-
Články
Top novinky
Reklama- Vzdělávání
- Časopisy
Top články
Nové číslo
- Témata
Top novinky
Reklama- Videa
- Podcasty
Nové podcasty
Reklama- Kariéra
Doporučené pozice
Reklama- Praxe
Top novinky
ReklamaEpigenetically-Inherited Centromere and Neocentromere DNA Replicates Earliest in S-Phase
Eukaryotic centromeres are maintained at specific chromosomal sites over many generations. In the budding yeast Saccharomyces cerevisiae, centromeres are genetic elements defined by a DNA sequence that is both necessary and sufficient for function; whereas, in most other eukaryotes, centromeres are maintained by poorly characterized epigenetic mechanisms in which DNA has a less definitive role. Here we use the pathogenic yeast Candida albicans as a model organism to study the DNA replication properties of centromeric DNA. By determining the genome-wide replication timing program of the C. albicans genome, we discovered that each centromere is associated with a replication origin that is the first to fire on its respective chromosome. Importantly, epigenetic formation of new ectopic centromeres (neocentromeres) was accompanied by shifts in replication timing, such that a neocentromere became the first to replicate and became associated with origin recognition complex (ORC) components. Furthermore, changing the level of the centromere-specific histone H3 isoform led to a concomitant change in levels of ORC association with centromere regions, further supporting the idea that centromere proteins determine origin activity. Finally, analysis of centromere-associated DNA revealed a replication-dependent sequence pattern characteristic of constitutively active replication origins. This strand-biased pattern is conserved, together with centromere position, among related strains and species, in a manner independent of primary DNA sequence. Thus, inheritance of centromere position is correlated with a constitutively active origin of replication that fires at a distinct early time. We suggest a model in which the distinct timing of DNA replication serves as an epigenetic mechanism for the inheritance of centromere position.
Published in the journal: . PLoS Genet 6(8): e32767. doi:10.1371/journal.pgen.1001068
Category: Research Article
doi: https://doi.org/10.1371/journal.pgen.1001068Summary
Eukaryotic centromeres are maintained at specific chromosomal sites over many generations. In the budding yeast Saccharomyces cerevisiae, centromeres are genetic elements defined by a DNA sequence that is both necessary and sufficient for function; whereas, in most other eukaryotes, centromeres are maintained by poorly characterized epigenetic mechanisms in which DNA has a less definitive role. Here we use the pathogenic yeast Candida albicans as a model organism to study the DNA replication properties of centromeric DNA. By determining the genome-wide replication timing program of the C. albicans genome, we discovered that each centromere is associated with a replication origin that is the first to fire on its respective chromosome. Importantly, epigenetic formation of new ectopic centromeres (neocentromeres) was accompanied by shifts in replication timing, such that a neocentromere became the first to replicate and became associated with origin recognition complex (ORC) components. Furthermore, changing the level of the centromere-specific histone H3 isoform led to a concomitant change in levels of ORC association with centromere regions, further supporting the idea that centromere proteins determine origin activity. Finally, analysis of centromere-associated DNA revealed a replication-dependent sequence pattern characteristic of constitutively active replication origins. This strand-biased pattern is conserved, together with centromere position, among related strains and species, in a manner independent of primary DNA sequence. Thus, inheritance of centromere position is correlated with a constitutively active origin of replication that fires at a distinct early time. We suggest a model in which the distinct timing of DNA replication serves as an epigenetic mechanism for the inheritance of centromere position.
Introduction
Centromeres are essential components of eukaryotic chromosomes required for proper chromosome segregation to daughter cells. Lack of a functional centromere, or the presence of multiple centromeres, renders chromosomes unstable and prone to mis-segregation and breakage. This genome instability is associated with carcinogenesis and can also result in cell death. An intriguing property of most eukaryotic centromeres that remains poorly explained is their mode of inheritance. In principle, the functional identity of a single locus on a chromosome, such as a centromere, requires that locus to have at least one unique property that distinguishes it from other loci on that chromosome. While a primary DNA consensus sequence would be sufficient to define a single locus per chromosome, most eukaryotic centromeres are not defined at the DNA sequence level (reviewed in [1]–[5]). Thus, for instance, centromeres on different chromosomes in any one species do not share primary DNA sequence between them; furthermore, centromeric DNA sequence diverges between closely-related species while centromeric loci remain syntenic; in rare cases, centromere proteins move to new loci that do not normally function as centromeres. These neocentromeres, which remain stable at their new ectopic loci, have been observed in humans as well as in several model organisms [6]–[8].
