-
Články
Top novinky
Reklama- Vzdělávání
- Časopisy
Top články
Nové číslo
- Témata
Top novinky
Reklama- Videa
- Podcasty
Nové podcasty
Reklama- Kariéra
Doporučené pozice
Reklama- Praxe
Top novinky
ReklamaMorphogenesis in Fungal Pathogenicity: Shape, Size, and Surface
article has not abstract
Published in the journal: . PLoS Pathog 8(12): e32767. doi:10.1371/journal.ppat.1003027
Category: Pearls
doi: https://doi.org/10.1371/journal.ppat.1003027Summary
article has not abstract
Introduction
Morphological changes are required for eukaryotic pathogens to cause disease. However, it is only now becoming clear how such transitions are linked to virulence in human pathogenic fungi. Changing cell size and shape are strategies employed by many of these fungi to survive in the environment and serendipitously also within the host. Conserved signaling pathways regulate morphogenic differentiation in response to environmental and host physiological stimuli. The alterations in cell-surface composition during morphogenesis, in addition to cell size and shape, further link virulence with morphogenesis.
Morphotype Transition Is Associated with Fungal Virulence
Changes in morphology are required by diverse microbes to be successful pathogens, and the well-known examples include bacterial pathogen Chlamydia trachomatis and protozoan parasite Plasmodium falciparum. Morphotype transitions are also required for fungi to cause disease in plants. For example, the rice blast ascomycete Magnaporthe oryzae undergoes a series of morphotype changes (conidia→appressoria→penetration peg→invasive hyphae) for infection. The corn smut basidiomycete Ustilago maydis grows as a yeast in vitro and infects plants only in the hyphal form after mating.
The majority of fungi grow either filamentously by apical extension or unicellularly by budding or fission. A few human pathogenic fungi switch between these two growth forms, and these are called dimorphic fungi. Here, “morphotype transition” or “dimorphism” refers to the ability to switch between two or more different growth forms. It has been established in the “classic” dimorphic pathogens (listed in the clade of Ascomycota in Figure 1) that proper morphotype transition is required for virulence: blocking transitions by chemicals or genetic mutations attenuates or abolishes their ability to cause diseases [1]–[6].
Fig. 1. Fungal pathogens from different phyla exhibit a dimorphic lifestyle. 
Candida albicans is a commensal or opportunistic pathogen, distinguishing it from the other species that are acquired from environmental exposure. The factors regulating dimorphism are provided: H2O− dehydration; H2O+ aqueous environment. Morphotype-associated pathogenicity is likely more widespread than just those species traditionally classified as dimorphic. For instance, Cryptococcus neoformans, a basidiomycetous pathogen, has both a yeast and a filamentous form. Early studies on natural Cryptococcus isolates implicated an inverse relationship between filamentation and virulence (see [7] and references therein). A molecular link between morphogenesis and virulence in this fungus was obtained recently through the characterization of the transcription factor Znf2. Overexpression of the ZNF2 gene confers filamentation and abolishes the ability of this fungus to cause disease, while the deletion of the ZNF2 gene locks cells in the yeast form and enhances virulence [8], [9]. The existence of morphotype-associated pathogenicity is also observed in some zygomycetes [10] and in dermatophytes [11]. Thus, it appears that pathogens of different major phyla in the fungal kingdom have adopted dimorphism as a common pathogenic strategy. The underlying question is what advantage this provides to these species.
Changes in Cell Size Impact Fungal Pathogenicity
Cell size affects multiple steps during infection and disease progression. As infectious propagules, spores of small sizes are ideal for dispersal and entry into host lungs. For instance, Cryptococcus spores (1–2 µm in diameter) are easily lodged into the lower respiratory tract and phagocytosed by lung macrophages [12], [13]. Spores then germinate into encapsulated yeasts of varied sizes. These yeast cells can withstand the antimicrobial activities mediated by macrophages, proliferate intracellularly, and escape from the host cell. Mycelia of Histoplasma capsulatum produce two types of conidia, macroconidia (8–16 µm), and microconidia (2–5 µm). The microconidia enter the alveolar spaces and germinate into budding yeasts that can persist intracellularly in phagocytotic cells, a critical step for Histoplasma infection [14]. However, for fungal pathogens that typically infect the upper respiratory tract, gastrointestinal tract, or subcutaneous tissues, cell size has a different impact on virulence. The zygomycete Mucor circinelloides f. lusitanicus is a heterothallic fungus with (+) or (−) mating types. Asexual spores produced by (−) strains are larger and can germinate inside macrophages and lyse host cells. Accordingly, the spores produced by (−) strains are more virulent [10]. A key observation is that allowing small spores to germinate and develop into large cells in vitro, prior to the infection, increases the virulence of the inoculum [10].
