-
Články
Top novinky
Reklama- Vzdělávání
- Časopisy
Top články
Nové číslo
- Témata
Top novinky
Reklama- Videa
- Podcasty
Nové podcasty
Reklama- Kariéra
Doporučené pozice
Reklama- Praxe
Top novinky
ReklamaGenetic Interaction Maps in Reveal Functional Crosstalk among Cell Envelope Biogenesis Pathways
As the interface between a microbe and its environment, the bacterial cell envelope has broad biological and clinical significance. While numerous biosynthesis genes and pathways have been identified and studied in isolation, how these intersect functionally to ensure envelope integrity during adaptive responses to environmental challenge remains unclear. To this end, we performed high-density synthetic genetic screens to generate quantitative functional association maps encompassing virtually the entire cell envelope biosynthetic machinery of Escherichia coli under both auxotrophic (rich medium) and prototrophic (minimal medium) culture conditions. The differential patterns of genetic interactions detected among >235,000 digenic mutant combinations tested reveal unexpected condition-specific functional crosstalk and genetic backup mechanisms that ensure stress-resistant envelope assembly and maintenance. These networks also provide insights into the global systems connectivity and dynamic functional reorganization of a universal bacterial structure that is both broadly conserved among eubacteria (including pathogens) and an important target.
Published in the journal: . PLoS Genet 7(11): e32767. doi:10.1371/journal.pgen.1002377
Category: Research Article
doi: https://doi.org/10.1371/journal.pgen.1002377Summary
As the interface between a microbe and its environment, the bacterial cell envelope has broad biological and clinical significance. While numerous biosynthesis genes and pathways have been identified and studied in isolation, how these intersect functionally to ensure envelope integrity during adaptive responses to environmental challenge remains unclear. To this end, we performed high-density synthetic genetic screens to generate quantitative functional association maps encompassing virtually the entire cell envelope biosynthetic machinery of Escherichia coli under both auxotrophic (rich medium) and prototrophic (minimal medium) culture conditions. The differential patterns of genetic interactions detected among >235,000 digenic mutant combinations tested reveal unexpected condition-specific functional crosstalk and genetic backup mechanisms that ensure stress-resistant envelope assembly and maintenance. These networks also provide insights into the global systems connectivity and dynamic functional reorganization of a universal bacterial structure that is both broadly conserved among eubacteria (including pathogens) and an important target.
Introduction
The bacterial cell envelope serves as a resilient macrostructure and permeability barrier that protects microbes from osmotic stress, xenobiotics and environmental insults while supporting cell morphology and transport of essential nutrients and waste. For Gram-negative species like Escherichia coli, the cell envelope consists sequentially of a phospholipidic inner membrane (IM), a peptidoglycan (PG) cell wall embedded in aqueous periplasm, and an outer membrane (OM) composed of phospholipids and lipopolysaccharide (LPS). Unique sets of proteins determine the functional identity of each of these compartments under physiological demand [1], [2]. These include diverse outer membrane proteins (OMP) such as β-barrel porins that mediate cell adhesion and passage of small molecules, inner membrane proteins (IMP) involved in active transport and adaptation to changing growth conditions, and soluble periplasmic enzymes and membrane-tethered lipoproteins that process metabolic precursors required for envelope assembly. Proper expression, transport and activity of these envelope-associated proteins is critical to bacterial cell viability, morphology and stress-resistance [3].
The cell envelope also plays a crucial role in bacterial pathogenesis [3]. One-quarter of prescription antimicrobials are currently directed against proteins involved in envelope biogenesis [4], including classical antibiotics such as the β-lactams (e.g. penicillin) and modern ‘last resort’ glycopeptide drugs like vancomycin. Yet despite the emergence of widespread clinical resistance, few new therapeutics targeting the bacterial cell envelope have been developed over the past two decades due in part to the extraordinary functional robustness of this structure [5]. The utility of combination therapies targeting redundant pathways as an alternate clinical strategy [2] underscores the need to identify functional dependencies among essential envelope bioprocesses that contribute to envelope formation, stability and drug tolerance.
Cell envelope biogenesis has been studied extensively in E. coli [3], including investigations of individual genes and pathways involved in the biogenesis of fatty acids [6], cell wall [7], LPS [8] and OMP [9]. Yet important gaps still exist in the understanding of the global mechanisms that ensure faithful envelope assembly and integrity under different growth conditions or in response to antibiotic challenges [3]. New components of envelope assembly pathways continue to be discovered [10], [11], [12], [13], yet nearly one-third of the membrane proteins of E. coli are currently functionally unannotated likely in part due to incomplete or biased historical experimental analyses [1], [2], [14]. Moreover, most (>90%) of the annotated biosynthetic genes of E. coli are dispensable for viability under standard laboratory culture conditions [15]. This redundancy, which presumably reflects in part a robust modular systems level organization [16], has hindered both functional annotation [2] and antibiotic development using traditional single gene/target-centric approaches [17].
Unbiased genetic screens can reveal unexpected functional dependencies between genetic loci (i.e. epistasis, wherein the phenotypic effects of mutation of one gene are modified by one or more other genes). For example, aggravating (negative) genetic interactions, manifesting as synthetic sickness or lethality, often result from loss-of-function mutations in pairs of genes in parallel or compensatory pathways that impinge on a common essential process [18]. Conversely, alleviating (positive) interactions can occur between genes in the same pathway if the loss of one gene alone inactivates the pathway such that loss of a second gene confers no additional defect [18]. Large-scale genetic screens in yeast have outlined the tightly integrated functional organization of essential biological systems [19], [20], and global network rewiring in response to environmental stress causing DNA damage [21]. No analogous systematic surveys have yet been reported for bacteria, and only ad hoc genetic studies on a few select components of the E. coli cell envelope have been reported to date [22]. As a result, the degree of functional redundancy, connectivity and modularity among the biosynthetic pathways supporting envelope assembly and maintenance remains unclear. Such knowledge is paramount for targeting envelope systems resistant to existing antibiotics.
To this end, we applied our high-throughput synthetic genetic array (eSGA) screening technology [23] in a comprehensive manner to identify and quantify epistatic relationships between all known and predicted components of the cell envelope biosynthetic machinery of E. coli during growth in both auxotrophic and prototrophic culture conditions. Unbiased scoring and filtering of the resulting genetic data revealed condition-specific genetic interaction networks required for the proper formation and integrity of the OM, cell wall, and LPS, and functional dependencies mediating membrane protein secretion and cell division, which were verified independently. These functional association maps provide a unique perspective into the global functional architecture and dynamic rewiring of widely-conserved envelope bioprocesses critical to bacterial morphology, fitness and environmental adaptation. All of the data are publicly-accessible as a community resource via a dedicated web portal.
Results
High-throughput genetic screens of cell envelope bioprocesses
Using a quantitative screening format originally developed to investigate pathway crosstalk in yeast [20], we performed 821 high-density eSGA screens to examine the fitness of all possible digenic mutant combinations of hypomorphic alleles (i.e. partial loss of gene function) of 128 essential biosynthetic genes and single-gene deletions of 683 non-essential protein-coding envelope genes and 10 small non-coding regulatory RNAs (sRNA) linked to post-transcriptional regulation of cell surface protein expression (Figure 1A, see Protocol S1). Target inclusion was based on an exhaustive survey of the literature and databases of envelope-related biosynthetic pathways and gene annotations [1] (Table S1).
Fig. 1. Cell envelope targets and schematic of cell envelope eSGA strategy. 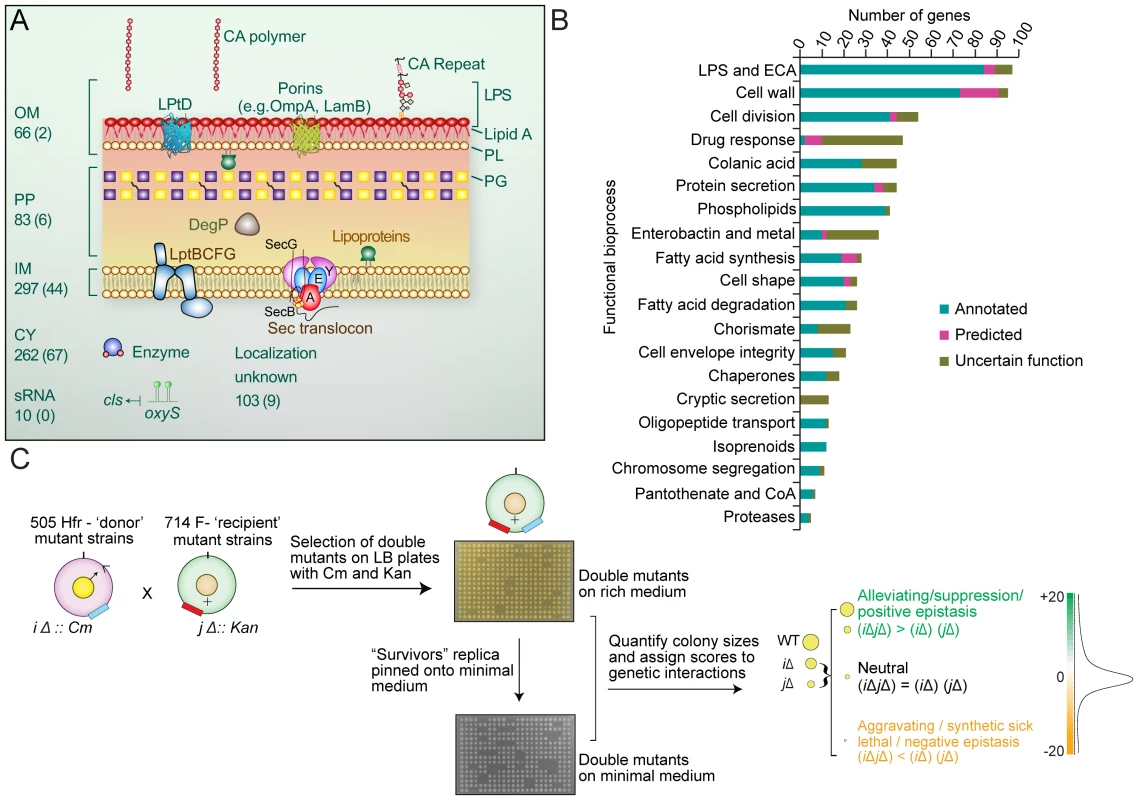
(A) Schematic summary of the eSGA gene targets associated with the E. coli outer membrane (OM), inner membrane (IM), periplasm (PP), cytoplasm (CY), regulatory RNA (sRNA), or of uncertain localization. Gene numbers are indicated, with essential genes shown in brackets. (B) Functional distribution of annotated (Group 1), uncertain function (Group 2) and predicted (Group 3) envelope target genes sorted into 20 broadly representative bioprocesses. (C) Schematic summarizing the experimental construction and computational analysis of two E. coli cell-envelope genetic interaction maps. After conjugation and genetic exchange, high density arrays of double-mutants were first selected on rich medium containing Cm and Kan. After outgrowth for 24 hours, surviving colonies were then replica pinned onto minimal M9 medium, to identify condition-dependent genetic interactions. Both sets of plates were digitally imaged and colony sizes quantified to determine gene pairs showing aggravating (synthetic lethal and synthetic sick) and alleviating (buffering) interactions. Targets came from 20 representative bioprocesses (Figure 1B), including 363 integral membrane proteins (66 OMPs, 297 IMPs), and consisted of three basic types: Group 1 (Annotated), comprising 357 core biosynthetic genes (‘building block’ enzymes) with experimental evidence supporting direct involvement in a specific step of an annotated envelope biogenesis pathway or bioprocess; Group 2 (Uncertain Function), encompassing 286 genes, where existing experimental evidence indicates a role in envelope biogenesis but without certainty as to a particular pathway or bioprocess; Group 3 (Predicted), comprising 178 unannotated genes predicted to participate in envelope biogenesis based on protein-protein interactions, genomic context inferences [14], or tentative EcoCyc, Gene Ontology (GO) or GenProtEC assignments. Despite significant differences in bacterial morphology and envelope composition (e.g. Gram-positives versus Gram-negatives), many of these genes are broadly evolutionary conserved consistent with fundamental roles in cell envelope biology beyond E. coli (Figure S1 and Table S2).