Rather than a specific DNA sequence, a unique, conserved histone H3 variant, termed CENP-A/CenH3 (Cse4 in yeasts), distinguishes eukaryotic centromeres from the rest of the chromosome and has inspired the majority of current models of epigenetic centromere inheritance. These models propose that centromeric chromatin structure contains the information necessary to form and maintain centromeres at a given locus. One model suggests that CENP-A and histone H3 are expressed and/or deposited at different times during the cell cycle [9]–[12]. Consistent with this, S. pombe CENP-A expression reaches maximal levels just before the G1-S boundary [11] and can be deposited in either G1/early S-phase, or via a different pathway in G2 [12]. A related hypothesis suggests that centromeric DNA might replicate at a distinct time during S-phase, and that this may be coordinated with the timing of CENP-A deposition [13] (reviewed in [14]). However, studies in flies and mammalian cells that utilized microscopy measurements of BrdU incorporation to follow the replication timing of centromeric DNA failed to detect a distinct time of replication at centromeres [9], [15]–[17] and thus such models were abandoned. It is important to note that these experiments may lack the resolution and precision necessary to detect replication events within specific regions of the genome. Replication timing microarrays could provide the necessary precision, however centromeres in most eukaryotes typically span hundreds to thousands of kilobases of highly repetitive DNA and are usually not fully sequenced, thereby obviating such analyses. In contrast, centromeres in the pathogenic yeast Candida albicans exhibit all the hallmarks of epigenetically inherited centromeres, yet are short (∼ 3 kb), simple and fully sequenced [7], [18]–[21], making this yeast an attractive model system for the study of centromeric DNA properties.
In this study, we used C. albicans to study the replication of centromeric DNA. We found that on all chromosomes, the centromere was associated with a replication origin that fires first, and well before all other origins, on that chromosome. Manipulating Cse4 binding by either deletion of a canonical centromere locus or by placing it under the control of a conditional promoter revealed that centromere determinants attract replication components and specify early DNA replication. In addition, we describe a sequence feature of C. albicans centromeres - asymmetrical nucleotide composition - that is indicative of stable replication activity over evolutionary time scales. Using phylogenetic comparison, we provide evidence linking the epigenetic inheritance of centromere position with replication activity. Thus, DNA replication timing can serve as the basis for the inheritance of functional centromeres at specific chromosomal sites, representing a novel mechanism of epigenetic inheritance.
Results
Centromeres replicate at a distinct time during S-phase
We determined the genome-wide DNA replication timing profile at high temporal and spatial resolution for all eight Candida albicans chromosomes (Figure 1A). Briefly, asynchronous log phase cells were sorted by fluorescence activated cell sorting into G1 and S phase fractions. DNA from 2×106 cells from each fraction was extracted, differentially labeled with fluorescent dyes and hybridized to genomic tiling arrays. The data were smoothed and replication timing was displayed as a function of chromosome position. In these replication profiles, peaks represent replication origins and the height of each peak reflects the relative timing and/or efficiency of replication from that origin. When applied to S. cerevisiae, this method identifies known replication origins at a resolution of ∼5 Kb ([22]; Materials and Methods).
