-
Články
Top novinky
Reklama- Vzdělávání
- Časopisy
Top články
Nové číslo
- Témata
Top novinky
Reklama- Videa
- Podcasty
Nové podcasty
Reklama- Volná místa
Doporučené pozice
Reklama- Praxe
Top novinky
ReklamaShared Information between Residues Is Sufficient to Detect Pairwise Epistasis in a Protein
article has not abstract
Published in the journal: . PLoS Genet 12(12): e32767. doi:10.1371/journal.pgen.1006471
Category: Formal Comment
doi: https://doi.org/10.1371/journal.pgen.1006471Summary
article has not abstract
In a comment [1] on our manuscript “Strong Selection Significantly Increases Epistatic Interactions in the Long-Term Evolution of a Protein” [2], Dr. Crona challenges our assertion that shared entropy (that is, information) between two residues implies epistasis between those residues by constructing an explicit example of three loci (say A, B, and C), in which A and B are epistatically linked (leading to shared entropy between A and B) and A and C are also epistatically dependent (leading to shared entropy between A and C) so that loci B and C are correlated (share entropy). She goes on to assert that (as per her examples) even though there will be correlations (and thus shared entropy) between the residues at loci B and C, there is no pairwise epistasis between loci B and C, contradicting our assertion in [1] that shared entropy implies epistasis.
The disagreement is based on two different interpretations of the meaning of pairwise epistasis, and the comment gives us an opportunity to discuss those.
We do not disagree that epistasis refers to mutational effects that are conditional on the states of other alleles. In our paper, we are in particular interested in pairwise epistasis—that is, how the fitness effects of mutations at two loci depend on each other. Of course, the dependence between those two loci could depend on the state of many other alleles in the genome. The different interpretations of epistasis hinge upon whether a quantitative assessment of the epistasis between two loci should be conditional on the state of other loci in the genome, or whether instead we should consider the state of these loci averaged over what their state would be in a population at mutation–selection balance. Crona’s example helps us illustrate that distinction.
A fitness landscape constructed in the supplementary information of [1] (example 1) has three loci: A, B, and C. The values given are (note that in [1], the log-transformed relative fitnesses are given instead):
The values are judiciously chosen such that w000w011w010w001=w100w111w110w101=1.
Crona defines the epistasis between loci B and C as either
orNote that these values are conditional on the state of locus A, but for the case discussed here they happen to be equal and vanishing. Clearly, this is a special case. In general, the pairwise epistasis conditional on the state of another locus can depend on that state, and if there are n other binary loci, then there could be in principle 2n different values for the pairwise epistasis. Surely this is not satisfactory, as pairwise epistasis then would not be defined. Instead, pairwise epistasis should be unconditional on the state of other loci in the genome. How do you calculate this?
We assert that pairwise epistasis between two loci should depend on the fitness effect of mutations at those loci where the states of the other loci are determined by mutation–selection balance in a population. In other words, we assert that fitness effects should be measured by the effect on the growth rate of a population. For the three locus system, the fitness of the BC system depends on the frequency of the A = 0 allele and the A = 1 allele in the population. Let p0 stand for the frequency of the A = 0 allele, with p0 + p1 = 1. Then
These four values can be used to calculate the epistasis between loci B and C unconditional on the state of A as