Heterogeneity in cell size is a bet-hedging strategy that poises a population of cells of identical genotype for different contingencies in the host. For instance, a subset of Cryptococcus yeast cells enlarge from the typical 3–5 µm to up to 100 µm in diameter during later stages of lung infection [15], [16]. These giant cryptococcal cells can evade phagocytosis and better tolerate oxidative and nitrosative stresses. A mutant defective in producing these large cells has attenuated virulence [17]. Similarly, Paracoccidioides brasiliensis wild-type populations show varied cell size, and mutant cells homogenously reduced in size are more susceptible to phagocytosis by macrophages and are unable to cause disease [18].
Changing Cell Shape or Size May Have Evolved as a Response to Environmental Stresses
The majority of the human fungal pathogens are environmental saprophytes (Figure 1), and their ability to survive in a mammalian host is a consequence of selective pressures posed by their environment. Changing cell shape or size is likely an adaptive trait that is also advantageous when these pathogens encounter the host. Accordingly, signaling pathways that play important roles in coordinating morphological changes and virulence (see the next section) are typically involved in sensing environmental stresses.
Of these stresses, high body temperature presents a major barrier for many fungal species to infect mammals. This barrier may explain the rarity of fungal diseases in humans, in contrast to the prevalence of fungal diseases in plants, insects, amphibians, or even bats during hibernation [19]. Interestingly, elevated temperatures are the primary signal directing dimorphic fungi to switch from the saprotrophic filamentous phase to the pathogenic yeast phase (Figure 1).
Conserved Signaling Pathways Orchestrate both Morphogenesis and Virulence
Ancient signaling pathways that respond to environmental cues control fungal morphogenesis and virulence. These include the cAMP/PKA pathway, the PKC cell wall integrity pathway, the pheromone sensing pathway, the Ca2+-calcineurin pathway, and the two-component pathways [2], [20] that are found across the fungal kingdom. Most of these signaling pathways are involved in regulating temperature stress. However, those pathways are also present in nonpathogenic species: how temperature is translated into morphogenesis in the pathogens remains an open question and the responsible thermosensors elusive. One promising candidate thermosensor is the histidine kinase Drk1, which triggers the temperature-dependent dimorphic transition in Histoplasma and Blastomyces [2]. Interestingly, its bacterial homolog DesK is the first documented histidine kinase that senses temperature fluctuations by detecting changes in membrane thickness [21]. The heat-shock chaperone Hsp90 plays important roles in the temperature-regulated dimorphism in Candida, and regulators Ryp1-3 are critical for the temperature-induced filament-to-yeast switch of Histoplasma [5], [6], [22]. However, these molecules likely sense temperatures indirectly via some unfolded proteins or act downstream of thermosensors. Besides proteins, RNAs with temperature-sensitive structures could also act as thermosensors, as shown in some bacterial pathogens [23].
In addition to exogenous factors (e.g., temperature, osmolarity, serum, pH, and sugar), endogenously produced small molecules such as quorum-sensing molecules (QSMs) affect fungal morphogenesis. QSMs regulate biological processes in a population density–dependent manner. In C. albicans, farnesol inhibits yeast-to-filament conversion in correlation with the inoculum size [24]. Tyrosol, on the other hand, stimulates yeast-to-filament conversion [25]. Multiple QSMs (e.g., farnesol, tyrosol, and morphogenic autoregulatory substance) likely fine-tune the dimorphic transition in C. albicans. Inoculum size is shown to affect morphotype transition in other dimorphic fungi [25], implying that QS systems are widely used to synchronize fungal cellular differentiation. Given that “anti-quorum sensing” is being exploited to curb bacterial virulence and that QS quenchers are less likely to select for resistance compared to conventional antimicrobial drugs [26], investigation into the QS response systems in fungi could help design effective treatments against mycoses.