Fig. 1. Centromeres are first to replicate in Candida albicans. 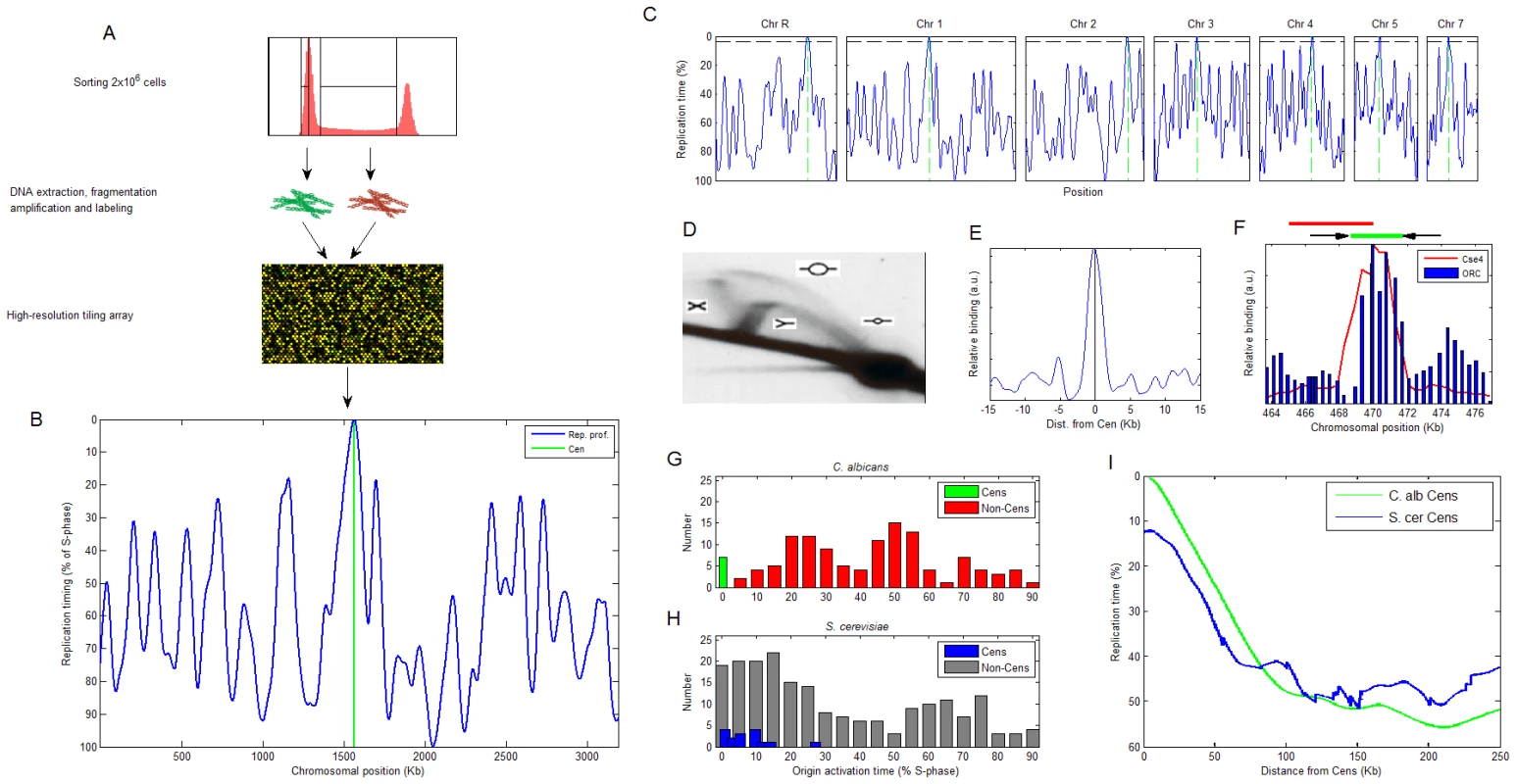
(A) Experimental method for measuring DNA replication timing. The earlier a certain locus replicates, the higher its DNA content will be when averaged over an asynchronous population of S-phase cells. Therefore, measuring copy number along the chromosomes in S-phase cells enables deduction of the genomic DNA replication timing program [22]. (B) The replication profile of chromosome 1. Peaks are predicted origin locations with their height corresponding to activation time. (C) The replication profiles of all chromosomes other than Chr6 (Materials and Methods and Figure S1). Green: centromeres. Horizontal black line: replication timing of the first non-centromeric origin activated in the genome, to denote the clear separation of timing for centromeric versus other origins. (D) 2D gel identifies replication intermediates in CEN5. The replication structures represented by each arc are shown [32]. The position of the probe used for the 2D gel is shown in red above (F). (E) ORC ChIP-chip values averaged over all chromosomes, aligned at the GC skew zero-intersection point (see text). See Figure S2 for the individual chromosome data. (F) ChIP of ORC retrieves CEN5 sequence. The ChIP result for Cse4 [7] is shown for comparison. The location of the centromere (green line) and flanking inverted repeats (black arrows) are shown above. (G,H) Distribution of relative activation times for centromeric and non-centromeric replication origins in (G) C. albicans and (H) S. cerevisiae (data from [22]). (I) Centromere position effect. The replication profiles exhibited a striking feature: centromeres were always proximal to a predicted replication origin and remarkably, these centromeric origins were the first to fire on each chromosome. Furthermore, the activation timing of these centromere-associated origins was clearly separate from the activation time of non-centromeric origins (Figure 1B and 1C and Figure S1). The colocalization of replication origins with centromere position is further supported by several lines of evidence. First, two-dimensional DNA gels detected a replication bubble structure, indicative of an active origin, at the chromosome 5 centromere DNA region (CEN5) (Figure 1D). Second, genome-wide chromatin immunoprecipitation (ChIP-chip) experiments of the Origin Recognition Complex (ORC), that is essential for replication initiation, revealed the presence of ORC within the centromere regions of each of the eight chromosomes (Figure 1E and Figure S2). These ORC binding sites were among the strongest in the genome (Figure S2 and data not shown). Third, ChIP followed by quantitative PCR of CEN5 detected ORC localization within CEN5 in correlation with the position of Cse4 (Figure 1F; [7]). Thus, centromeres in C. albicans contain replication origins that are the first to replicate on each chromosome.
