatin and epigenetically regulate the cold-specific GAP leading to establishment of cold specific transcriptional programs and consequently lignin and extracellular matrix remodeling. Although BCB level was almost the same in tcf1-1 and amiRNA-BCB lines, tcf1-1 showed higher survival rate than amRNAi-BCB lines under freezing treatments (Figs 3D and 6B), pointing out that BCB was not the only gene to involve in TCF1-mediated freezing tolerance. Further identification of the target gene(s) of TCF1 will help us to uncover the molecular mechanism underlying TCF1-mediated plant cold acclimation and freezing tolerance. Since UVR8 can also modulate plant response to UV-B through direct interaction with COP1 [62]. We do not exclude the possibility that TCF1 regulates cold acclimation and freezing through association with the key regulator(s) in the cold signal transduction pathway.
It is well known that lignin fills the spaces in the cell wall to reduce water permeability and increase the stiffness of the cell wall [45,50]. Extensive cellular studies have shown that freezing tolerance is directly related to cell permeability and cell wall properties, in particular lignin content, so that water outflows and ice forms in the extracellular spaces without damaging cellular structures [18,35,63–65]. Although it is still technically difficult to measure the cell wall permeability of plants during cold acclimation, the important role of lignin in plant cold acclimation and freezing tolerance has been well documented. For example, the freezing tolerant Miscanthus contains lower lignin content and higher PAL gene expression than the freezing sensitive ecotype [66]. Therefore, it is conceivable that lignin content is closely related to the cell wall permeability and freezing tolerance. However, the molecular mechanism by which lignin content is regulated during cellular adaptation to low temperature still remains a mystery. Here we demonstrate that during cold acclimation tcf1-1, amiRNA-BCB and pal1pal2 had increased freezing tolerance which correlates with their reduced lignin contents (Figs 6 and 8). Therefore, reduction of lignin deposition within the cell wall of the tcf1-1, amiRNA-BCB and pal1pal2 plants during acclimation and freezing may increase cell wall permeability and protect the cells from freezing damage. Reduction of lignin may also enhance elasticity of the cell wall to increase the capacity to accommodate growth of ice crystals with less damage to both the dehydrated cell and cell wall. Together, our data reveal a novel regulatory mechanism in cold acclimation and freezing tolerance in Arabidopsis that involves chromatin based regulation of lignification and cell wall remodeling.
The immediate question is what the biological significance of TCF1 induction is because absence of TCF1 confers freezing tolerance. It is known that cold hardy (freezing tolerant) plants frequently employ extracellular freezing to cope with the freezing temperature. Arabidopsis Col-0 is not hardy plant although it has moderate freezing tolerance compared with other ecotypes [67]. It is possible that TCF1-mediated signaling is activated to maintain lignin content of cells that can enhance cell rigidity and reduce cell expansion, which is required for plant growth arrest under low temperature. Thus, it is conceivable that TCF1-mediated signaling modulates plastic development of the plants during cold acclimation, but not freezing tolerance. A mechanistic working model is presented in Fig 9. The fact that absence of TCF1 enhances freezing tolerance of Col-0 plants suggests that low expression or TCF1 absence may be related to plant freezing tolerance. Thus, our study also identifies a new gene that can be used for genetic improvement of plant freezing tolerance. It will be interesting to see whether TCF1 gene acts differently in cold hardy plants. Given that natural variation in TCF1 may contribute substantially to cold acclimation and freezing tolerance among Arabidopsis accessions, the role of TCF1 expression and its mediated signaling in cold acclimation and freezing tolerance is worthy of special focus. Furthermore, further study will also help to differentiate the molecular mechanisms in plant cold acclimation and freezing tolerance.