Alterations in the Cell Surface Are Underpinned by Cellular Morphology
Changes in cell shape or size are a visual manifestation of alterations in cell-wall properties. Although important, the physical aspects of shape and size may not by themselves be the key factor controlling virulence. Rather, alterations in cell surface molecules during morphogenesis help fungi to adapt to host conditions and to avoid or defend against host immune attacks. In B. dermatitidis, H. capsulatum, and P. brasiliensis, filament-to-yeast transition is accompanied by the increased production of α-1,3-glucan [14], [27], which masks the immunostimulatory β-glucan [28]. In C. albicans, the pathogenic hyphal growth form does not expose β-glucan to trigger antimicrobial responses [29]. In C. neoformans, spores germinate into yeast cells with enlarged capsule under host-relevant conditions. The capsule is immune-suppressive and antiphagocytotic, and it conceals antigens that could be recognized by the host.
A repertoire of cell-surface adhesion proteins (adhesins) are highly regulated during morphotype switches: some adhesins are specifically expressed in the pathogenic form, while others in the saprotrophic/commensal form. In M. oryzae, the integral membrane protein Pls1 regulates adhesion with Teflon-binding affinity to the plant surface and is specifically expressed in the appressorium [30], a cell type used for penetrating the plant tissue. In the human pathogenic fungi, adhesin Bad1 of B. dermatitidis is specifically expressed in the pathogenic yeast form [1], and it controls multiple processes during infection [31]. Adhesin Hwp1 in C. albicans is specifically expressed in hyphae to assist fungal attachment to epithelial cells, and Hwp1 is required for systemic candidiasis [32]. Als3, another adhesin expressed in Candida hyphae, binds to multiple host receptors and induces its own endocytosis to facilitate fungal penetration of epithelial cells [33]. By contrast, the adhesin Cfl1 in C. neoformans is associated with nonpathogenic filaments, and forced expression of Cfl1 in the pathogenic yeast form attenuates virulence [8]. Cfl1 is a downstream target of Znf2, which controls the expression of multiple adhesion genes in Cryptococcus (unpublished results). Accordingly, the znf2Δ mutant is more virulent than the wild type, even though both strains are in the yeast form during infection [8], [9]. Taken together, alterations in cell surface in addition to the physical cell shape and size likely underpin the link between morphogenesis and fungal virulence.
In the pathogenic fungi that are acquired from environmental sources, the filamentous form is generally not pathogenic. Nonetheless, adhesins or other molecules on filaments or spores help shape the initial interactions between the host and the pathogen (Figure 2), which establishes subsequent host-pathogen interactions. Success by these fungi to colonize the host, establish infections, and disseminate systemically is a combination of the downregulation of filament - or spore-specific molecules and upregulation of the yeast-specific ones. Therefore, investigation of cell-surface molecules specific to each morphological form will provide a new opportunity to comprehend host-fungus interactions.