The distinct replication timing of the centromeres is manifested in the distribution of origin activation times in the C. albicans genome: non-centromeric origins are largely absent from the beginning of S-phase and are otherwise activated throughout S-phase (Figure 1G). In contrast, in S. cerevisiae (that has genetically-inherited point centromeres), most origins are activated in the beginning of S-phase, at a time that is not distinct from that of centromere-proximal origins (Figure 1H; [23]). Thus, C. albicans achieves genomic replication with significantly fewer early-activated origins relative to S. cerevisiae. However, the paucity of early origins does not result in a longer S-phase in C. albicans compared to S. cerevisiae. This could be explained by a difference in the distribution of active origins with different activation timing along the chromosome: in S. cerevisiae, early origins tend to cluster in close proximity, ultimately resulting in ∼2-fold more active origins per DNA length than in the C. albicans genome. In C. albicans, the spacing of early and late origins is more evenly interspersed (Figure S3). This more regular spacing of origins along the chromosome is presumably more efficient, enabling utilization of fewer origins in C. albicans.
Previous work described a replication timing position effect in S. cerevisiae [23], in which centromeres are located within a region of several tens of kilobases of early replicating DNA (and see below). A similar, albeit significantly stronger effect is evident in C. albicans (Figure 1I) where a region of up to ∼100 kb surrounding the centromere replicates earlier than the genomic average, further emphasizing the distinctiveness of centromere replication timing in C. albicans.
Centromeres are effectors of DNA replication timing
The colocalization of centromeres and early replication origins could be caused by replication origins recruiting centromeric determinants. Alternatively, centromeres could recruit replication determinants. To address the direction of dependency between centromeres and replication origins, we exploited C. albicans strains in which the native chromosome 5 centromere had been deleted and a heritable neocentromere had formed at a different novel locus on the chromosome in each strain [7]. These strains provide a unique opportunity to address the order of this dependency. In four neocentromere strains, the centromere shifted to non-telomeric loci that were not ORC-binding sites in the wild-type progenitor strain (Figure 2A and 2C). This indicates that neocentromeres do not form at pre-existing replication origins.