Following the conjugation of single mutant strains and genetic transfer (Figure 1C), double mutants were first plated as replicate arrays onto solid agar containing rich (Luria Broth) medium. After outgrowth for 24 hrs at 32°C, viable stationary phase colonies were then replica pinned onto minimal (M9) medium to identify additional genetic interactions under limiting nitrogen and carbon levels (Figure 1C). Both sets of plates were digitally imaged, and colony sizes measured and normalized to account for experimental variation (see Protocol S2 and S3). Screen reproducibility was uniformly high (r typically >0.8; Figure S2A) and comparable to high-quality quantitative genetic interaction screens reported previously for yeast [19], [20].
Although the Hfr donor strain is not isogenic with the BW25113 KEIO deletion mutant strain background, a pilot test set of 30×30 conjugations showed that the growth rates of double mutants produced by crossing 30 diverse F - ‘recipient’ strains from the Keio collection [15] with the corresponding set of 30 ‘donor’ mutants in either an Hfr C or an isogenic strain background were comparable (r = 0.7; Figure S2B).
Digenic mutant fitness was estimated using an established multiplicative model [19] which reports both the strength and confidence of genetic interactions between any two genes (see Protocol S4). Briefly, if two genes are functionally unrelated, the growth rates of the respective single mutations are predicted to combine in a simple multiplicative manner in the double mutant; significant deviations from this expected fitness imply a functional association [18].
The resulting E-scores of the double mutants on both rich medium and minimal medium (Table S3) showed a bimodal distribution (Figure S2C), with the major peak approximating a normal distribution centered on neutrality, confirming the expectation that genetic interactions are relatively uncommon (i.e. limited to only certain gene pairs). Positive E-scores suggestive of alleviating interactions (double mutants grew more rapidly than expected) were found in the right tail of the distributions, reflecting factors operating in the same pathway [24], while aggravating interactions (double mutants grew more slowly than expected) occurred in the heavy left tail, with a prominent peak of highly negative E-scores representing gene pairs exhibiting synthetic lethality [19].
Deriving high-confidence genetic interaction maps
Genetic interactions by a given biogenesis gene reflect its functional associations with other envelope components and thus serve as a high-resolution phenotype. We applied a statistical framework to define suitable E-score cutoffs to derive biologically-relevant interactions by minimizing the false discovery rate. Specifically, we evaluated the extent to which pairs of envelope genes involved in different biogenesis bioprocesses showed significant (p-value≤0.05) enrichment at various E-score values.
Enrichment increased progressively as the E-score threshold was raised (Figure S2D), reaching a maximum at −2 (aggravating interactions) and +2 (alleviating interactions). We used these apex values (E≤−2, E≥2) as cutoffs, and assigned p-values using a null distribution background (see Methods).
Strikingly, the patterns of genetic interactions found in rich and minimal (Table S3) medium were markedly different, reflecting a profound reorganization in the envelope biogenesis machinery. For example, although more highly connected on average than non-essential genes (Figure S2E and S2F), essential genes exhibited a higher ratio of alleviating interactions (p-value = 1.22×10−50) in rich medium (Figure 2A, Figure S3A, Protocol S5). Conversely, aggravating interactions were far more common (p-value = 3.70×10−57) for essential genes in minimal medium (Figure 2A), suggesting compensatory relationships emerge under environmental constraint (i.e. nutrient limitation). The likelihood of a genetic interaction by any one particular envelope biogenesis gene in either condition was correlated both with gene essentiality and mRNA expression levels in culture (Figure S3B). As with yeast [19], [25], essential genes producing abundant transcripts had significantly more aggravating interactions, while envelope factors with multiple annotated functions suggestive of pleiotropy were more likely to display synthetic lethality (Table S4).
Fig. 2. Analysis on the genetic interaction networks derived from two growth conditions. 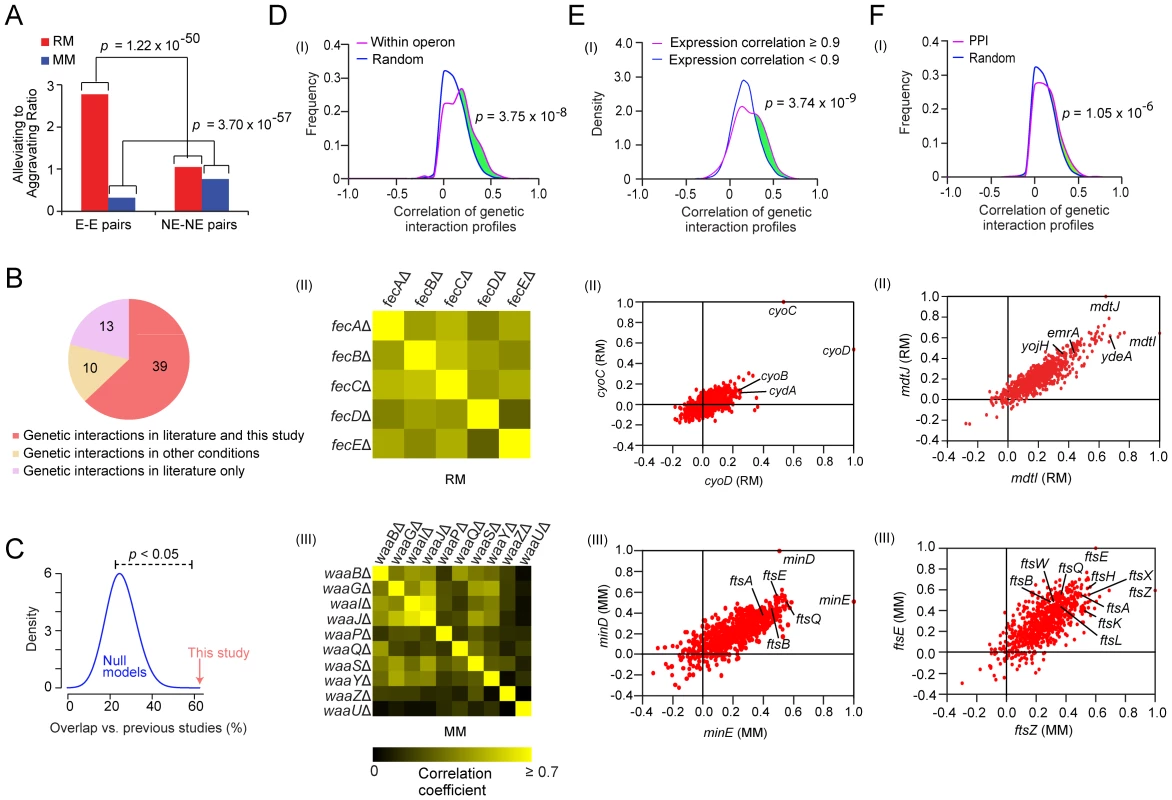
(A) Comparison of ratios of alleviating (E-score ≥2) to aggravating (E- score ≤−2) genetic interaction involving essential (E) and non-essential (NE) gene pairs on both growth conditions, p-values were computed using Fisher's exact test. (B) Venn diagram showing the overlap of the genetic interaction pairs derived from this study and the literature. (C) Significance testing of the agreement between the literature and this study (red arrow) compared against expected gene pair frequencies by randomly sampling (blue distribution represents 10,000 null models). (D) Distribution of correlations coefficients between the genetic interaction profiles for gene pairs within the same operon versus randomly drawn gene pairs (panel I). Pair-wise profile correlation coefficient of genes in the Fe-enterobactin “fec” operon in rich medium (panel II) and for LPS 1, 2-glucosyltransferase components of waa operon in minimal medium (panel III). (E) Distribution of genetic interaction profile correlation for gene pairs with correlated co-expression (panel I). Scatter-plot of correlated genetic profiles is shown for cyoD (x-axis) and cyoC (y-axis) in panel II, and for minE (x-axis) and minD (y-axis) in panel III. (F) Distribution of correlation coefficients between the genetic interaction profiles of gene pairs encoding interacting proteins versus randomly drawn gene pairs (panel I), where the p-value was computed using the two-sample Kolmogorov-Smirnov (KS) test. Scatter plot of correlated genetic profiles for mdtI (x-axis) and mdtJ (y-axis) (panel II). Scatter plot of correlated genetic profiles for ftsZ (x-axis) and ftsE (y-axis) (panel III). To independently gauge the accuracy of these networks, we examined the screen results obtained for 62 gene pairs reported previously to exhibit aggravating (53 pairs) or alleviating (9 pairs) phenotypes. In total, we correctly captured two-thirds (63%) of the published GI (Figure 2B and Table S5), which is statistically significant (p-value<0.05 by random sampling; Figure 2C). For example, the rich medium network recapitulated aggravating interactions between the periplasmic chaperone surA and the β-barrel protein assembly machinery (BAM) [26] and a functionally redundant chaperone skp [27]. Conversely, the minimal medium network captured the conditional synthetic lethality reported between cell division proteins zapB and ftsZ [28]. Half (10 of 23) of the discordant gene pairs were found using different growth conditions in the literature (Table S5), reinforcing the notion of conditional-dependency.