Fig. 9. A proposed model for TCF1 in the Arabidopsis response to low temperature. 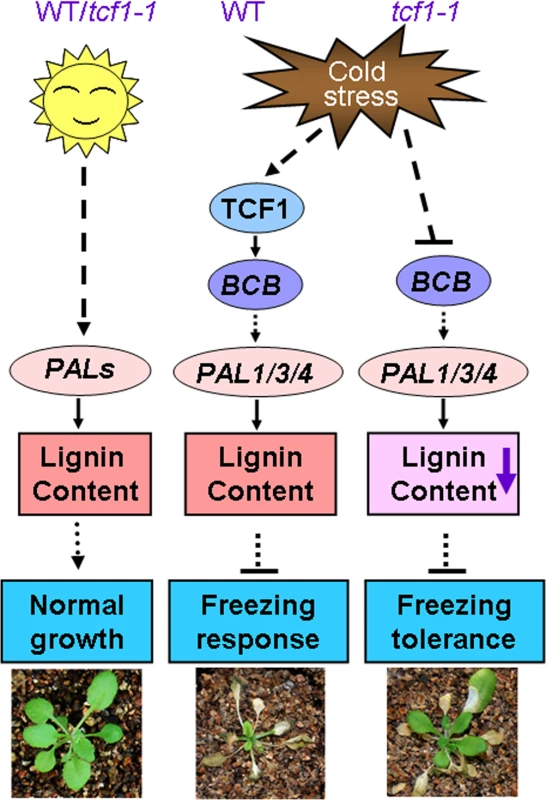
Under normal condition, PAL genes are modulated by the developmental signals to synthesize lignin that favor optimal plant growth and water transport. During cold acclimation, TCF1 is rapidly induced to activate BCB transcription and then stimulates expression of PAL1/3/4 genes to maintain lignin accumulation of the stressed cells. However, when the plants are exposed to freezing temperature, reduction of lignin deposition within the cell wall of the tcf1-1 plants may increase cell wall permeability and protect the cells from freezing damage. Reduction of lignin may also enhance elasticity of the cell wall to increase the capacity to accommodate growth of ice crystals with less damage to both the dehydrated cell and cell wall which is required for plant growth arrest. Lines with arrowheads denote direct regulation, and lines with blunt heads represent indirect regulation. Materials and Methods
Plant Materials and Freezing Treatments
A. thaliana (L.) Heynh Col-0 and tcf1-1 (SAIL_97_D05) and tcf1-2 (SAIL_749_A09) from ABRC were germinated on MS medium containing 2% sucrose, pH 5.7. Three-week-old tcf1-1 and wild-type plants grown in soil with cold-acclimation (4°C for 7 days) and non-cold acclimation were used for whole plant freezing tolerance tests and an electrolyte leakage assay (ELA) as previously described [13]. Briefly, the plants were grown in soil under a long-day photoperiod (16 h light/8 h dark) for 3 weeks in a growth chamber (22°C), and then placed in a temperature chamber (ZSP-A0160, ZHCHENG) at -6°C, -8°C or -10°C for 2 hours. Freezing tolerance was determined as the capacity of plants to resume growth after 7 days under control conditions. For ELA, the rosettes were placed in a temperature chamber starting at 0°C. The temperature was reduced by 2°C after 30 min and maintained for 1 h. Then an identical timing sequence was used for successive 2°C decreases until -10°C was reached. The percentage of EL was calculated as the percentage of the conductivity before autoclaving over that after autoclaving.
Gene Expression and Microarray Analysis
Total RNA was extracted from plants’ leaves by using the TRizol Reagent. First-strand cDNA synthesis was performed according to standard procedures using reverse transcriptase (Promega, 18064–014) following the manufacturer’s instructions. Semi-quantitative RT-PCR and qRT-PCR were done as described previously [68]. A positive control was provided by a parallel analysis based on the ACT2 gene, and three independent replicates were performed per experiment. Gene-specific primers for CBF1, CBF2, CBF3 etc are shown in S3 Table.
For Microarray analysis, 3-week-old plants were treated with or without low temperature at 4°C for 7 day, 2 μg of total RNA was used to produce cyanine dye-tagged cRNA (cy5-WT, cy3-tcf1) and was hybridized to JingXin Array (CapitalBio Company) containing 29K Arabidopsis transcripts. Three biological replicates with 6 Microarray slides were used to check differential expression genes. Data from the GeneChip arrays were scanned on a GeneChip Scanner 3000 and analyzed using GeneChip Operating software (GCOS 1.4). The Significant Analysis of Microarray software (SAM) was used to identify significantly differentially expressed genes between tcf1 and WT groups. Different genes were determined to be significantly differentially expressed with a selection threshold of false discovery rate, FDR = 5% and fold change > 1.5 in the SAM output result. The Microarray data had submitted to GEO (Accession number: GSE70682) at NCBI website.
Plasmid Constructs and Transformation
For histochemical analysis of TCF1 expression, a genomic fragment including 1,337 bp upstream of the translation initiation codon was amplified by PCR and cloned into the binary vector pCAMBIA1391 between the HindIII and BamHI sites. β-glucuronidase (GUS) activity was assayed as previously described [68]. To make the TCF1pro::GFP-TCF1 fusion, the TCF1 CDS was first amplified by PCR and cloned in-frame into the binary vector pEZR(K)-LC [40] between the EcoRI and SalI sites, and then the 35S promoter was replaced by the 1,337 bp TCF1 promoter between the HindIII and SacI sites to generate TCF1pro::GFP-TCF1. The construct was introduced into wild-type and/or tcf1-1 plants through A. tumefaciens-mediated transformation. At least three independent homozygous T3 lines were tested for TCF1pro::GUS expression, protein subcellular localization, and gene expression analysis.