Fig. 2. The transition between morphotype and virulence in fungi. 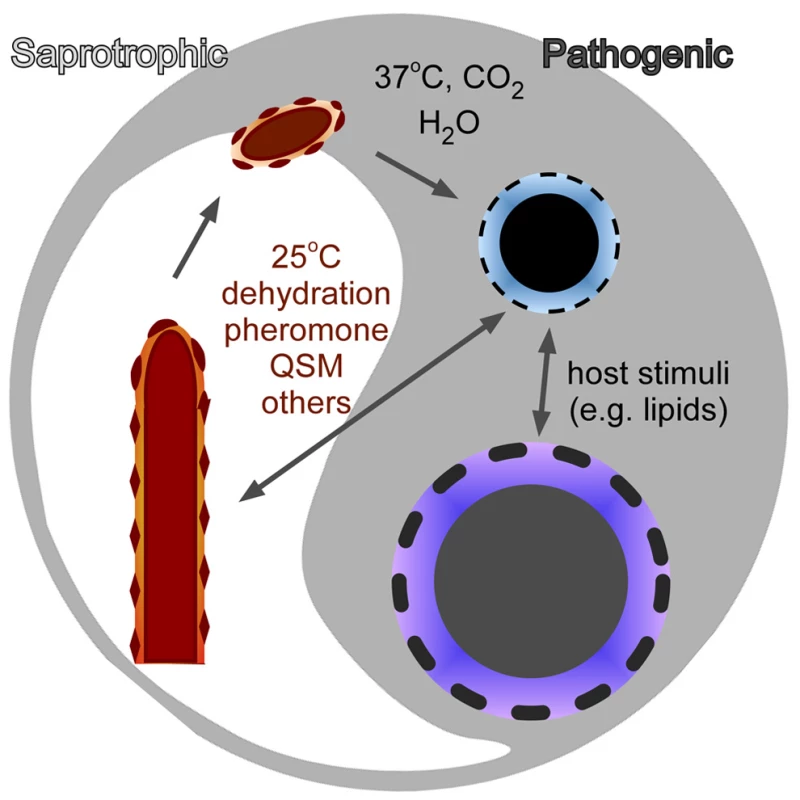
Each morphotype has a unique cell-surface structure and composition. The differences in cell surface reflect differences in fungal cell physiology and contribute to the differences in the host immune responses elicited by these cells.
Zdroje
1. KleinBS, TebbetsB (2007) Dimorphism and virulence in fungi. Curr Opin Microbiol 10 : 314–319.
2. NemecekJC, WuthrichM, KleinBS (2006) Global control of dimorphism and virulence in fungi. Science 312 : 583–588.
3. LiuH, KohlerJ, FinkGR (1994) Suppression of hyphal formation in Candida albicans by mutation of a STE12 homolog. Science 266 : 1723–1726.
4. LoHJ, KohlerJR, DiDomenicoB, LoebenbergD, CacciapuotiA, et al. (1997) Nonfilamentous C. albicans mutants are avirulent. Cell 90 : 939–949.
5. NguyenVQ, SilA (2008) Temperature-induced switch to the pathogenic yeast form of Histoplasma capsulatum requires Ryp1, a conserved transcriptional regulator. Proc Natl Acad Sci U S A 105 : 4880–4885.
6. WebsterRH, SilA (2008) Conserved factors Ryp2 and Ryp3 control cell morphology and infectious spore formation in the fungal pathogen Histoplasma capsulatum. Proc Natl Acad Sci U S A 105 : 14573–14578.
7. LinX (2009) Cryptococcus neoformans: morphogenesis, infection, and evolution. Infect Genet Evol 9 : 401–416.
8. WangL, ZhaiB, LinX (2012) The link between morphotype transition and virulence in Cryptococcus neoformans. PLoS Pathog 8: e1002765 doi:10.1371/journal.ppat.1002765.
9. LinX, JacksonJC, FeretzakiM, XueC, HeitmanJ (2010) Transcription factors Mat2 and Znf2 operate cellular circuits orchestrating opposite - and same-sex mating in Cryptococcus neoformans. PLoS Genet 6: e1000953 doi:10.1371/journal.pgen.1000953.
10. LiCH, CervantesM, SpringerDJ, BoekhoutT, Ruiz-VazquezRM, et al. (2011) Sporangiospore size dimorphism is linked to virulence of Mucor circinelloides. PLoS Pathog 7: e1002086 doi:10.1371/journal.ppat.1002086.
11. LillisJV, DawsonES, ChangR, WhiteCRJr (2010) Disseminated dermal Trichophyton rubrum infection - an expression of dermatophyte dimorphism? J Cutan Pathol 37 : 1168–1169.
12. GilesSS, DagenaisTR, BottsMR, KellerNP, HullCM (2009) Elucidating the pathogenesis of spores from the human fungal pathogen Cryptococcus neoformans. Infect Immun 77 : 3491–3500.