Many components of the cell envelope biosynthetic machinery are predicted to be functionally-associated based on physical association (Figure S4A), transcriptional co-expression (Figure S4B), and/or genomic context (Figure S4C). Consistent with this, cell envelope genes with positively correlated genetic profiles were more likely to be present within the same operon in E. coli compared to random pairs of genes (Figure 2D), consistent with the natural chromosomal clustering of functionally-related genes in bacteria [29]. For example, components of the fec Fe-enterobactin uptake system were correlated in rich medium (Figure 2D), consistent with joint participation in the import of cellular iron [30]. Similarly, the genetic interaction patterns of the LPS 1, 2-glucosyltransferase components of waa operon (formerly rfa) were correlated in minimal medium (Figure 2D), consistent with the coordinated role in LPS core biosynthesis [31]. Likewise, pairs of co-expressed envelope genes also tended to show more positively correlated genetic interaction profiles (Figure 2E). For example, the genetic interaction profiles of the co-expressed cytochrome bo terminal oxidase subunits (cyoC, cyoD) were correlated in rich medium (r = 0.53; Figure 2E), reflecting elevated aerobic respiration. In contrast, the genetic interaction patterns of the co-expressed cell division components (minD, minE) were closely correlated in minimal medium (r = 0.51; Figure 2E), consistent with the cooperative role in modulating the division potential of cellular sites located at mid cell and at the cell poles [32].
As in yeast [19], [20], envelope biogenesis factors with highly correlated genetic profiles were also significantly more likely to be connected by protein-protein interaction (PPI) (Figure 2F). For example, the genetic profiles of two transporters (mdtI, mdtJ) that form a heterodimeric complex required for spermidine excretion [33] were highly correlated in rich medium (r = 0.65; Figure 2F). The profiles of mdtJ/mdtI were also similar to other translocases linked to multidrug resistance such as resistance-nodulation-cell division, major facilitator, and ATP-binding cassette proteins (Figure S5). Conversely, the genetic profiles of ftsE and its interacting partner ftsZ, which are required for assembly of the cytokinetic Z-ring [34], were closely correlated in minimal medium (r = 0.60; Figure 2F).
We concluded that the filtered networks reliably captured functional dependencies, and hence could be used to infer biologically-relevant relationships. For example, the genetic profile of the recently characterized OM lipoprotein ycfM/lpoB was similar to that of the penicillin binding protein mrcA under both rich and minimal conditions (Table S6), consistent with a recently proposed role as joint regulators of PG synthesis [12].
Bioprocess coupling
Having established that the genetic maps were informative about biological relationships at the level of individual envelope components, multiprotein complexes and biosynthetic pathways, we explored the global functional connections linking envelope bioprocesses in the two networks (Table S7 and S8). Using functional enrichment analysis (see Materials and Methods), we identified significant (p-value≤0.05) crosstalk (elevated patterns of aggravating or alleviating interactions) between bioprocesses (Figure 3A).
Fig. 3. Bioprocesses crosstalk critical for envelope integrity. 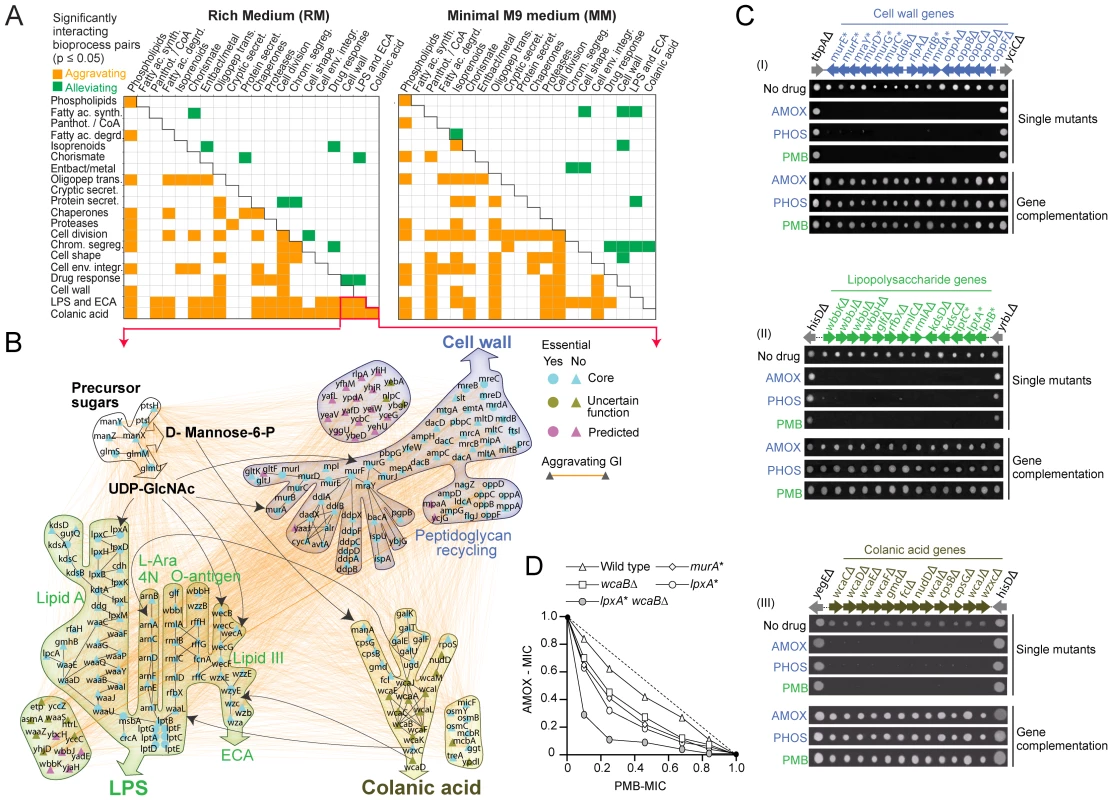
(A) Distinct patterns of enrichment in aggravating and alleviating genetic interactions between select envelope bioprocesses in rich and minimal medium. (B) Aggravating genetic interactions (orange edges) and metabolic links (black arrows) between the CA, LPS/ECA and cell wall biosynthetic pathways. (C) Antibiotic sensitivity of select E. coli mutants; bioprocess and gene identity indicated. Phenotypic complementation based on plasmid rescue. (D) Isobologram portraying the enhanced combination potency of amoxicillin (AMOX) and phosphomycin (PMB) against E. coli mutants deficient in cell wall (murA), LPS (lpxA) or CA (wcaB) production. Dotted line indicates additivity; MIC, minimal inhibitory concentration normalized relative to no drug control. We found that certain patterns were prevalent in both culture conditions (Figure 3A). For example, aggravating interactions were prominent (p-value≤0.05) between the oligopeptide transport, cell wall, cell shape and cell division machineries, consistent with the tight integration of these bioprocesses. As bacteria proliferate, the murein sacculus elongates in such a way that the distinctive rod shape of E. coli is maintained [7]. Murein peptides liberated by dynamic degradative recycling of the cell wall are actively imported for reuse by multiple reuptake systems [7]. Defective oligopeptide transport exacerbates slow growth and morphological deficiencies [7], which is reflected in our genetic networks.
In some cases, this crosstalk was unexpected. For example, Figure 3B shows a sub-network of aggravating interactions between the pathways generating colanic acid (CA), an exopolysaccharide (also known as M-antigen) produced by E. coli K-12 in response to hypotonic conditions or membrane perturbations [35], and the pathways responsible for producing the cell wall, LPS and enterobacterial common antigen (ECA), another surface glycophospholipid [36]. This crosstalk presumably reflects use of shared metabolic intermediates. For example, UDP-N acetylglucosamine is required for biosynthesis of ECA, LPS (lipid A) and PG in E. coli [36]. While disturbances in envelope integrity can lead to increased CA synthesis via activation of the Rcs regulon [37], defects in LPS formation do not affect CA expression under standard laboratory growth conditions [38], and dependencies between these systems have not been reported before [37].
To verify our results, we challenged CA mutants with amoxicillin and phosphomycin, antibiotics, which block cell wall/PG biogenesis, or with polymyxin B, which targets the lipid A component of LPS [2]. Consistent with our genetic data, CA strains were nearly as hypersensitive to the compounds as mutants deficient in cell wall and LPS formation (Figure 3C). A synergistic inhibitory effect was also observed when CA/LPS double (wcaB lpxA) mutants were challenged with amoxicillin and polymyxin B simultaneously (Figure 3D). Collectively, these data indicate that an underlying functional coordination (direct or indirect) of envelope glycoconjugate production is essential to sustain envelope integrity.
Conditional rewiring of cell envelope bioprocesses
Comparison of the two networks revealed differential physiological demands placed by the two culture conditions. For instance, alleviating interactions were preferentially detected in rich medium (p-value = 1.42×10−3; Figure 3A) between the pathways producing fatty acids and chorismate, the last branch point of the shikimate pathway that produces aromatic amino acid precursors. When amino acid pools exceed the requirements for protein synthesis, surplus aromatic precursors are directed towards fatty acid biosynthesis [39]. Hence, the lipid composition of the bacterial cell envelope reflects the combined output of both the shikimate and fatty acid pathways, which is mirrored as an alleviating phenotype during double mutant growth in abundant nutrient conditions.
Conversely, alternate selective pressures were placed on envelope biosynthetic pathways in minimal medium, where aggravating interactions became more pronounced (p-value = 9.4×10−4) between the cell division (Fts) and protein secretion (Sec) systems (Figure 4A). For example, SecY and Ftsk mutants showed alleviating interactions on rich medium, but synthetic lethality on minimal medium. This differential dependency was confirmed by liquid culture growth curve assays (Figure 4B). The genetic interaction profiles of cell division and secretion mutants were also more highly correlated on minimal medium than rich medium (Figure 4C). Genes exhibiting alleviating interactions are more likely to encode proteins that are physically associated [40]. Consistent with this, we found that affinity purified endogenous Fts interacts physically with the Sec export apparatus (Figure 4D, see Methods), implicating the Sec translocon directly in targeting of critical cell division determinants.