Yeast Two-Hybrid Assays
The TCF1 coding region was amplified by PCR and cloned in-frame between the EcoRI and BamHI sites of pGBKT7. HTA1 [histone H2A (AT5g54640)] and HTR9 [histone H3 (AT5g10400)] coding regions were amplified and cloned in-frame into pGADT7 between the NdeI and XhoI sites to generate prey constructs. pACT2-HTB1 (histone H2B) and pACT2-HFO1 (histone H4) were from Dr. R. A. Bressan at Purdue University. For analysis of specific histone H4 interaction with TCF1, TCF1 coding region was cloned in-frame between the EcoRI and BamHI sites of pGADT7, another two histone H4 variants (At5g59690 and At1g07820) with high expression in tcf1-1 were introduced into pGBKT7 at EcoRI and SalI sites. Plasmid DNA of bait and prey constructs was transformed into the S. cerevisiae strain Y190. Individual transformants were streaked on plates containing a synthetic, minimal (SD) medium lacking tryptophan and leucine and grown for 24 h. Yeast cells were transferred onto a filter paper, and β-galactosidase (β-gal) filter assays were performed [14].
Determination of Histone Acetylation
Nuclei were isolated as described by [14]. Twenty micrograms of nuclear protein and 1 μg of purified core histones from chicken (Upstate Biotechnology, 13–107) were separated by SDS/PAGE and blotted onto a PVDF membrane (Millipore, GVPPEAC12). Anti-tetra-acetylated-histone H4 (1 : 1,000) or anti-histone H4 (1 : 100,000) (Upstate Biotechnology) primary antibodies were used to detect acetylated and unacetylated histone H4, bands were visualized by using the BCIP/NBT Kit (Invitrogen). Data shown are representative of six independent experiments.
GEF Activity Assays, Histone Interaction and Histone Competition
GEF activity assay was performed as described in [40]. Briefly, RCC1, TCF1, and human Ran were expressed in Escherichia coli as fusions with GST. The Ran clone was provided by Dr. Murray Stewart (Medical Research Council Laboratory for Molecular Biology, Cambridge, U.K.). Assays of guanine nucleotide exchange activity were performed by using [3H]GDP to load 30 pmol GST-Ran and subsequent incubation with 0.5 nM recombinant RCC1 or TCF1 for 3 min. The exchange activity was calculated as ln (Ct/C0), where C0 and Ct are radioactive counts at the start and end of the reaction, respectively. The GEF assays were repeated four times. Analysis of TCF1-histone interaction was performed as previously described [40]. Competition assays between TCF1 and histones were conducted as previously described by Cloix and Jenkins [58].
Chromatin Association
Chromatin was isolated and the chromatin immunoprecipitation assay was carried out [40,69] by using an anti-GFP antibody (Invitrogen A-11122). Before antibody treatment, the samples were precleared with protein A Dynabeads (Dynal Biotech, Great Neck, NY, 100.02). The immunoprecipitated DNA was used in PCR reactions to amplify fragments from the BCB and ACT2 genes, using primers shown in S3 Table. Data shown are representative of three independent experiments.
Lignin Analysis
Cell wall fractions were isolated as described by Tokunaga et al [70]. Briefly, three-week-old plants at rosette stage were ground in liquid nitrogen, and then washed with 95% ethanol and ethanol:hexane (1 : 2, v/v) in turn. The washed pellet was allowed to air-dry at 70°C overnight. Lignin content was measured according to the method of Fukuda and Komamine [71] with some modifications. Five mg of the air-dried samples suspended in a 1 ml aliquot of 25% acetyl bromide in acetic acid were treated at 70°C for 30 min. After cooling down, 0.9 ml of 2 M NaOH, 5 ml of acetic acid, 0.1 ml of 7.5 M hydroxylamine hydrochloride, and 3 ml of glacial acetic acid were added. The 10 ml samples were centrifuged and the absorbance of the supernatant was measured at 280 nm to determine the lignin content.
Supporting Information
Zdroje
1. Thomashow MF (1999) Plant cold acclimation: freezing tolerance genes and regulatory mechanisms. Annu Rev Plant Biol 50 : 571–599.
2. Steponkus PL (1984) Role of the plasma membrane in freezing injury and cold acclimation. Ann Rev Plant Physiol 35 : 543–584.