We plot this quantity in Fig 1 as a function of the frequency of the A = 0 allele p0 and see that it is everywhere positive except for p0 = 0 or p0 = 1, which are the conditional epistasis values of Crona (Eqs 2 and 3).
Fig. 1. Unconditional epistasis. 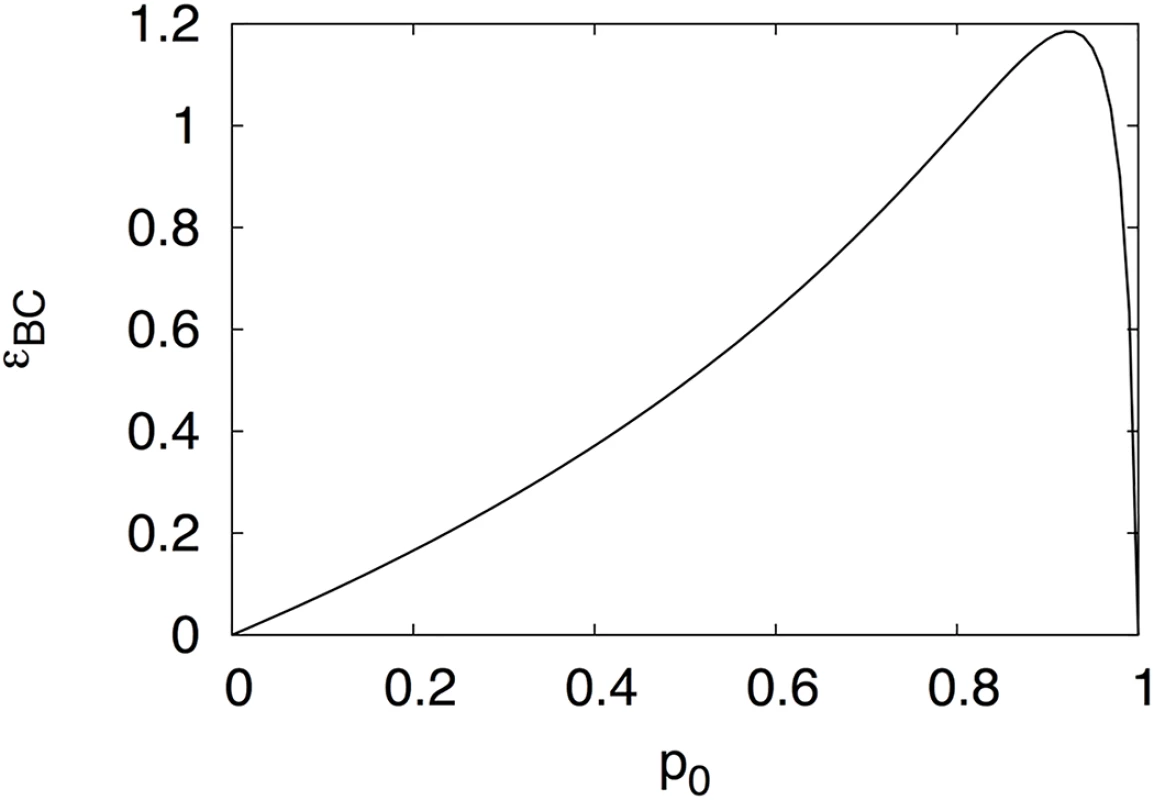
Pairwise epistasis between loci B and C calculated using Eqs 4 and 5 as a function of locus A allele frequency p0, with fitnesses as in Eq 1. However, given the fitness landscape Eq 1, these extreme values (a population composed purely of one allelic state of A) are impossible. As long as the mutation rate is nonvanishing, there will always be a mixture of both alleles at locus A. Indeed, Table 1 of the supplementary information of [1], which tabulates an evolutionary simulation on that precise landscape, makes that point for us. Crona finds that p0 ≈ 0.998 in equilibrium, leading to εBC ≈ 0.191, which is nonvanishing. Thus, the positive shared entropy between those loci is indeed sufficient to determine nonvanishing pairwise epistasis between them. We also remark that at that frequency p0, the information between B and C is exceedingly small: I≈0.0013.
Needless to say, the example discussed here is a fairly contrived one, and we show in Fig 9 of [2] that the correlation between epistasis and information is robust when testing random fitness landscapes. The same argument holds for examples 2 and 3 in the comment [1].
In conclusion, the assertion in [1] that detecting epistasis via shared entropy gives false positives for epistasis is based on a calculation of conditional epistasis, a concept that is ambiguous at best as it depends on the allelic state of all the other loci on the genome and could take on arbitrary values. If epistasis is calculated by averaging over the allelic state of the other loci, then our assertion that correlation (positive information) implies positive pairwise epistasis holds without exception.
Zdroje
1. Crona K. Epistasis and Entropy. PLoS Genet. 2016;12(12):e1006322. doi: 10.1371/journal.pgen.1006322
2. Gupta A, Adami C. Strong Selection Significantly Increases Epistatic Interactions in the Long-Term Evolution of a Protein. PLoS Genet. 2016;12(12):e1005960.
Štítky
Genetika Reprodukční medicína
Článek Epistasis and Entropy
Článek vyšel v časopisePLOS Genetics
Nejčtenější tento týden
2016 Číslo 12
-
Všechny články tohoto čísla
- Bringing Editors to Preprint Servers
- Epistasis and Entropy
- Middle Age Has Its Advantages
- Craniofacial Ciliopathies and the Interpretation of Hedgehog Signal Transduction
- Comment on " Is Dispensable for Mouse Development"
- Shared Information between Residues Is Sufficient to Detect Pairwise Epistasis in a Protein
- A Hox-Embedded Long Noncoding RNA: Is It All Hot Air?
- Deep Reads: Favorites from a Few Different Shelves
- Buffers Inherent Left/Right Asymmetry Ensuring Symmetric Forelimb Formation
- Intracellular Zn(II) Intoxication Leads to Dysregulation of the PerR Regulon Resulting in Heme Toxicity in
- Mutations in the Heme Exporter Cause Sensory Neurodegeneration with Loss of Pain Perception
- PLOS Genetics
- Archiv čísel
- Aktuální číslo
- Informace o časopisu
Nejčtenější v tomto čísle- Intracellular Zn(II) Intoxication Leads to Dysregulation of the PerR Regulon Resulting in Heme Toxicity in
- Craniofacial Ciliopathies and the Interpretation of Hedgehog Signal Transduction
- Mutations in the Heme Exporter Cause Sensory Neurodegeneration with Loss of Pain Perception
- Shared Information between Residues Is Sufficient to Detect Pairwise Epistasis in a Protein
Kurzy
Zvyšte si kvalifikaci online z pohodlí domova
Současné možnosti léčby obezity
nový kurzAutoři: MUDr. Martin Hrubý
Všechny kurzyPřihlášení#ADS_BOTTOM_SCRIPTS#Zapomenuté hesloZadejte e-mailovou adresu, se kterou jste vytvářel(a) účet, budou Vám na ni zaslány informace k nastavení nového hesla.
- Vzdělávání