Fig. 4. Genetic dependencies reflect functional relationships. 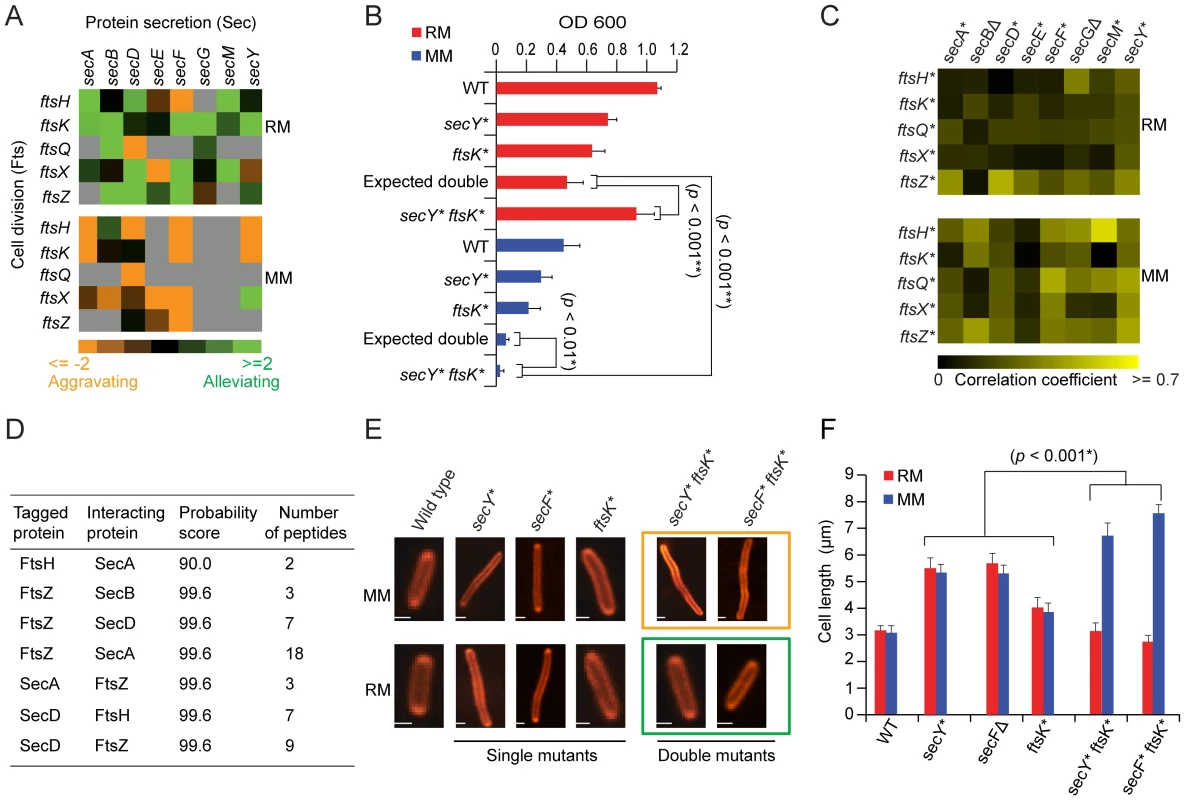
(A) Genetic interaction patterns between secretory and cell division factors. (B) Growth rates (fitness) of secY and ftsK single and double mutants in liquid rich (RM) or minimal medium (MM) at 32°C over 24 h; hypomorphic alleles indicated by asterisks. (C) Pair-wise correlation coefficient profiles of secretory and cell division mutants grown on rich medium or minimal medium. (D) Identification of Sec proteins co-purified from the endogenous affinity tagged Fts protein by tandem mass spectrometry. (E) Fluorescent microscopy of E. coli stains (stained with FM4-64) after growth in minimal or rich medium; scale-bars 2 µm. (F) Quantitative measurements of changes in mutant cell length after growth in rich and minimal medium. Error bars show measurement of standard deviation from three independent experiments. Protein secretion is an important determinant of nutrient import, and is known to be slowed along with cell division when bacteria are grown with limiting nutrients, favoring cell elongation [41]. Moreover, the cell surface localization of several Fts septation proteins including the essential ftsI protein (penicillin-binding protein 3) with a membrane spanning segment is one such process that requires sec translocon gene functions for correct membrane integration [41], [42]. Consistent with this, cell division/secretion double mutants formed exaggerated filamentous cell aggregates in minimal medium, whereas this phenotype was rescued by growth in rich medium (Figure 4E and 4F), suggesting transport of essential cleavage factors is limiting in nutrient-poor conditions.
Using a variant of differential interaction network strategy [21], slightly over 1, 400 gene pairs showed contrasting genetic interactions in rich versus minimal medium (Table S9, Protocol S6). These differential interactions were enriched significantly (p-value≤0.05) for genes functioning in cell division, chromosome segregation and protein secretion pathways (Figure S6A). For example, annotated murein amidases (e.g. amiABC) involved in septum splitting, and cell division proteins localized to the membrane-associated ring structure (e.g. ftsAEKHLQXYZ) showed differential interactions (i.e. aggravating in minimal and alleviating in rich medium) (Figure S6B). Conversely, we observed significantly fewer differential interactions (p-value≤0.05) among genes annotated to fatty acid degradation, stress response and chaperone pathways (Figure S6A), suggesting that functional connections between these genes remains to a larger extent unchanged.
Functional coupling of murein hydrolases to cell septation
Since functionally-related genes often have similar genetic profiles [19], [20], [40], the biological roles of incompletely characterized components can potentially be inferred based on correlation to annotated components. An illustrative example is a sub-network of strong alleviating interactions found in rich medium between the unannotated genes yceG and yebA and with several well-known cell division PG hydrolases (Figure 5A), each with distinct domain-architectures (Figure 5B). Binary fission in E. coli and other Gram-negatives depends on localized cell wall assembly at the division site by PG synthases e.g., penicillin-binding proteins [43]; subsequent splitting of these septa by PG hydrolases is required for cell division [44], [45]. Consistent with our genetic data, ultrastructure analysis revealed impaired membrane invagination and cytokinesis in yebA and yceG mutants (Figure 5C), with chains of daughter cells connected by uncleaved septa (Figure 5D and Figure S7A). Over-expression of either factor in trans fully rescued the phenotype (Figure 5D). Single yebA and yceG mutants also showed reduced sensitivity to ampicillin (Figure 5E), which blocks PG assembly, whereas wild-type E. coli rapidly lyse due to imbalanced PG hydrolase activity [44]. Consistent with joint promotion of septal PG splitting, growth curve assays confirmed an alleviating interaction between yebA and yceG in rich medium (Figure 5F).
Fig. 5. Novel hydrolase factors in septal PG splitting. 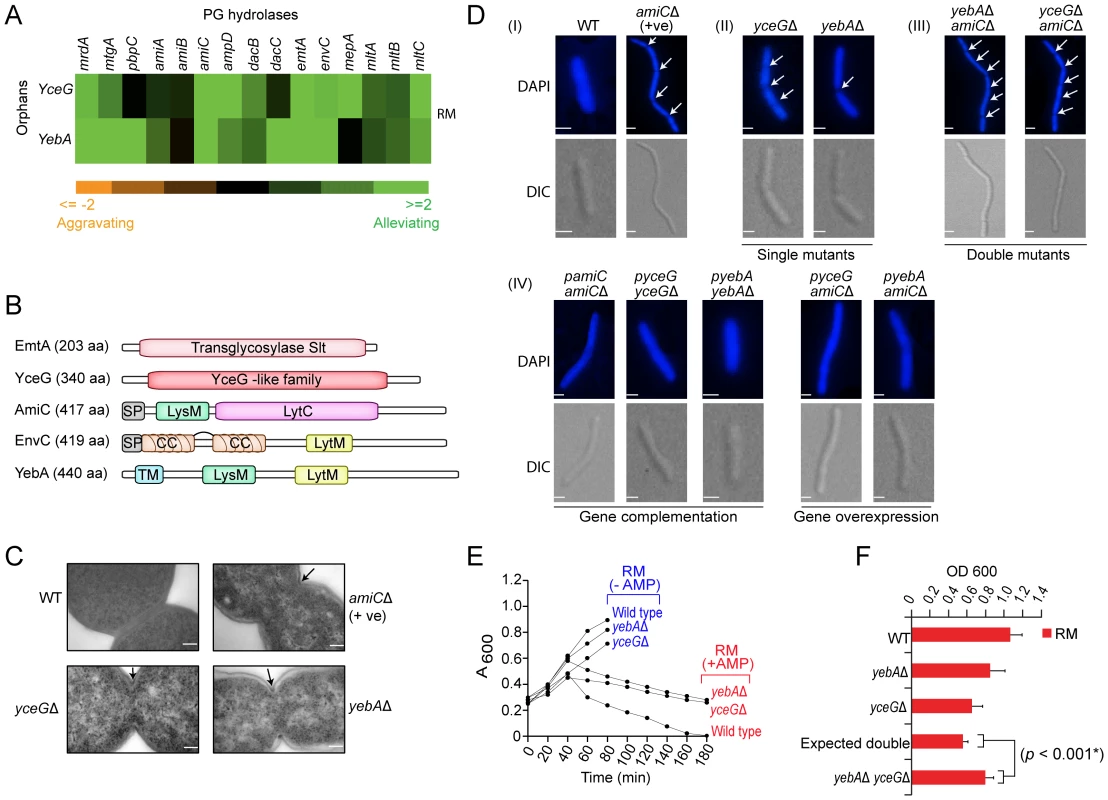
(A) Alleviating genetic interactions between annotated PG hydrolases and YceG/YebA. (B) Protein architecture (amino acid length indicated); domain structure of YceG unknown. CC, coiled coil; TM, transmembrane helix; SP, signal peptide. (C) Electron microscopy showing impaired membrane invagination and uncleaved PG septa (arrows) in E. coli mutants relative to wild-type cells; scale-bar 500 nm. (D) Cell division defects (arrows), phenotypic complementation (plasmid-based wild-type gene copy) or transgenic rescue. Strains stained with DAPI were visualized using a high content microscopy with differential interference contrast (DIC) and fluorescence optics; scale-bar 2 µm. (E) Differential strain growth with or without ampicillin (AMP, 5 µg/ml). (F) Alleviating double mutant interaction in liquid rich medium at 32°C over 24 h. Novel factors for OM integrity
A sub-network of strong aggravating interactions was also found in rich medium (Figure 6A and 6B), linking 3 unannotated genes (one encoding a putative β-barrel protein, ytfN and the other two lipoproteins, yfgH, yceK) to the lptDE complex involved in the final stages of LPS assembly at the OM outer leaflet [46]. Since impaired LPS destabilizes the envelope integrity [10], we challenged the mutant strains with vancomycin, an inhibitor of PG formation normally excluded by the intact OM of Gram-negatives [47]. Unlike drug-resistant wild-type E. coli, vancomycin induced morphological defects manifested by prominent mid-cell membranous bulges in single mutants and cell wall failure/lysis in double mutants (Figure 6C). Impaired OM integrity was also evident by drug hypersensitivity (Figure 6D), lowered OMP abundance (Figure 6E), and elevated levels of the σE-stress response DegP protease [10]. Taken together, these data point to the tight participation of yfgH, yceK, ytfN in LPS/OM formation and OM integrity