3. Uemura M, Joseph RA, Steponkus PL (1995) Cold acclimation of Arabidopsis thaliana (effect on plasma membrane lipid composition and freeze-induced lesions). Plant Physiol 109 : 15–30. 12228580
4. Stockinger EJ, Gilmour SJ, Thomashow MF (1997) Arabidopsis thaliana CBF1 encodes an AP2 domain-containing transcriptional activator that binds to the C-repeat/DRE, a cis-acting DNA regulatory element that stimulates transcription in response to low temperature and water deficit. Proc Natl Acad Sci USA 94 : 1035–1040. 9023378
5. Jaglo-Ottosen KR, Gilmour SJ, Zarka DG, Schabenberger O, Thomashow MF (1998) Arabidopsis CBF1 overexpression induces COR genes and enhances freezing tolerance. Science 280 : 104–106. 9525853
6. Novillo F, Alonso JM, Ecker JR, Salinas J (2004) CBF2/DREB1C is a negative regulator of CBF1/DREB1B and CBF3/DREB1A expression and plays a central role in stress tolerance in Arabidopsis. Proc Natl Acad Sci USA 101 : 3985–3990. 15004278
7. Chinnusamy V, Ohta M, Kanrar S, Lee BH, Hong X, et al. (2003) ICE1: a regulator of cold-induced transcriptome and freezing tolerance in Arabidopsis. Genes Dev 17 : 1043–1054. 12672693
8. Agarwal M, Hao Y, Kapoor A, Dong CH, Fujii H, et al. (2006) A R2R3 type MYB transcription factor is involved in the cold regulation of CBF genes and in acquired freezing tolerance. J Biol Chem 281 : 37636–37645. 17015446
9. Doherty CJ, Van Buskirk HA, Myers SJ, Thomashow MF (2009) Roles for Arabidopsis CAMTA transcription factors in cold-regulated gene expression and freezing tolerance. Plant Cell 21 : 972–984. doi: 10.1105/tpc.108.063958 19270186
10. Shi Y, Tian S, Hou L, Huang X, Zhang X, et al. (2012) Ethylene signaling negatively regulates freezing tolerance by repressing expression of CBF and type-A ARR genes in Arabidopsis. Plant Cell 24 : 2578–2595. doi: 10.1105/tpc.112.098640 22706288
11. Ding Y, Li H, Zhang X, Xie Q, Gong Z, et al. (2015) OST1 Kinase Modulates Freezing Tolerance by Enhancing ICE1 Stability in Arabidopsis.http://www.sciencedirect.com/science/article/pii/S1534580714008442-cor1 Dev Cell mailto:yangshuhua@cau.edu.cn32 : 278–289.
12. Fowler S, Thomashow MF (2002) Arabidopsis transcriptome profiling indicates that multiple regulatory pathways are activated during cold acclimation in addition to the CBF cold response pathway. Plant Cell 14 : 1675–1690. 12172015
13. Zhu J, Shi H, Lee BH, Damsz B, Cheng S, et al. (2004) An Arabidopsis homeodomain transcription factor gene, HOS9, mediates cold tolerance through a CBF-independent pathway. Proc Natl Acad Sci USA 101 : 9873–9878. 15205481
14. Zhu J, Jeong JC, Zhu Y, Sokolchik I, Miyazaki S, et al. (2008) Involvement of Arabidopsis HOS15 in histone deacetylation and cold tolerance. Proc Natl Acad Sci USA 105 : 4945–4950. doi: 10.1073/pnas.0801029105 18356294
15. Moellering ER, Muthan B, Benning C (2010). Freezing tolerance in plants requires lipid remodeling at the outer chloroplast membrane. Science 330 : 226–228. doi: 10.1126/science.1191803 20798281
16. Shi H, Ye T, Zhong B, Liu X, Jin R, Chan Z (2014) AtHAP5A modulates freezing stress resistance in Arabidopsis through binding to CCAAT motif of AtXTH21. New Phytol 203 : 554–567. doi: 10.1111/nph.12812 24739069
17. Xin Z, Browse J (2000) Cold comfort farm: the acclimation of plants to freezing temperatures. Plant Cell Environ 23 : 893–902.
18. Yamada T, Kuroda K, Jitsuyama Y, Takezawa D, Arakawa K, Fujikawa S (2002) Roles of the plasma membrane and the cell wall in the responses of plant cells to freezing. Planta 215 : 770–778. 12244442
19. Thalhammer A, Bryant G, Sulpice R, Hincha DK (2014). Disordered cold regulated15 proteins protect chloroplast membranes during freezing through binding and folding, but do not stabilize chloroplast enzymes in vivo. Plant Physiol 166 : 190–201. doi: 10.1104/pp.114.245399 25096979
20. Hématy K, Cherk C, Somerville S (2009). Host-pathogen warfare at the plant cell wall. Curr Opin Plant Boil 12 : 406–413.
21. Szymanski DB, Cosgrove DJ (2009) Dynamic coordination of cytoskeletal and cell wall systems during plant cell morphogenesis. Curr Biol 19 : 800–811.