Fig. 2. Neocentromere formation causes replication timing advancement. 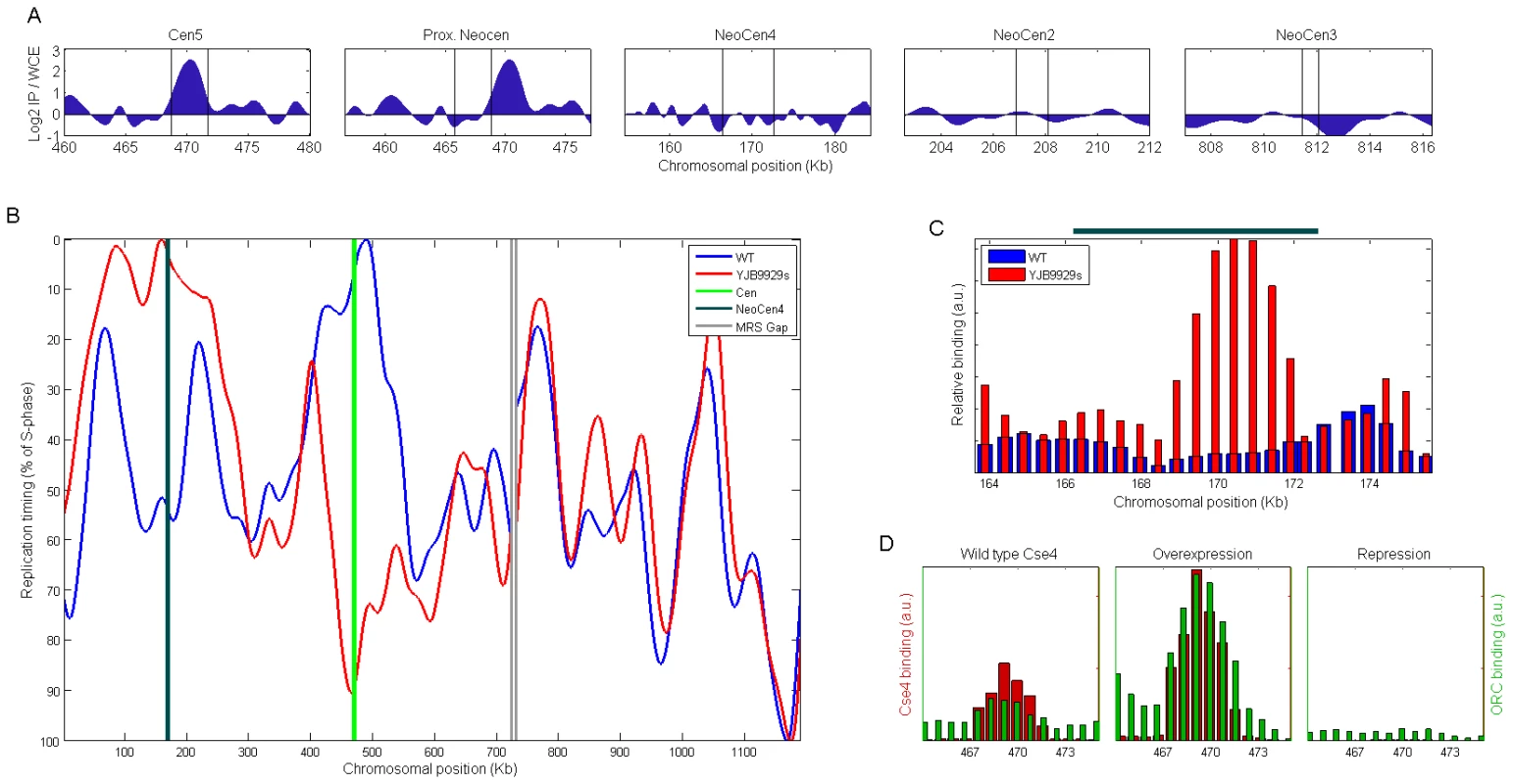
(A) ORC binding (determined by ChIP-chip) in wild-type cells at CEN5 and at chromosome 5 loci with the potential to form a neocentromere [7]. ORC clearly binds at CEN5 and is not evident within the loci that have been shown to form neocentromeres following deletion of CEN5. Regions identified as centromeric or capable of forming neocentromeres (by virtue of Cse4 binding in the corresponding cen5Δ strains) are located between the two vertical lines. (B) The replication profile of chromosome 5 in wild-type and in strain YJB9929s, in which CEN5 was deleted and a neocentromere formed on the left chromosome arm [7]. MRS: Major Repeat Sequence. (C) ChIP reveals an ORC binding site within the neocentromere region (dark green line above the plot; [7]) in strain YJB9929s that is not present in the wild-type strain. We note that ORC binding provides a more precise indication of the actual location of an origin than the replication timing profiles; thus, the offset of the replication peak in (B) from the neocentromere site is likely explained by experimental noise. (D) Overexpression or repression of CSE4 (Materials and Methods) is accompanied by consistent changes in ORC binding strength at CEN5. We then assayed replication timing in a strain that acquired a stable homozygous neocentromere following deletion of CEN5 DNA and homozygosis of the entire chromosome (neoCEN4, strain YJB9929s; see Materials and Methods; [7]). Origin activity at the CEN5 region was completely lost, indicating that origin determinants had resided within the deleted centromeric DNA (Figure 2B). Strikingly, neocentromere formation was associated with the appearance, in the neoCEN4 region, of a new replication origin that was the first to replicate in the chromosome (Figure 2B). Consistent with the idea that a de novo origin formed at neoCEN4, a new ORC binding site in the neoCEN4 region was readily evident by ChIP-PCR only in the strain that had formed a neoCEN and not in its wild-type parental strain (Figure 2C). Of note, the replication timing of origins flanking the centromere and neocentromere was also altered, possibly reflecting a broad position effect exerted by centromeres on the replication timing of flanking origins of replication. This resembles the position effect that mediates generally earlier replication timing within tens of kilobases surrounding conventional centromeres (Figure 1I). The profiles of chromosomes 2 and 3, which would not be expected to be altered by deletion of CEN5, were indeed similar between the neocentromeric and wild type strains (correlation between the profiles r = 0.92−0.95 versus 0.36 for chromosome 5).