Fig. 6. Novel factors required for OM integrity. 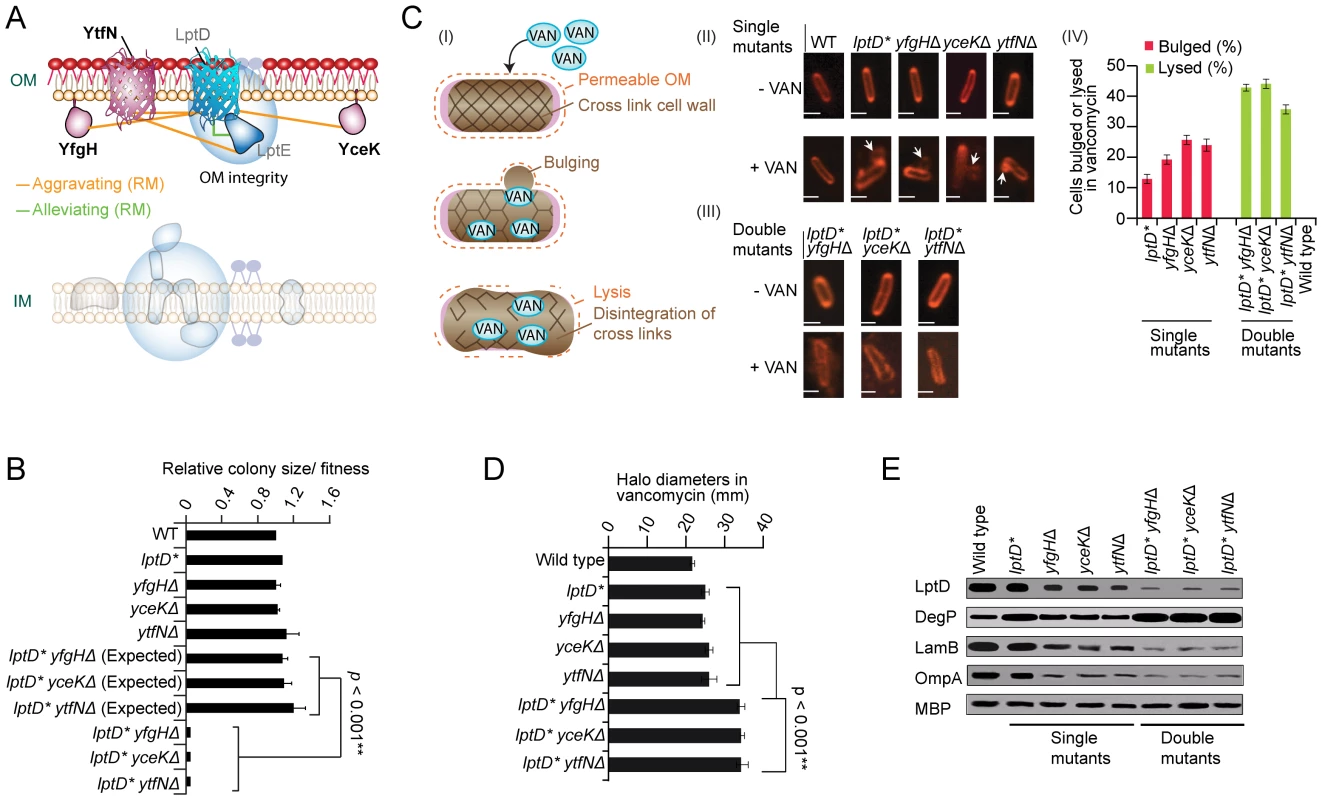
(A) Genetic interaction sub-network in rich medium. (B) Aggravating interactions between lptD and yfgH, yceK, and ytfN. (C) Proposed mechanism of drug hypersensitivity (I) modeled on [68]: envelope deficiency results in a porous OM (dashes), thereby allowing vancomycin (VAN) into the periplasm where it blocks PG cross-linking, weakening the cell wall and resulting in cytoplasmic membrane bulges in single mutant strains (II) or catastrophic cell wall failure and lysis in double mutants (III); scale bar equals 2 µm. Quantification (percentage) of bulged or lysed cells following vancomycin treatment according to strain genotype; error bars indicate standard deviation (IV). (D) Growth inhibition of strains treated with vancomycin. Graph shows mean drug disk halo diameter and standard deviation (error bars) in three independent experiments; p-value calculated using Student's t-test. (E) SDS-PAGE immunoblot analyses of marker protein levels in indicated strains during logarithmic growth in rich medium; periplasmic maltose-binding protein (MBP) used as loading control. YhjD participates in LPS transport
Another envelope gene of uncertain function, yhjD, which encodes a putative IMP with (5–6 TMH) previously implicated in Lipid A precursor IVA transport [48], showed a similar genetic profile (Table S6) and alleviating interactions (Figure 7A and 7B) with components of the lptBCFG complex that extracts LPS intermediates from the IM for passage to the OM [49], and aggravating interactions with other LPS transport factors (Figure S7B and S7C).
Fig. 7. YhjD is required for LPS transport. 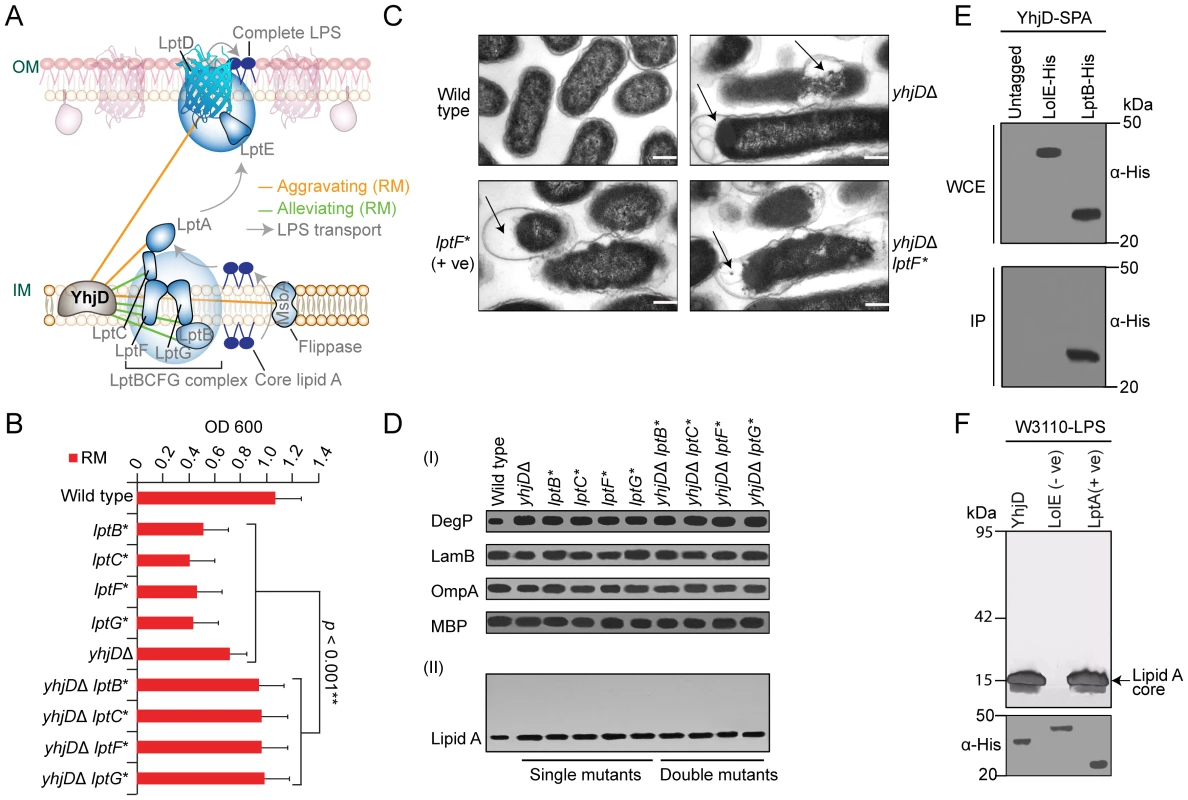
(A) Genetic interaction sub-network (rich medium) involving LPS transport factors. LPS is synthesized at the IM cytoplasmic face, flipped across the bilayer by MsbA, extracted by LptBCFG, and transported to the OM by LptA for final assembly by LptDE. (B) Strain growth in rich liquid medium at 32°C over 24 h. (C) Electron microscopy showing morphological alterations (arrows) in mutant E. coli strains relative to wild-type cells; scale-bar 500 nm. (D) SDS-PAGE immunoblots of marker proteins (I) and silver staining LPS (II). (E) Co-immunoprecipitation of SPA-tagged YhjD with His6-tagged LPS transport component. (F) LPS-binding by immobilized recombinant YhjD (test) and LptA (positive control) but not LolE (negative control). After extensive washing, bound LPS molecules were visualized by SDS-PAGE and silver staining. Mutant lptBCFG strains have morphologically perturbed envelopes [10], [50]. Transmission electron microscopy (TEM) likewise revealed a defective ultrastructure, membranous projections and periplasmic bodies characteristic of compromised LPS transport in strains lacking yhjD (Figure 7C). Like lptBCFG mutants [10], [50], strains lacking yhjD also showed an activated DegP stress response and accumulated intracellular lipid A precursors whereas OMP abundance was not affected (Figure 7D). Consistent with the alleviating epistasis, these defects were not exacerbated in yhjD lptBCFG double mutants.
Since alleviating interactions arise with in pathways or physical complexes [40], we affinity purified endogenous YhjD from detergent solubilized E. coli cell extracts to identify stably associated proteins (see Methods). As shown in Figure 7E, YhjD co-purified specifically with LptB, consistent with association as a multiprotein complex. Moreover, recombinant YhjD also bound selectively to core-lipid A (rough LPS) in vitro (Figure 7F). Since site-specific suppressor mutation in YhjD has been shown to reduce the toxic-side effects of lipid IVA accumulation by rendering the LPS transporter MsbA critical for lipid IVA trafficking [48], it is conceivable that the deletion of YhjD eliminates the buildup of toxic lipid A intermediates in lptBCFG mutants, resulting in alleviating phenotype.
Genetic networks capture regulatory relationships
Antisense sRNAs regulate gene expression by base-pairing to mRNAs via imperfect complementarity [51], often in response to environmental contingency such as stationary phase cells in nutrient rich medium or in minimal medium challenge [52], [53]. We therefore examined the genetic interactions of the 10 sRNAs in our networks (cyaR, dicF, gcvB, micA, micC, micF, omrA, oxyS, rybB, sgrS) with both verified and computationally-predicted targets (Table S10).
Cognate target-regulator pairs showed predominantly alleviating phenotypes under both culture conditions (Figure 8A). For example, oxyS with both the OM lipoprotein ybaY [54] and cls, which encodes cardiolipin synthase, whose levels are both significantly elevated in E. coli mutants lacking oxyS in rich and minimal medium (Figure 8B). However, condition-specific regulator-target relationships were also observed consistent with differential sRNA activity (Figure 8B). For instance, alleviating interactions were preferentially observed in rich medium between gcvB and mtr stationary phase culture cells, which encodes a tryptophan-specific permease feedback inhibited via the Trp repressor [55], and oppA, which encodes a periplasmic oligopeptide transporter. Deletion of gcvB causes constitutive expression of oppA [56], which is normally repressed in nutrient-rich conditions. Similarly, omrA displayed an alleviating phenotype in rich medium with iron transporters (fecCDE). Although essential for growth, iron is toxic and its import is tightly control [57].