22. Denness L, McKenna JF, Segonzac C, Wormit A, Madhou P, et al. (2011) Cell wall damage-induced lignin biosynthesis is regulated by a Reactive Oxygen Species-and Jasmonic Acid-dependent process in Arabidopsis. Plant Physiol 156 : 1364–1374. doi: 10.1104/pp.111.175737 21546454
23. Temple BRS, Jones AM (2007) The plant heterotrimeric G-protein complex. Annu Rev Plant Biol 58 : 249–266. 17201690
24. Seifert GJ, Blaukopf C (2010) Irritable walls: the plant extracellular matrix and signaling. Plant Physiol 153 : 467–478. doi: 10.1104/pp.110.153940 20154095
25. Tsang DL, Edmond C, Harrington JL, Nühse TS (2011) Cell wall integrity controls root elongation via a general 1-Aminocyclopropane-1-Carboxylic Acid-dependent, Ethylene-independent pathway. Plant Physiol 156 : 596–604. doi: 10.1104/pp.111.175372 21508182
26. Taylor-Teeples M, Lin L, de Lucas M, Turco G, Toal TW, et al. (2015) An Arabidopsis gene regulatory network for secondary cell wall synthesis. Nature 517 : 571–575. doi: 10.1038/nature14099 25533953
27. Ellis C, Karafyllidis I, Wasternack C, Turner JG (2002) The Arabidopsis mutant cev1 links cell wall signaling to jasmonate and ethylene responses. Plant Cell 14 : 1557–1566. 12119374
28. Manfield IW, Orfila C, Mccartney L, Harholt J, Bernal AJ, et al. (2004) Novel cell wall architecture of isoxaben-habituated Arabidopsis suspension-cultured cells: global transcript profiling and cellular analysis. Plant J 40 : 260–275. 15447652
29. Hamann T, Bennett M, Mansfield J, Somerville C (2009) Identification of cell-wall stress as a hexose-dependent and osmosensitive regulator of plant responses. Plant J 57 : 1015–1026. doi: 10.1111/j.1365-313X.2008.03744.x 19036034
30. Boorsma A, de Nobel H, ter Riet B, Bargmann B, Brul S, et al. (2004) Characterization of the transcriptional response to cell wall stress in Saccharomyces cerevisiae. Yeast 21 : 413–427. 15116342
31. Levin DE (2005) Cell wall integrity signaling in Saccharomyces cerevisiae. Microbiol. Mol Bio Rev 69 : 262.
32. Moura JC, Bonine CA, de Oliveira FVJ, Dornelas MC, Mazzafera P (2010) Abiotic and biotic stresses and changes in lignin content and composition in plants. J Integr Plant Biol 52 : 360–76. doi: 10.1111/j.1744-7909.2010.00892.x 20377698
33. Le MQ, Pagter M, Hincha DK (2015) Global changes in gene expression, assayed by microarray hybridization and quantitative RT-PCR, during acclimation of three Arabidopsis thaliana accessions to sub-zero temperatures after cold acclimation. Plant Mol Biol 87 : 1–15 doi: 10.1007/s11103-014-0256-z 25311197
34. Rajashekar C, Burke MJ (1996) Freezing characteristics of rigid plant tissues (development of cell tension during extracellular freezing). Plant Physiol 111 : 597–603. 12226313
35. Ball MC, Canny MJ, Huang CX, Heady RD (2004) Structural changes in acclimated and unacclimated leaves during freezing and thawing. Func Plant Biol 31 : 29–40.
36. Ball MC, Canny MJ, Huang CX, Egerton JJG, Wolfe J (2006) Freeze/thaw-induced embolism depends on nadir temperature: the heterogeneous hydration hypothesis. Plant Cell Environ 29 : 729–745. 17087458
37. Ferrer JL, Austin MB, Stewart C Jr, Noel JP (2008) Structure and function of enzymes involved in the biosynthesis of phenylpropanoids. Plant Physiol Biochem 46 : 356–370. doi: 10.1016/j.plaphy.2007.12.009 18272377
38. Shafi A, Dogra V, Gill T, Ahuja PS, Sreenivasulu Y (2014) Simultaneous over-expression of PaSOD and RaAPX in transgenic Arabidopsis thaliana confers cold stress tolerance through increase in vascular lignifications. PLoS One 9: e110302. doi: 10.1371/journal.pone.0110302 25330211
39. Raes J, Rohde A, Christensen JH, Van de Peer Y, Boerjan W (2003) Genome-wide characterization of the lignification toolbox in Arabidopsis. Plant Physiol 133 : 1051–1071. 14612585
40. Brown DM, Zeef LA, Ellis J, Goodacre R, Turner SR (2005) Identification of novel genes in Arabidopsis involved in secondary cell wall formation using expression profiling and reverse genetics. Plant Cell 17 : 2281–2295. 15980264
41. Cassan-Wang H, Goué N, Saidi MN, Legay S, Sivadon P, et al. (2013) Identification of novel transcription factors regulating secondary cell wall formation in Arabidopsis. Front Plant Sci 11 : 189.