13. VelagapudiR, HsuehYP, Geunes-BoyerS, WrightJR, HeitmanJ (2009) Spores as infectious propagules of Cryptococcus neoformans. Infect Immun 77 : 4345–4355.
14. SeiderK, HeykenA, LuttichA, MiramonP, HubeB (2010) Interaction of pathogenic yeasts with phagocytes: survival, persistence and escape. Curr Opin Microbiol 13 : 392–400.
15. OkagakiLH, StrainAK, NielsenJN, CharlierC, BaltesNJ, et al. (2010) Cryptococcal cell morphology affects host cell interactions and pathogenicity. PLoS Pathog 6: e1000953 doi:10.1371/journal.ppat.1000953.
16. ZaragozaO, García-RodasR, NosanchukJD, Cuenca-EstrellaM, Rodríguez-TudelaJL, et al. (2010) Fungal cell gigantism during mammalian infection. PLoS Pathog 6: e1000945 doi:10.1371/journal.ppat.1000945.
17. CrabtreeJN, OkagakiLH, WiesnerDL, StrainAK, NielsenJN, et al. (2012) Titan cell production enhances the virulence of Cryptococcus neoformans. Infect Immun 80 : 3776–85 doi:10.1128/IAI.00507-12.
18. AlmeidaAJ, CunhaC, CarmonaJA, Sampaio-MarquesB, CarvalhoA, et al. (2009) Cdc42p controls yeast-cell shape and virulence of Paracoccidioides brasiliensis. Fungal Genet Biol 46 : 919–926.
19. BergmanA, CasadevallA (2012) Mammalian endothermy optimally restricts fungi and metabolic costs. MBio 1: e00212–10 doi:10.1128/mBio.00212-10.
20. BahnYS, KojimaK, CoxGM, HeitmanJ (2006) A unique fungal two-component system regulates stress responses, drug sensitivity, sexual development, and virulence of Cryptococcus neoformans. Mol Biol Cell 17 : 3122–3135.
21. AlbanesiD, MartinM, TrajtenbergF, MansillaMC, HaouzA, et al. (2009) Structural plasticity and catalysis regulation of a thermosensor histidine kinase. Proc Natl Acad Sci U S A 106 : 16185–16190.
22. ShapiroRS, CowenLE (2012) Uncovering cellular circuitry controlling temperature-dependent fungal morphogenesis. Virulence 3 : 400–404 doi:10.4161/viru.20979.
23. BöhmeK, SteinmannR, KortmannJ, SeekircherS, HerovenAK, et al. (2012) Concerted actions of a thermo-labile regulator and a unique intergenic RNA thermosensor control Yersinia virulence. PLoS Pathog 8: e1002518 doi:10.1371/journal.ppat.1002518.
24. HornbyJM, JensenEC, LisecAD, TastoJJ, JahnkeB, et al. (2001) Quorum sensing in the dimorphic fungus Candida albicans is mediated by farnesol. Appl Environ Microbiol 67 : 2982–2992.
25. AlbuquerqueP, CasadevallA (2012) Quorum sensing in fungi–a review. Med Mycol 50 : 337–345.
26. RomeroM, AcunaL, OteroA (2012) Patents on quorum quenching: interfering with bacterial communication as a strategy to fight infections. Recent Pat Biotechnol 6 : 2–12.
27. KanetsunaF, CarbonellLM (1971) Cell wall composition of the yeastlike and mycelial forms of Blastomyces dermatitidis. J Bacteriol 106 : 946–948.
28. RappleyeCA, EissenbergLG, GoldmanWE (2007) Histoplasma capsulatum a-(1,3)-glucan blocks innate immune recognition by the b-glucan receptor. Proc Natl Acad Sci U S A 104 : 1366–1370.
29. GantnerBN, SimmonsRM, UnderhillDM (2005) Dectin-1 mediates macrophage recognition of Candida albicans yeast but not filaments. EMBO J 24 : 1277–1286.
30. ClergeotPH, GourguesM, CotsJ, LauransF, LatorseMP, et al. (2001) PLS1, a gene encoding a tetraspanin-like protein, is required for penetration of rice leaf by the fungal pathogen Magnaporthe grisea. Proc Natl Acad Sci U S A 98 : 6963–6968.