Fig. 8. Target-regulator genetic interactions. 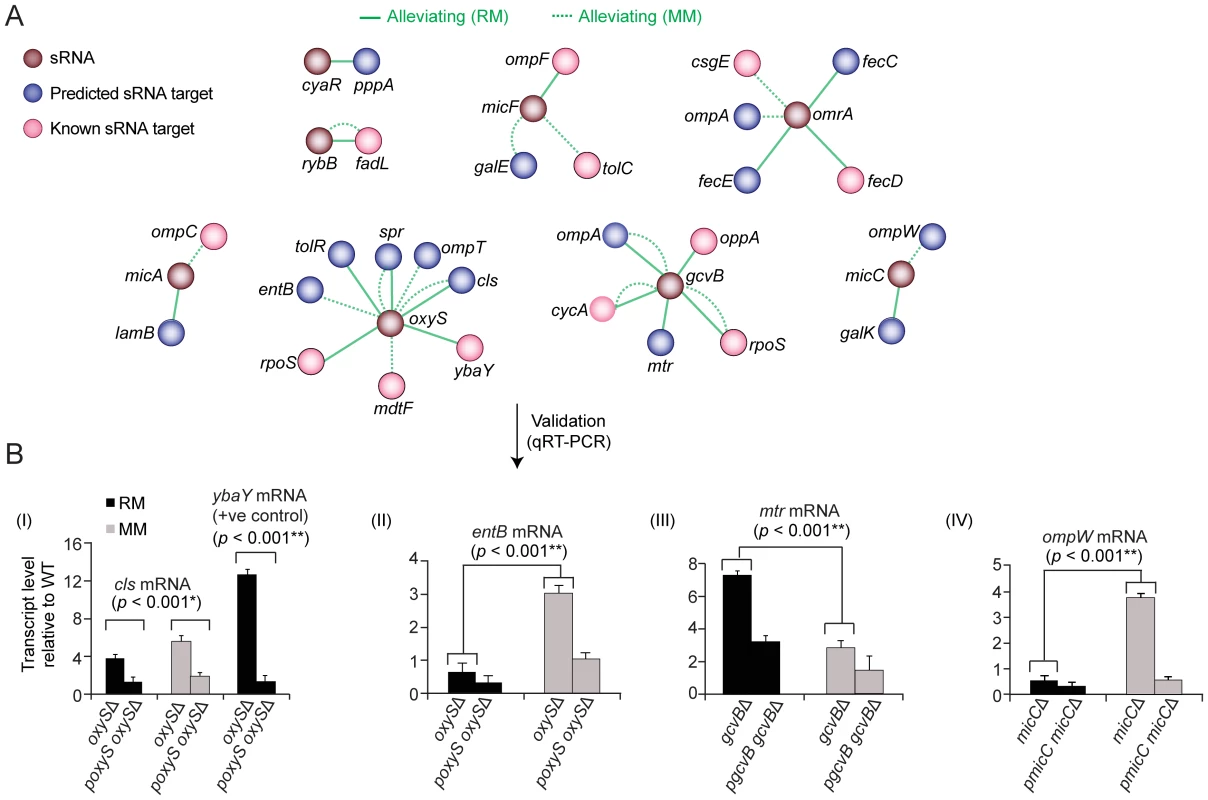
(A) Alleviating interactions between known (pink) or predicted (blue) gene targets and cognate sRNA regulators (brown). (B) Quantitative real-time PCR analysis of target transcript levels in regulator mutants on rich or minimal medium. Values indicate mean fold-change ± standard deviation (error bars) of triplicate biological measurements normalized to a housekeeping gene product (glutathione S-transferase). Conversely, the regulators micC and oxyS displayed alleviating interactions with certain targets preferentially in minimal medium (Figure 8A), implying repression is lost in nutrient-limiting conditions. We confirmed that the mRNA levels of ompW and entB increase substantively in minimal medium in micC and oxyS, mutants, respectively (Figure 8B). Collectively, these results demonstrate how quantitative genetic interaction maps can be used to probe regulatory relationships orchestrating cell envelope homeostasis.
Discussion
The cell envelope forms an essential interface between a microbe and its environment, or host in the case of pathogens. While E. coli has clearly evolved well-developed adaptive mechanisms to withstand diverse perturbations [2], our genetic results show that the K-12 laboratory isolate is unable to compensate for the loss of different combinations of individual cell envelope components and bioprocesses in culture. We have provided diverse lines of evidence supporting the unexpected biological participation of multiple novel factors in disparate core envelope processes. While additional follow-up mechanistic studies are warranted, the genetic interaction networks serve as a valuable resource for gleaning mechanistic and cell biological insights into E. coli envelope bioprocesses at the gene, pathway and physiological level.
These genetic interactions networks also inform on the global modular biological architecture of the bacterial cell envelope biosynthetic machinery, and its pronounced re-organization as an adaptive environmental response. Although our screens missed additional cases of epistasis emergent under alternate growth or stress conditions, the size and scope of the genetic map provides a fresh perspective into the functional interplay between convergent and compensatory systems that collectively ensure cell envelope assembly and integrity under two commonly studied culture conditions. The functional relationships we detected extend beyond established biochemical pathways and metabolic fluxes, and are consistent with the recently introduced notions of conditional and induced essentiality [58] and the substantive rewiring of genetic interaction networks in yeast following genotoxic stress [21]. Although the evolutionary basis for these emergent systems properties is unclear, it suggests a ubiquity of dynamic bioprocess crosstalk among microbes [3].
E. coli has an envelope architecture that is very similar to that of infectious species like Salmonella enterica (serovar Typhimurium), Pseudomonas aeruginosa and Neisseria meningitidis. Just as genetic screens in model eukaryotes have facilitated the characterization of conserved disease-related biological systems in humans [18], the generation of genetic interaction maps for E. coli suggests functional vulnerabilities that could potentially be exploited to combat these and other pathogens. For example, the coupling of cell wall and LPS glycoconjugate production we observed implies that a new family of peptidomimetic antibiotics (e.g. protegrin) targeting LPS assembly in P. aeruginosa [59] might be potentiated by inhibitors of either PG assembly or exopolysaccharide virulence factors analogous to CA in E. coli.
To facilitate exploration of these connectivity maps, our datasets are publicly accessible via a dedicated web portal (http://ecoli.med.utoronto.ca/eMap/CE/). The fact that a wide breadth of biological information could be derived from a defined collection of double mutants using a simple measure of colony growth provides strong motivation for future efforts aimed at examining other bacterial traits of broad biological, clinical and pharmacological significance.
Materials and Methods
Functional annotations
E. coli cell envelope gene targets suitable for screening were compiled based on published functional studies and database surveys [1], pathway annotations in EcoCyc, and relevant GO and GenProtEC annotations. Literature curated genetic interactions were manually compiled from low-throughput experimental studies. To generate functional association networks (see Protocol S7 and S8), high-quality E. coli PPI curated in the eNET, BIND, DIP, IntAct and MPI_LIT databases were compiled; microarray based mRNA transcript profiles were downloaded from the M3D database to derive a network of co-expressed genes based on correlation; and functional connections inferred by genomic context methods were computed as reported before [14]. Metabolic networks were reconstructed using Ecocyc annotations.
Strains, plasmids, and genetic screens
Bacterial strains and plasmids used in this study are listed in Table S11. The F - ‘recipient’ single gene deletion mutants were from the Keio knock-out strain collection [15], marked with kanamycin resistance (KanR), and from the recently developed ASKA single-gene deletion mutant library (Yamamoto et al., unpublished data) marked with chloramphenicol-resistance (CmR). Donor mutant strain construction, conjugation and imaging was performed essentially as described [23]. Briefly, to construct Hfr Cavalli (Hfr C) donor knockout mutants, λ-Red recombination was used to replace the entire coding sequence of each open reading frame of non-essential genes with a drug selection marker, whereas the selection cassette was integrated into 3′-UTR of essential genes to perturb transcript abundance [23]. In total, 505 ‘query’ gene mutations in Hfr C were systematically transferred via conjugation to an arrayed collection of 714 F - ‘recipient’ knockout and hypomorphic mutant strains [23]. Robotic colony pinning and drug selection procedures are detailed in Protocol S2. An “Hfr” donor (marked with KanR) isogenic with the E. coli single-gene deletion knock libraries was constructed for testing essentially as described [60].
Bioinformatics analysis
Plate image processing and colony size quantization were adapted from an automated image processing system originally devised for yeast [61]. Epistasis scores were calculated based on a multiplicative model [61] where a GI between a pair of genes (i, j) is identified if the fitness phenotype of the double mutant (Wij) deviates significantly from that predicted for non-interacting gene pairs (Wi×Wj). Enrichment was determined by contrasting the frequency of observed genetic interactions between or within functional modules against the expected frequencies according to the hypergeometric distribution using the Fisher's exact test [62] with correction for multiple hypothesis testing [63]. This enrichment approach was used to determine the optimal E-score cutoff values for selecting aggravating and alleviating genetic interactions. Additional details are provided in Protocols S2, S3, S4, S5, S6, S7.
Computational prediction of regulatory sRNA targets was performed using RNAup [64] from the Vienna RNA package (-Xp-w20-b parameters) to measure sequence complementarity between each sncRNA and the 5′ regions (−50 and +50 nt relative to translation start site) of all protein coding genes in E. coli. Genes were ranked according to free energy, with the lowest scoring predicted as targets. Known mRNA targets were compiled from published functional studies and the NPInter and sRNAMap databases.
Western blotting and protein affinity purification
Immunoblots were probed with polyclonal (LptD, OmpA, LamB, DegP) or monoclonal (MBP, LPS, His epitopes) antisera. Hexahistidine (His6)-tagged E. coli LptB or LolE fusion protein were expressed using an IPTG inducible promoter from a high copy pCA24N plasmid [65]. Sequential peptide affinity (SPA) tagging and purification of Fts, Sec and YhjD was performed essentially as previously described [66]. The affinity-purified proteins were subjected to gel-free liquid chromatography-electrospray-linear ion trap tandem mass spectrometry (LC-MS) to identify the stably and weakly associated interacting proteins with high sensitivity. Spectral searches were performed against a database containing complete set of E. coli protein-coding sequences and filtered for high-confidence matches essentially as described [66]. Details of the immunoprecipitation are provided in Protocol S9.
Phenotypic and drug interaction assays
For growth curve analyses, overnight cultures were inoculated into 100-well microtitre plates containing 100 µl of liquid medium, incubated with shaking at 32°C for over 24 hrs with optical density (600 nm) measured every 15 min using an automated Bioscreen-C (Thermolabsystems, Helsinki, Finland). For drug assays, overnight E. coli cultures in liquid LB medium were serially diluted and pinned onto solid LB agar plates in the absence or presence of drug (see Protocol S10). To examine cell morphology, strains were grown to log-phase (A600∼0.5) in LB medium at 32°C; 30 min prior to imaging, cultures were treated with vancomycin hydrochloride (1.5 µg/ml).