42. Lu S, Li Q, Wei H, Chang MJ, Tunlaya-Anukit S, et al. (2013) Ptr-miR397a is a negative regulator of laccase genes affecting lignin content in Populus trichocarpa. Proc. Natl. Acad. Sci. U S A 110 : 10848–10853. doi: 10.1073/pnas.1308936110 23754401
43. Petrik DL, Karlen SD, Cass CL, Padmakshan D, Lu F, et al. (2014) p-Coumaroyl-CoA:monolignol transferase (PMT) acts specifically in the lignin biosynthetic pathway in Brachypodium distachyon. Plant J 77 : 713–726. doi: 10.1111/tpj.12420 24372757
44. Dixon RA, Paiva NL (1995) Stress-induced phenylpropanoid metabolism. Plant Cell 7 : 1085–1097. 12242399
45. Ross W, Ron S (1995) Lignin Biosynthesis. Plant Cell 7 : 1001–1013. 12242395
46. Kim DS, Hwang BK (2014) An important role of the pepper phenylalanine ammonia-lyase gene (PAL1) in salicylic acid-dependent signalling of the defence response to microbial pathogens. J Exp Bot 65 : 2295–2306. doi: 10.1093/jxb/eru109 24642849
47. Olsen KM, Lea US, Slimestad R, Verheul M, Lillo C (2008) Differential expression of four Arabidopsis PAL genes; PAL1 and PAL2 have functional specialization in abiotic environmental-triggered flavonoid synthesis. J Plant Physiol 165 : 1491–1499. doi: 10.1016/j.jplph.2007.11.005 18242769
48. Cass CL, Peraldi A, Dowd PF, Mottiar Y, Santoro N, et al. (2015) Effects of PHENYLALANINE AMMONIA LYASE (PAL) knockdown on cell wall composition, biomass digestibility, and biotic and abiotic stress responses in Brachypodium. J Exp Bot 66 : 4317–4335. doi: 10.1093/jxb/erv269 26093023
49. Ezaki B, Sasaki K, Matsumoto H, Nakashima S (2005) Functions of two genes in aluminium (Al) stress resistance: repression of oxidative damage by the AtBCB gene and promotion of efflux of Al ions by the NtGDI1gene. J Exp Bot 56 : 2661–2671. 16143720
50. Huang J, Gu M, Lai Z, Fan B, Shi K, et al. (2010) Functional analysis of the Arabidopsis PAL gene family in plant growth, development, and response to environmental stress. Plant Physiol 153 : 1526–1538. doi: 10.1104/pp.110.157370 20566705
51. Kilian J, Whitehead D, Horak J, Wanke D, Weinl S, et al. (2007) The AtGenExpress global stress expression data set: protocols, evaluation and model data analysis of UV-B light, drought and cold stress responses. Plant J 50 : 347–363. 17376166
52. Ohtsubo M, Okazaki H, Nishimoto T (1989) The RCC1 protein, a regulator for the onset of chromosome condensation locates in the nucleus and binds to DNA. J Cell Biol 109 : 1389–1397. 2677018
53. Renault L, Nassar N, Vetter I, Becker J, Klebe C, et al. (1998) The 1.7 A crystal structure of the regulator of chromosome condensation (RCC1) reveals a seven-bladed propeller. Nature 392 : 97–101. 9510255
54. Bischoff FR, Ponstingl H (1991) Catalysis of guanine nucleotide exchange on Ran by the mitotic regulator RCC1. Nature 354 : 80–82. 1944575
55. Nemergut ME, Lindsay ME, Brownawell AM, Macara IG (2002) Ran-binding protein 3 links Crm1 to the Ran guanine nucleotide exchange factor. J Biol Chem 277 : 17385–17388. 11932251
56. Rohde A, Morreel K, Ralph J, Goeminne G, Hostyn V, et al. (2004) Molecular phenotyping of the pal1 and pal2 mutants of Arabidopsis thaliana reveals far-reaching consequences on phenylpropanoid, amino acid, and carbohydrate metabolism. Plant Cell 16 : 2749–2771. 15377757
57. Li HY, Wirtz D, Zheng Y (2003) A mechanism of coupling RCC1 mobility to RanGTP production on the chromatin in vivo. J Cell Biol 160 : 635–644. 12604592
58. Cloix C, Jenkins GI (2008) Interaction of the Arabidopsis UV-B-specific signaling component UVR8 with chromatin. Mol Plant 1 : 118–128. doi: 10.1093/mp/ssm012 20031919
59. Van Gysel A, Van Montagu M, Inzé D (1993) A negatively light-regulated gene from Arabidopsis thaliana encodes a protein showing high similarity to blue copper-binding proteins. Gene 136 : 79–85. 8294044
60. Borner GH, Lilley KS, Stevens TJ, Dupree P (2003) Identification of glycosylphosphatidylinositol-anchored proteins in Arabidopsis. A proteomic and genomic analysis. Plant Physiol 132 : 568–77. 12805588
61. Mishina TE, Zeier J (2007) Bacterial non-host resistance: interactions of Arabidopsis with non-adapted Pseudomonas syringae strains. Physiol Plant 131 : 448–461. doi: 10.1111/j.1399-3054.2007.00977.x 18251883
62. Huang X, Yang P, Ouyang X, Chen L, Deng XW (2014) Photoactivated UVR8-COP1 module determines photomorphogenic UV-B signaling output in Arabidopsis. PLoS Genet 10: e1004218. doi: 10.1371/journal.pgen.1004218 24651064
63. Levitt J (1980) Chilling, freezing, and high temperature stress. Responses of plants to environmental stresses; London: Academic Press I: 497.