To further test the hypothesis that centromeric activity can recruit replication determinants, we used a conditional promoter to manipulate the level of Cse4 expression and assayed the effect of Cse4 levels on the level of ORC-binding at centromeres. Consistent with the idea that centromeres recruit origins of replication, we found that over-expression of Cse4 resulted in increased levels of ORC at CEN5, while repression of Cse4 expression resulted in reduced levels of ORC (Figure 2D). Taken together, we conclude that centromeric determinants confer both the presence of replication origins and a distinct, early replication timing to the loci at which they reside.
Sequence asymmetry at centromeric DNA suggests constitutive origin activity correlated with epigenetic inheritance of centromere positions
As in higher eukaryotes, C. albicans has regional centromeres with different primary sequences at each chromosome [18], [20]. Furthermore, centromeric DNA sequences are highly divergent between strains and related species, mutating at rates higher than those at intergenic regions and synonymous sites [21]. Despite this lack of homology, sequence analysis revealed a common feature among C. albicans centromeres: all of them have a sequence pattern that has been detected previously at replication origin sites of hundreds of bacterial, archeal, viral and organellar genomes that have a single replication origin in their genome [24]. This pattern, indicative of constitutive replication origin activity, is an asymmetric GC skew, in which G nucleotides are more abundant than C nucleotides on one side of the centromeric replication origins, and G nucleotides are less abundant than C nucleotides on the same strand on the other side of the centromere (Figure 3A, 3B, 3D). For A versus T nucleotides, a similar skew pattern was observed, in the opposite direction relative to the GC skew, with a lower magnitude, and with essentially the same position of switch in skew direction. A similar AT skew pattern is present in many of the species with single-origin genomes.