Cell structures were stained using membrane dye FM4-64 (Molecular Probes; 1 µg/ml) or of DAPI/ml (250 ng/ml), and 1.5 µl of cell suspension was spotted onto a glass slide for microscopy. Digital images were captured using a Quorum WaveFX Spinning Disc Confocal System. Cell length was measured using the Volocity program (Improvision Ltd.).
For drug interaction assays, drugs were tested over varying concentration ranges. The minimum inhibitory drug concentration was deemed to substantially reduce the growth rate by at least 20–50% relative to a no-drug control. Drug combinations were compared to single drugs using an isobologram on an arithmetic scale. For further details, see Protocol S11.
LPS/OM and ampicillin assays
The in vitro LPS purification and binding assay was based on an approach reported previously [67]. Total soluble LPS was separated by SDS-PAGE and examined by silver staining (see Protocol S12). Defects in OM protein biogenesis were assayed as previously described [26].
To assess ampicillin sensitivity, overnight cultures were diluted in fresh LB and grown at 32°C to an OD600 of ∼0.6. The cells were then diluted 1∶3 in fresh LB medium with or without 5 µg of ampicillin/ml and growth continued at 32°C, with OD600 measured every 20 min.
Electron microscopy
Cells were fixed with 2% glutaraldehyde in 0.1 M sodium cacodylate buffer overnight, post-fixed with 1% osmium tetroxide, dehydrated in a graded ethanol series followed by absolute propylene oxide, and embedded in Quetol-Spurr resin. Sections (90 nm thick) were cut on a RMC MT6000 ultramicrotome, stained with uranyl acetate and lead citrate, and viewed on a FEI CM100 TEM.
Quantitative real-time PCR (qRT-PCR)
To measure mRNA levels, target RNA was isolated from stationary phase mutant cultures grown in rich or minimal medium using standard hot phenol extraction procedure, followed by DNase I treatment. cDNA was synthesized from ∼0.5 µg total RNA using iScript™ cDNA synthesis kit with SYBR green supermix (Bio-Rad) following the manufacture's protocol. Primer sequences used for qRT-PCR were as follows: forward primer of cls gene, 5′ GATTATATTTCGCGTTCACGTCTG-3′, reverse primer of cls gene, 5′-TCCGACTAACGGCAGAATGTAA-3′; forward primer of ybaY gene, 5′ATTACCGTGAATGACAAACTGGTA-3′, reverse primer of ybaY gene, 5′ - CAGGTTGTTGTGTTGCTGAAATAG-3′; forward primer of mtr gene, 5′ - TCTGCATCACACCTTCGCAGAGAT-3′, reverse primer of mtr gene, 5′-TTACGCCAAGGAACGAACTCGCTA -3′; forward primer of ompW gene, 5′-GCGGCTTTGGCAGTAACAACTCTT-3′, reverse primer of ompW gene, 5′-TGATGAACGGTTGCAATATCGCCG-3′; forward primer of entB gene, CGCGACTACTGCAAACAGCACAAT-3′ and reverse primer of entB gene, 5′-ATCAGGTTGTCGTCATCGAACGGT-3′. A primer set specific to glutathione S - transferase (gst) (forward primer of gst gene, 5′ - CTTTGCCGTTAA CCCTAAGGG -3′; reverse primer of gst gene, 5′ - GCTGCAATGTGCTCTAACCC -3′) was used as an internal control for normalization. mRNA quantification was performed on a Rotor-Gene RG-300 qRT-PCR system (Corbett Research) under standard reaction conditions. Relative mRNA expression level were quantified by comparing the cycle threshold (Ct) values of each deletion mutant strain in rich medium or minimal medium to the Ct value of WT strain RNA, normalizing the sample value to gst expression. All reactions were performed independently from three biological replicates.
Supporting Information
Zdroje
1. Díaz-MejíaJJBabuMEmiliA 2009 Computational and experimental approaches to chart the Escherichia coli cell-envelope-associated proteome and interactome. FEMS Microbiol Rev 33 66 97
2. NicholsRJSenSChooYJBeltraoPZietekM 2011 Phenotypic landscape of a bacterial cell. Cell 144 143 156
3. SilhavyTJKahneDWalkerS 2010 The bacterial cell envelope. Cold Spring Harb Perspect Biol 2 a000414
4. HaselbeckRWallDJiangBKetelaTZyskindJ 2002 Comprehensive essential gene identification as a platform for novel anti-infective drug discovery. Curr Pharm Des 8 1155 1172
5. FalconerSBBrownED 2009 New screens and targets in antibacterial drug discovery. Curr Opin Microbiol 12 497 504
6. BabuMGreenblattJFEmiliAStrynadkaNCReithmeierRA 2010 Structure of a SLC26 Anion Transporter STAS Domain in Complex with Acyl Carrier Protein: Implications for E. coli YchM in Fatty Acid Metabolism. Structure 18 1450 1462
7. ParkJTUeharaT 2008 How bacteria consume their own exoskeletons (turnover and recycling of cell wall peptidoglycan). Microbiol Mol Biol Rev 72 211 227
8. RuizNKahneDSilhavyTJ 2009 Transport of lipopolysaccharide across the cell envelope: the long road of discovery. Nat Rev Microbiol 7 677 683
9. KnowlesTJScott-TuckerAOverduinMHendersonIR 2009 Membrane protein architects: the role of the BAM complex in outer membrane protein assembly. Nat Rev Microbiol 7 206 214
10. RuizNGronenbergLSKahneDSilhavyTJ 2008 Identification of two inner-membrane proteins required for the transport of lipopolysaccharide to the outer membrane of Escherichia coli. Proc Natl Acad Sci U S A 105 5537 5542
11. SperandeoPVillaRMartoranaASamalikovaMGrandoriR 2010 New insights into the Lpt machinery for LPS transport to the cell surface: LptA-LptC interaction and LptA stability as a sensors of a properly assembled transenvelope complex. J Bacteriol In Press
12. TypasABanzhafMvan den Berg van SaparoeaBVerheulJBiboyJ 2010 Regulation of peptidoglycan synthesis by outer-membrane proteins. Cell 143 1097 1109
13. WuTMalinverniJRuizNKimSSilhavyTJ 2005 Identification of a multicomponent complex required for outer membrane biogenesis in Escherichia coli. Cell 121 235 245
14. HuPJangaSCBabuMDiaz-MejiaJJButlandG 2009 Global functional atlas of Escherichia coli encompassing previously uncharacterized proteins. PLoS Biol 7 e1000096 doi:10.1371/journal.pbio.1000096
15. BabaTAraTHasegawaMTakaiYOkumuraY 2006 Construction of Escherichia coli K-12 in-frame, single-gene knockout mutants: the Keio collection. Mol Syst Biol 2 2006.0008
16. HartwellLHHopfieldJJLeiblerSMurrayAW 1999 From molecular to modular cell biology. Nature 402 C47 52
17. GirgisHSHottesAKTavazoieS 2009 Genetic architecture of intrinsic antibiotic susceptibility. PLoS ONE 4 e5629 doi:10.1371/journal.pone.0005629
18. DixonSJCostanzoMBaryshnikovaAAndrewsBBooneC 2009 Systematic mapping of genetic interaction networks. Annu Rev Genet 43 601 625
19. CostanzoMBaryshnikovaABellayJKimYSpearED 2010 The genetic landscape of a cell. Science 327 425 431
20. SchuldinerMCollinsSRThompsonNJDenicVBhamidipatiA 2005 Exploration of the function and organization of the yeast early secretory pathway through an epistatic miniarray profile. Cell 123 507 519
21. BandyopadhyaySMehtaMKuoDSungMKChuangR 2010 Rewiring of genetic networks in response to DNA damage. Science 330 1385 1389
22. BabuMMussoGDiaz-MejiaJJButlandGGreenblattJF 2009 Systems-level approaches for identifying and analyzing genetic interaction networks in Escherichia coli and extensions to other prokaryotes. Mol Biosyst 12 1439 1455
23. ButlandGBabuMDíaz-MejíaJJBohdanaFPhanseS 2008 eSGA: E. coli synthetic genetic array analysis. Nat Methods 5 789 795
24. BeltraoPCagneyGKroganNJ 2010 Quantitative genetic interactions reveal biological modularity. Cell 141 739 745
25. BandyopadhyaySKelleyRKroganNJIdekerT 2008 Functional maps of protein complexes from quantitative genetic interaction data. PLoS Comput Biol 4 e1000065 doi:10.1371/journal.pcbi.1000065
26. RuizNFalconeBKahneDSilhavyTJ 2005 Chemical conditionality: a genetic strategy to probe organelle assembly. Cell 121 307 317
27. SklarJGWuTKahneDSilhavyTJ 2007 Defining the roles of the periplasmic chaperones SurA, Skp, and DegP in Escherichia coli. Genes Dev 21 2473 2484
28. EbersbachGGalliEMoller-JensenJLoweJGerdesK 2008 Novel coiled-coil cell division factor ZapB stimulates Z ring assembly and cell division. Mol Microbiol 68 720 735
29. PriceMNArkinAPAlmEJ 2006 The life-cycle of operons. PLoS Genet 2 e96 doi:10.1371/journal.pgen.0020096
30. FaithJJHayeteBThadenJTMognoIWierzbowskiJ 2007 Large-scale mapping and validation of Escherichia coli transcriptional regulation from a compendium of expression profiles. PLoS Biol 5 e8 doi:10.1371/journal.pbio.0050008
31. LamarcheMGKimSHCrepinSMourezMBertrandN 2008 Modulation of hexa-acyl pyrophosphate lipid A population under Escherichia coli phosphate (Pho) regulon activation. J Bacteriol 190 5256 5264
32. RowlandSLFuXSayedMAZhangYCookWR 2000 Membrane redistribution of the Escherichia coli MinD protein induced by MinE. J Bacteriol 182 613 619
33. HigashiKIshigureHDemizuRUemuraTNishinoK 2008 Identification of a spermidine excretion protein complex (MdtJI) in Escherichia coli. J Bacteriol 190 872 878
34. CorbinBDWangYBeuriaTKMargolinW 2007 Interaction between cell division proteins FtsE and FtsZ. J Bacteriol 189 3026 3035
35. WhitfieldC 2006 Biosynthesis and assembly of capsular polysaccharides in Escherichia coli. Annu Rev Biochem 75 39 68
36. HugIFeldmanMF 2010 Analogies and homologies in lipopolysaccharide and glycoprotein biosynthesis in bacteria. Glycobiology 21 138 151
37. MajdalaniNGottesmanS 2005 The Rcs phosphorelay: a complex signal transduction system. Annu Rev Microbiol 59 379 405
38. GottesmanSStoutV 1991 Regulation of capsular polysaccharide synthesis in Escherichia coli K12. Mol Microbiol 5 1599 1606