64. Sakai A, Larcher W (1987) Ecological Studies. Frost survival of plants. Responses and adaptation to freezing stress; Berlin: Springer-Verlag 62 : 321.
65. Alberdi M, Corcuera LJ (1991) Cold acclimation in plants. Phytochemistry 30 : 3177–3184.
66. Domon JM, Baldwin L, Acket S, Caudeville E, Arnoult S, et al. (2013) Cell wall compositional modifications of Miscanthus ecotypes in response to cold acclimation. Phytochemistry 85 : 51–61. doi: 10.1016/j.phytochem.2012.09.001 23079767
67. Zuther E, Schulz E, Childs LH, Hincha DK. (2012) Clinal variation in the non-acclimated and cold-acclimated freezing tolerance of Arabidopsis thaliana accessions. Plant Cell Environ 35 : 1860–1878. doi: 10.1111/j.1365-3040.2012.02522.x 22512351
68. Ji H, Wang S, Li K, Szakonyi D, Koncz C, et al. (2014) PRL1 modulates root stem cell niche activity and meristem size through WOX5 and PLTs in Arabidopsis. Plant J 81 : 399–412. doi: 10.1111/tpj.12733 25438658
69. Gendrel AV, Lippman Z, Yordan C, Colot V, Martienssen RA (2002) Dependence of heterochromatic histone H3 methylation patterns on the Arabidopsis gene DDM1. Science 297 : 1871–1873. 12077425
70. Tokunaga N, Sakakibara N, Umezawa T, Ito Y, Fukuda H, et al. (2005). Involvement of extracellular dilignols in lignification during tracheary element differentiation of isolated Zinnia mesophyll cells. Plant Cell Physiol 46 : 224–232. 15659440
71. Fukuda H, Komamine A (1982) Lignin synthesis and its related enzymes as markers of tracheary-element differentiation in single cells isolated from the mesophyll of Zinnia elegans. Planta 155 : 423–430 doi: 10.1007/BF00394471 24271974
Štítky
Genetika Reprodukční medicína
Článek The Chromatin Protein DUET/MMD1 Controls Expression of the Meiotic Gene during Male Meiosis inČlánek Tissue-Specific Gain of RTK Signalling Uncovers Selective Cell Vulnerability during Embryogenesis
Článek vyšel v časopisePLOS Genetics
Nejčtenější tento týden
2015 Číslo 9
-
Všechny články tohoto čísla
- Retraction: RNAi-Dependent and Independent Control of LINE1 Accumulation and Mobility in Mouse Embryonic Stem Cells
- Signaling from Within: Endocytic Trafficking of the Robo Receptor Is Required for Midline Axon Repulsion
- A Splice Region Variant in Lowers Non-high Density Lipoprotein Cholesterol and Protects against Coronary Artery Disease
- The Chromatin Protein DUET/MMD1 Controls Expression of the Meiotic Gene during Male Meiosis in
- A NIMA-Related Kinase Suppresses the Flagellar Instability Associated with the Loss of Multiple Axonemal Structures
- Slit-Dependent Endocytic Trafficking of the Robo Receptor Is Required for Son of Sevenless Recruitment and Midline Axon Repulsion
- Expression of Concern: Protein Under-Wrapping Causes Dosage Sensitivity and Decreases Gene Duplicability
- Mutagenesis by AID: Being in the Right Place at the Right Time
- Identification of as a Genetic Modifier That Regulates the Global Orientation of Mammalian Hair Follicles
- Bridges Meristem and Organ Primordia Boundaries through , , and during Flower Development in
- Evaluating the Performance of Fine-Mapping Strategies at Common Variant GWAS Loci
- KLK5 Inactivation Reverses Cutaneous Hallmarks of Netherton Syndrome
- Differential Expression of Ecdysone Receptor Leads to Variation in Phenotypic Plasticity across Serial Homologs
- Receptor Polymorphism and Genomic Structure Interact to Shape Bitter Taste Perception
- Cognitive Function Related to the Gene Acquired from an LTR Retrotransposon in Eutherians
- Critical Function of γH2A in S-Phase
- Arabidopsis AtPLC2 Is a Primary Phosphoinositide-Specific Phospholipase C in Phosphoinositide Metabolism and the Endoplasmic Reticulum Stress Response
- XBP1-Independent UPR Pathways Suppress C/EBP-β Mediated Chondrocyte Differentiation in ER-Stress Related Skeletal Disease
- Integration of Genome-Wide SNP Data and Gene-Expression Profiles Reveals Six Novel Loci and Regulatory Mechanisms for Amino Acids and Acylcarnitines in Whole Blood
- A Genome-Wide Association Study of a Biomarker of Nicotine Metabolism
- Cell Cycle Regulates Nuclear Stability of AID and Determines the Cellular Response to AID
- A Genome-Wide Association Analysis Reveals Epistatic Cancellation of Additive Genetic Variance for Root Length in