Fig. 3. Nucleotide skew patterns and their correlation to syntenic conservation of centromere position. 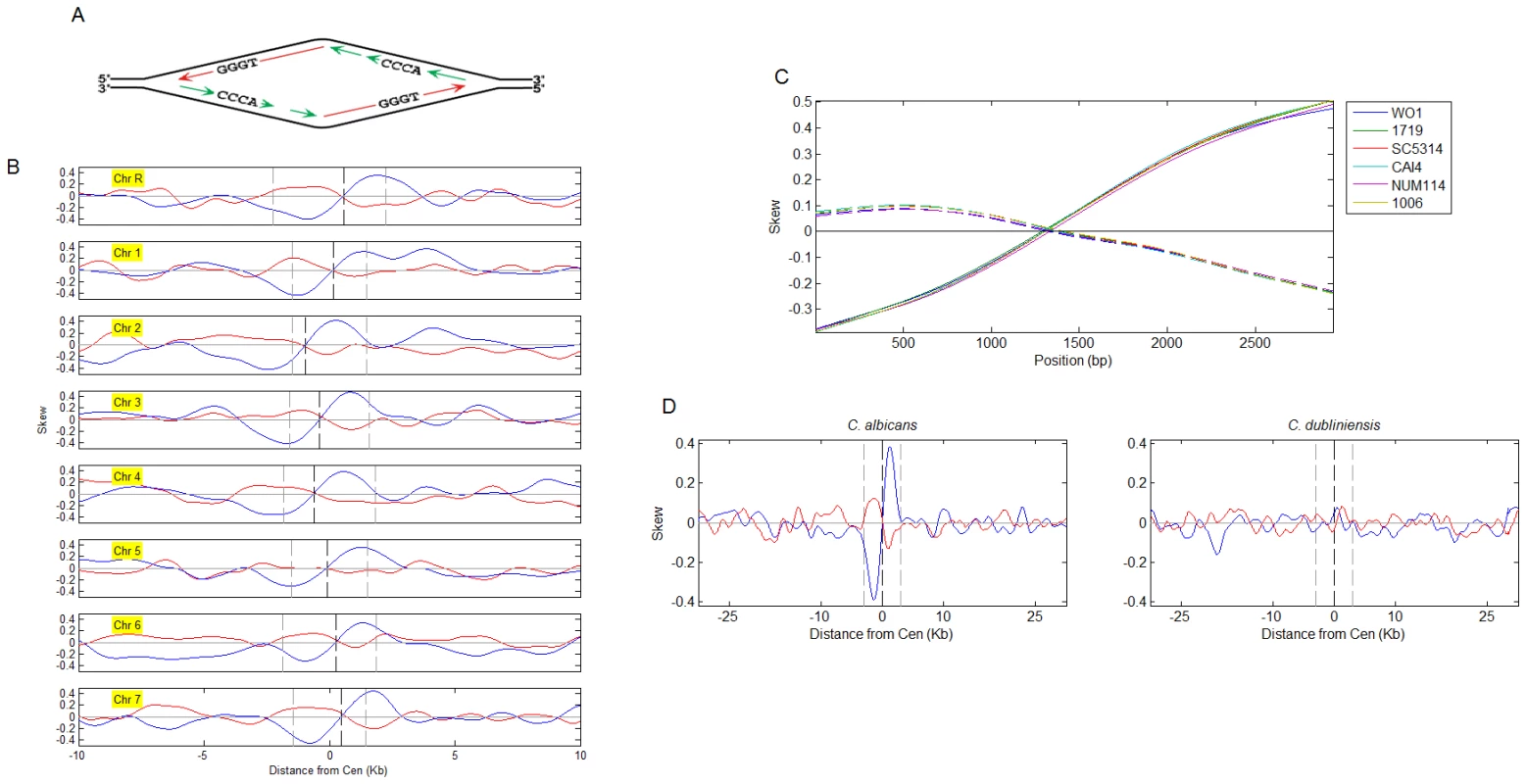
(A) A diagram of GC skew formation. G, and to a lesser extent, T nucleotides, are enriched on the leading strand. The location of the replication origin is the point of replication strand switch as well as the point of GC and AT skew orientation reversal. (B) GC (blue) and AT (red) skew at the C. albicans centromere regions. The opposite orientation of the GC and AT skews depends on the way the skews are defined (i.e. the same analysis with relation to ‘TA’ instead of ‘AT’ skews would result in the same orientation). Skew curves represent the degree to which base-pairs are oriented with a particular base on one (value of 1) or another (−1) of the strands (0- no skew). Centromeres are aligned at the center of the published centromere borders (dashed grey lines). Dashed black lines: GC skew zero-intersection points. GC-skewed regions also exhibited high GC content (Figures S4, S5, S6, S7); GC content at the centromeres exhibited a local drop within a region of increased GC content; nucleosome occupancy appeared to follow a similar pattern (Figure S6). (C) GC skew (solid lines) and AT skew (dashed lines) for CEN1 of the C. albicans strains indicated. Based on sequence data for a DNA fragment containing CEN1 from Ref. [20] (x-axis is position on the sequenced fragment). For strain WO-1, where the complete genome sequence is available, skew patterns were conserved at all centromeres (data not shown). (D) Average GC and AT skew patterns for C. albicans (based on individual chromosomes shown in (A) and C. dubliniensis (individual chromsomes are shown in Figure S9). Chromosomes were aligned at the GC skew zero-intersection points (dashed black line) and skew signals were averaged. Dashed grey lines delimit 3 kb regions to each side. Such skew patterns are inferred to be a consequence of mutations that accumulate in a strand-specific manner: specific substitutions occur at different rates on the leading and lagging strands and the leading and lagging strands switch identities at the point of replication initiation (Figure 3A). Thus, a nucleotide skew pattern implies the presence of a replication origin at the point of symmetry switch [24]. This is consistent with our results detecting the presence of replication origins within centromeric regions. In particular, strong ORC-binding sites reside in very close proximity to the points of skew sign switch (Figure 1E and Figure S2)).