31. BrandhorstTT, WuthrichM, Finkel-JimenezB, WarnerT, KleinBS (2004) Exploiting type 3 complement receptor for TNF-alpha suppression, immune evasion, and progressive pulmonary fungal infection. J Immunol 173 : 7444–7453.
32. StaabJF, BradwaySD, FidelPL, SundstromP (1999) Adhesive and mammalian transglutaminase substrate properties of Candida albicans Hwp1. Science 283 : 1535–1538.
33. ZhuW, PhanQT, BoontheungP, SolisNV, LooJA, et al. (2012) EGFR and HER2 receptor kinase signaling mediate epithelial cell invasion by Candida albicans during oropharyngeal infection. Proc Natl Acad Sci U S A 109 : 14194–14199.
Štítky
Hygiena a epidemiologie Infekční lékařství Laboratoř
Článek vyšel v časopisePLOS Pathogens
Nejčtenější tento týden
2012 Číslo 12- Jak souvisí postcovidový syndrom s poškozením mozku?
- Měli bychom postcovidový syndrom léčit antidepresivy?
- Farmakovigilanční studie perorálních antivirotik indikovaných v léčbě COVID-19
- 10 bodů k očkování proti COVID-19: stanovisko České společnosti alergologie a klinické imunologie ČLS JEP
-
Všechny články tohoto čísla
- Virus-Encoded microRNAs: An Overview and a Look to the Future
- Reactive Oxygen Species Production and Survivorship in with Artificial Infection Types
- Zinc Exploitation by Pathogenic Fungi
- Attenuated Typhimurium Lacking the Pathogenicity Island-2 Type 3 Secretion System Grow to High Bacterial Numbers inside Phagocytes in Mice
- The Polyfunctionality of Human Memory CD8+ T Cells Elicited by Acute and Chronic Virus Infections Is Not Influenced by Age
- How the Fly Balances Its Ability to Combat Different Pathogens
- MiniCD4 Microbicide Prevents HIV Infection of Human Mucosal Explants and Vaginal Transmission of SHIV in Cynomolgus Macaques
- Bidirectional Transfer of RNAi between Honey Bee and : Gene Silencing Reduces Population
- Global Gene Transcriptome Analysis in Vaccinated Cattle Revealed a Dominant Role of IL-22 for Protection against Bovine Tuberculosis
- Morphogenesis in Fungal Pathogenicity: Shape, Size, and Surface
- Inflammatory Responses Associated with the Induction of Cerebral Malaria: Lessons from Experimental Murine Models
- News from the Fungal Front: Wall Proteome Dynamics and Host–Pathogen Interplay
- Blood Flukes Exploit Peyer's Patch Lymphoid Tissue to Facilitate Transmission from the Mammalian Host
- Parallels in Intercellular Communication in Oomycete and Fungal Pathogens of Plants and Humans
- Influenza Human Monoclonal Antibody 1F1 Interacts with Three Major Antigenic Sites and Residues Mediating Human Receptor Specificity in H1N1 Viruses
- PLOS Pathogens
- Archiv čísel
- Aktuální číslo
- Informace o časopisu
Nejčtenější v tomto čísle- Influenza Human Monoclonal Antibody 1F1 Interacts with Three Major Antigenic Sites and Residues Mediating Human Receptor Specificity in H1N1 Viruses
- Parallels in Intercellular Communication in Oomycete and Fungal Pathogens of Plants and Humans
- Virus-Encoded microRNAs: An Overview and a Look to the Future
- Reactive Oxygen Species Production and Survivorship in with Artificial Infection Types
Kurzy
Zvyšte si kvalifikaci online z pohodlí domova
Současné možnosti léčby obezity
nový kurzAutoři: MUDr. Martin Hrubý
Všechny kurzyPřihlášení#ADS_BOTTOM_SCRIPTS#Zapomenuté hesloZadejte e-mailovou adresu, se kterou jste vytvářel(a) účet, budou Vám na ni zaslány informace k nastavení nového hesla.
- Vzdělávání