39. BergJMTymoczkoJLStryerL 2002 Protein Turnover and Amino Acid Catabolism New York W. H. Freeman 1100
40. CollinsSRMillerKMMaasNLRoguevAFillinghamJ 2007 Functional dissection of protein complexes involved in yeast chromosome biology using a genetic interaction map. Nature 446 806 810
41. TauraTItoK 1991 Does protein secretion activity vary during the cell cycle of Escherichia coli? J Biochem 109 811 815
42. OliverDBBeckwithJ 1981 E. coli mutant pleiotropically defective in the export of secreted proteins. Cell 25 765 772
43. ChungHSYaoZGoehringNWKishonyRBeckwithJ 2009 Rapid beta-lactam-induced lysis requires successful assembly of the cell division machinery. Proc Natl Acad Sci U S A 106 21872 21877
44. HeidrichCUrsinusABergerJSchwarzHHoltjeJV 2002 Effects of multiple deletions of murein hydrolases on viability, septum cleavage, and sensitivity to large toxic molecules in Escherichia coli. J Bacteriol 184 6093 6099
45. UeharaTDinhTBernhardtTG 2009 LytM-domain factors are required for daughter cell separation and rapid ampicillin-induced lysis in Escherichia coli. J Bacteriol 191 5094 5107
46. ChngSSRuizNChimalakondaGSilhavyTJKahneD 2010 Characterization of the two-protein complex in Escherichia coli responsible for lipopolysaccharide assembly at the outer membrane. Proc Natl Acad Sci U S A 107 5363 5368
47. VaaraMNurminenM 1999 Outer membrane permeability barrier in Escherichia coli mutants that are defective in the late acyltransferases of lipid A biosynthesis. Antimicrob Agents Chemother 43 1459 1462
48. MamatUMeredithTCAggarwalPKühlAKirchhoffP 2008 Single amino acid substitutions in either YhjD or MsbA confer viability to 3-deoxy-d-manno-oct-2-ulosonic acid-depleted Escherichia coli. Mol Microbiol 67 633 648
49. NaritaSTokudaH 2009 Biochemical characterization of an ABC transporter LptBFGC complex required for the outer membrane sorting of lipopolysaccharides. FEBS Lett 583 2160 2164
50. SperandeoPLauFKCarpentieriADe CastroCMolinaroA 2008 Functional analysis of the protein machinery required for transport of lipopolysaccharide to the outer membrane of Escherichia coli. J Bacteriol 190 4460 4469
51. VogelJPapenfortK 2006 Small non-coding RNAs and the bacterial outer membrane. Curr Opin Microbiol 9 605 611
52. EddySR 2001 Non-coding RNA genes and the modern RNA world. Nat Rev Genet 2 919 929
53. GeissmannTChevalierCCrosMJBoissetSFechterP 2009 A search for small noncoding RNAs in Staphylococcus aureus reveals a conserved sequence motif for regulation. Nucleic Acids Res 37 7239 7257
54. TjadenBGoodwinSSOpdykeJAGuillierMFuDX 2006 Target prediction for small, noncoding RNAs in bacteria. Nucleic Acids Res 34 2791 2802
55. SarseroJPWookeyPJPittardAJ 1991 Regulation of expression of the Escherichia coli K-12 mtr gene by TyrR protein and Trp repressor. J Bacteriol 173 4133 4143
56. PulvermacherSCStaufferLTStaufferGV 2009 Role of the sRNA GcvB in regulation of cycA in Escherichia coli. Microbiology 155 106 114
57. NoinajNGuillierMBarnardTJBuchananSK 2010 TonB-dependent transporters: regulation, structure, and function. Annu Rev Microbiol 64 43 60
58. TischlerJLehnerBFraserAG 2008 Evolutionary plasticity of genetic interaction networks. Nat Genet 40 390 391
59. SrinivasNJetterPUeberbacherBJWerneburgMZerbeK 2010 Peptidomimetic antibiotics target outer-membrane biogenesis in Pseudomonas aeruginosa. Science 327 1010 1013
60. TypasANicholsRJSiegeleDAShalesMCollinsSR 2008 High-throughput, quantitative analyses of genetic interactions in E. coli. Nat Methods 5 781 787
61. CollinsSRSchuldinerMKroganNJWeissmanJS 2006 A strategy for extracting and analyzing large-scale quantitative epistatic interaction data. Genome Biol 7 R63
62. Le MeurNGentlemanR 2008 Modeling synthetic lethality. Genome Biol 9 R135
63. BenjaminiYHochbergY 1995 Controlling the false discovery rate: a practical and powerful approach to multiple testing. J Roy Statist Soc Ser B 57 589 300
64. MucksteinUTaferHHackermullerJBernhartSHStadlerPF 2006 Thermodynamics of RNA-RNA binding. Bioinformatics 22 1177 1182
65. KitagawaMAraTArifuzzamanMIoka-NakamichiTInamotoE 2005 Complete set of ORF clones of Escherichia coli ASKA library (a complete set of E. coli K-12 ORF archive): unique resources for biological research. DNA Res 12 291 299
66. BabuMButlandGPogoutseOLiJGreenblattJF 2009 Sequential peptide affinity purification system for the systematic isolation and identification of protein complexes from Escherichia coli. Methods Mol Biol 564 373 400
67. TranAXTrentMSWhitfieldC 2008 The LptA protein of Escherichia coli is a periplasmic lipid A-binding protein involved in the lipopolysaccharide export pathway. J Biol Chem 283 20342 20349
68. HuangKCMukhopadhyayRWenBGitaiZWingreenNS 2008 Cell shape and cell-wall organization in Gram-negative bacteria. Proc Natl Acad Sci U S A 105 19282 19287
Štítky
Genetika Reprodukční medicína
Článek vyšel v časopisePLOS Genetics
Nejčtenější tento týden
2011 Číslo 11
-
Všechny články tohoto čísla
- De Novo Origins of Human Genes
- Duplication Hotspots Are Associated with Late-Replicating Regions of the Genome
- De Novo Origin of Human Protein-Coding Genes
- Cyclin D/CDK4 and Cyclin E/CDK2 Induce Distinct Cell Cycle Re-Entry Programs in Differentiated Muscle Cells
- Short Day–Mediated Cessation of Growth Requires the Downregulation of AINTEGUMENTALIKE1 Transcription Factor in Hybrid Aspen
- Physiological IRE-1-XBP-1 and PEK-1 Signaling in Larval Development and Immunity
- Role of Pirh2 in Mediating the Regulation of p53 and c-Myc
- Signatures of Environmental Genetic Adaptation Pinpoint Pathogens as the Main Selective Pressure through Human Evolution
- FOXO Regulates Organ-Specific Phenotypic Plasticity In
- Heritable Epigenetic Variation among Maize Inbreds
- Foxn1 Regulates Lineage Progression in Cortical and Medullary Thymic Epithelial Cells But Is Dispensable for Medullary Sublineage Divergence
- Attenuation of the Sensing Capabilities of PhoQ in Transition to Obligate Insect–Bacterial Association
- A Novel Protein LZTFL1 Regulates Ciliary Trafficking of the BBSome and Smoothened
- Activation of Bmp2-Smad1 Signal and Its Regulation by Coordinated Alteration of H3K27 Trimethylation in -Induced Senescence
- Histone H3K56 Acetylation, CAF1, and Rtt106 Coordinate Nucleosome Assembly and Stability of Advancing Replication Forks
- The SUN Protein Mps3 Is Required for Spindle Pole Body Insertion into the Nuclear Membrane and Nuclear Envelope Homeostasis
- Evidence-Based Annotation of Gene Function in MR-1 Using Genome-Wide Fitness Profiling across 121 Conditions
- Effect of Host Species on the Distribution of Mutational Fitness Effects for an RNA Virus
- Pch2 Acts through Xrs2 and Tel1/ATM to Modulate Interhomolog Bias and Checkpoint Function during Meiosis
- SOX9 Governs Differentiation Stage-Specific Gene Expression in Growth Plate Chondrocytes via Direct Concomitant Transactivation and Repression
- from the Aphid : A Missing Link from Facultative to Obligate Insect Endosymbiont
- Recessive Antimorphic Alleles Overcome Functionally Redundant Loci to Reveal Function in Flowers and Meristems
- Over-Expression of DSCAM and COL6A2 Cooperatively Generates Congenital Heart Defects
- Consequences of Eukaryotic Enhancer Architecture for Gene Expression Dynamics, Development, and Fitness
- Distinct Genetic Architectures for Male and Female Inflorescence Traits of Maize
- Capture of MicroRNA–Bound mRNAs Identifies the Tumor Suppressor miR-34a as a Regulator of Growth Factor Signaling
- For Male , Sperm Activation Is a “Just-in-Time” Event
- PcG Complexes Set the Stage for Epigenetic Inheritance of Gene Silencing in Early S Phase before Replication
- The Gene Contains Hotspots for L1 Endonuclease-Dependent Insertion
- Relative Burden of Large CNVs on a Range of Neurodevelopmental Phenotypes
- Multiple Means to the Same End: The Genetic Basis of Acquired Stress Resistance in Yeast
- Genome-Wide Crossover Distribution in Meiosis Reveals Sex-Specific Patterns along Chromosomes
- TRY-5 Is a Sperm-Activating Protease in Seminal Fluid
- Homologs of Retinoblastoma-Associated Protein 46/48 Associate with a Histone Deacetylase to Act Redundantly in Chromatin Silencing
- Genetic Interaction Maps in Reveal Functional Crosstalk among Cell Envelope Biogenesis Pathways
- The ERI-6/7 Helicase Acts at the First Stage of an siRNA Amplification Pathway That Targets Recent Gene Duplications
- PBX1 Genomic Pioneer Function Drives ERα Signaling Underlying Progression in Breast Cancer
- PLOS Genetics
- Archiv čísel
- Aktuální číslo
- Informace o časopisu
Nejčtenější v tomto čísle- Evidence-Based Annotation of Gene Function in MR-1 Using Genome-Wide Fitness Profiling across 121 Conditions
- De Novo Origins of Human Genes
- Capture of MicroRNA–Bound mRNAs Identifies the Tumor Suppressor miR-34a as a Regulator of Growth Factor Signaling
- TRY-5 Is a Sperm-Activating Protease in Seminal Fluid
Kurzy
Zvyšte si kvalifikaci online z pohodlí domova
Současné možnosti léčby obezity
nový kurzAutoři: MUDr. Martin Hrubý
Všechny kurzyPřihlášení#ADS_BOTTOM_SCRIPTS#Zapomenuté hesloZadejte e-mailovou adresu, se kterou jste vytvářel(a) účet, budou Vám na ni zaslány informace k nastavení nového hesla.
- Vzdělávání