- Tissue-Specific Gain of RTK Signalling Uncovers Selective Cell Vulnerability during Embryogenesis
- RAB-10-Dependent Membrane Transport Is Required for Dendrite Arborization
- Basolateral Endocytic Recycling Requires RAB-10 and AMPH-1 Mediated Recruitment of RAB-5 GAP TBC-2 to Endosomes
- Dynamic Contacts of U2, RES, Cwc25, Prp8 and Prp45 Proteins with the Pre-mRNA Branch-Site and 3' Splice Site during Catalytic Activation and Step 1 Catalysis in Yeast Spliceosomes
- ARID1A Is Essential for Endometrial Function during Early Pregnancy
- Predicting Carriers of Ongoing Selective Sweeps without Knowledge of the Favored Allele
- An Interaction between RRP6 and SU(VAR)3-9 Targets RRP6 to Heterochromatin and Contributes to Heterochromatin Maintenance in
- Photoreceptor Specificity in the Light-Induced and COP1-Mediated Rapid Degradation of the Repressor of Photomorphogenesis SPA2 in Arabidopsis
- Autophosphorylation of the Bacterial Tyrosine-Kinase CpsD Connects Capsule Synthesis with the Cell Cycle in
- Multimer Formation Explains Allelic Suppression of PRDM9 Recombination Hotspots
- Rescheduling Behavioral Subunits of a Fixed Action Pattern by Genetic Manipulation of Peptidergic Signaling
- A Gene Regulatory Program for Meiotic Prophase in the Fetal Ovary
- Cell-Autonomous Gβ Signaling Defines Neuron-Specific Steady State Serotonin Synthesis in
- Discovering Genetic Interactions in Large-Scale Association Studies by Stage-wise Likelihood Ratio Tests
- The RCC1 Family Protein TCF1 Regulates Freezing Tolerance and Cold Acclimation through Modulating Lignin Biosynthesis
- The AMPK, Snf1, Negatively Regulates the Hog1 MAPK Pathway in ER Stress Response
- The Parkinson’s Disease-Associated Protein Kinase LRRK2 Modulates Notch Signaling through the Endosomal Pathway
- Multicopy Single-Stranded DNA Directs Intestinal Colonization of Enteric Pathogens
- Recurrent Domestication by Lepidoptera of Genes from Their Parasites Mediated by Bracoviruses
- Three Different Pathways Prevent Chromosome Segregation in the Presence of DNA Damage or Replication Stress in Budding Yeast
- Identification of Four Mouse Diabetes Candidate Genes Altering β-Cell Proliferation
- The Intolerance of Regulatory Sequence to Genetic Variation Predicts Gene Dosage Sensitivity
- Synergistic and Dose-Controlled Regulation of Cellulase Gene Expression in
- Genome Sequence and Transcriptome Analyses of : Metabolic Tools for Enhanced Algal Fitness in the Prominent Order Prymnesiales (Haptophyceae)
- Ty3 Retrotransposon Hijacks Mating Yeast RNA Processing Bodies to Infect New Genomes
- FUS Interacts with HSP60 to Promote Mitochondrial Damage
- Point Mutations in Centromeric Histone Induce Post-zygotic Incompatibility and Uniparental Inheritance
- Genome-Wide Association Study with Targeted and Non-targeted NMR Metabolomics Identifies 15 Novel Loci of Urinary Human Metabolic Individuality
- Outer Hair Cell Lateral Wall Structure Constrains the Mobility of Plasma Membrane Proteins
- A Large-Scale Functional Analysis of Putative Target Genes of Mating-Type Loci Provides Insight into the Regulation of Sexual Development of the Cereal Pathogen
- A Genetic Selection for Mutants Reveals an Interaction between DNA Polymerase IV and the Replicative Polymerase That Is Required for Translesion Synthesis
- PLOS Genetics
- Archiv čísel
- Aktuální číslo
- Informace o časopisu
Nejčtenější v tomto čísle- Arabidopsis AtPLC2 Is a Primary Phosphoinositide-Specific Phospholipase C in Phosphoinositide Metabolism and the Endoplasmic Reticulum Stress Response
- Bridges Meristem and Organ Primordia Boundaries through , , and during Flower Development in
- KLK5 Inactivation Reverses Cutaneous Hallmarks of Netherton Syndrome
- XBP1-Independent UPR Pathways Suppress C/EBP-β Mediated Chondrocyte Differentiation in ER-Stress Related Skeletal Disease
Kurzy
Zvyšte si kvalifikaci online z pohodlí domova
Současné možnosti léčby obezity
nový kurzAutoři: MUDr. Martin Hrubý
Všechny kurzyPřihlášení#ADS_BOTTOM_SCRIPTS#Zapomenuté hesloZadejte e-mailovou adresu, se kterou jste vytvářel(a) účet, budou Vám na ni zaslány informace k nastavení nového hesla.
- Vzdělávání