Moreover, skew patterns accumulate over evolutionary time scales, only in cases where a replication origin is consistently active for many generations; hence skews are evident in most genomes with single origins. Conversely, and despite the observation that replication fork asymmetry also causes a strand bias in mutagenesis experiments [25] in S. cerevisiae, skew signals are rarely detected in eukaryotic genomes, presumably because eukaryotic replication origins are not constitutively active at specific loci over long time periods.
Thus, the identification of asymmetrical skew patterns at centromeric replication origins of C. albicans suggests that these origins have been active in most, or all, cell cycles for many generations. Indeed, skew patterns are conserved among C. albicans strains that represent divergence times of 1-3 million years (Figure 3C; [20]). Furthermore, skew patterns are virtually identical at all centromeres in C. albicans and C. dubliniensis (Figure 3D and Figure S9), which diverged from each other ∼20 million years ago and share complete conservation of centromere synteny, yet no conservation of centromere primary DNA sequence [21]. The conservation of skew patterns at centromeres in both organisms suggests that DNA replication activity is intimately associated with the mechanism that has ensured the epigenetic inheritance of centromere synteny.
In scanning the entire C. albicans genome sequence, we found that the strongest skew patterns were at centromeres, but that skew patterns were also identifiable at many telomeres (Figures S4, S5, S6, S7). No skew patterns different than background were seen in any of the loci where neocentromeres have been observed to form (data not shown), indicating that skewed DNA is not an attractor for centromeric proteins. Rather, the lack of skew patterns at neocentromeric loci is consistent with the idea that skews only arise following constitutive origin activity at centromeres over evolutionary time scales.
Evolutionary conservation of the centromere-early origin association
A DNA sequence pattern characterizing epigenetically-inherited centromeres provides a potential tool for predicting centromere locations in other species. Indeed, we found one distinctive skew pattern per chromosome, providing a prediction of centromere (and origin) locations, in two additional members of the Candida clade, L. elongisporus and C. lusitiniae, as well as in the more distantly related yeast, Yarrowia lipolytica (Figures S8, S9; Table S1). In Y. lipolytica, our approach re-identified the five known centromere locations (and predicts the centromere location of the sixth chromosome) in this species. Remarkably, Y. lipolytica centromeres were identified by searching for chromosomal sequences that function as DNA replication origins and subsequent work showed that, for Y. lipolytica plasmids, replication origin and centromere activity are inter-dependent [26], [27].
S. pombe centromeres also include several replication origins that fire very early in S-phase despite being embedded within heterochromatin [28]. We re-analyzed S. pombe replication timing microarray data (Materials and Methods), and found that CEN1, the only centromere that could be probed at sufficient resolution, is the first locus to replicate on chromosome 1 (Figure 4). Notably, no skew patterns were observed at S. pombe centromeric regions (data not shown). This is likely due to the presence of more than one origin within these regions. We suggest that centromeric skew patterns may be unique to species with smaller regional centromeres. In contrast, in a clade containing the Saccharomyces species (see Figure S9A), genetic, point-centromere positions have become fixed in DNA sequence [29] and do not coincide directly with replication origins. Nonetheless, generally early DNA replication surrounding centromeres (Figure 1H and 1I) could be an evolutionary remnant of replication-timing-dependent, epigenetically-inherited centromeres. Taken together, the association between centromeres and replication origins is conserved at least across a wide range of yeast phylogeny (Figure S9A). Since neither DNA sequence nor sufficient replication timing data are available for centromeric regions of higher model organisms, it remains to be determined whether this association is common among other eukaryotic species.
Fig. 4. Replication timing profile of S. pombe chromosome 1. 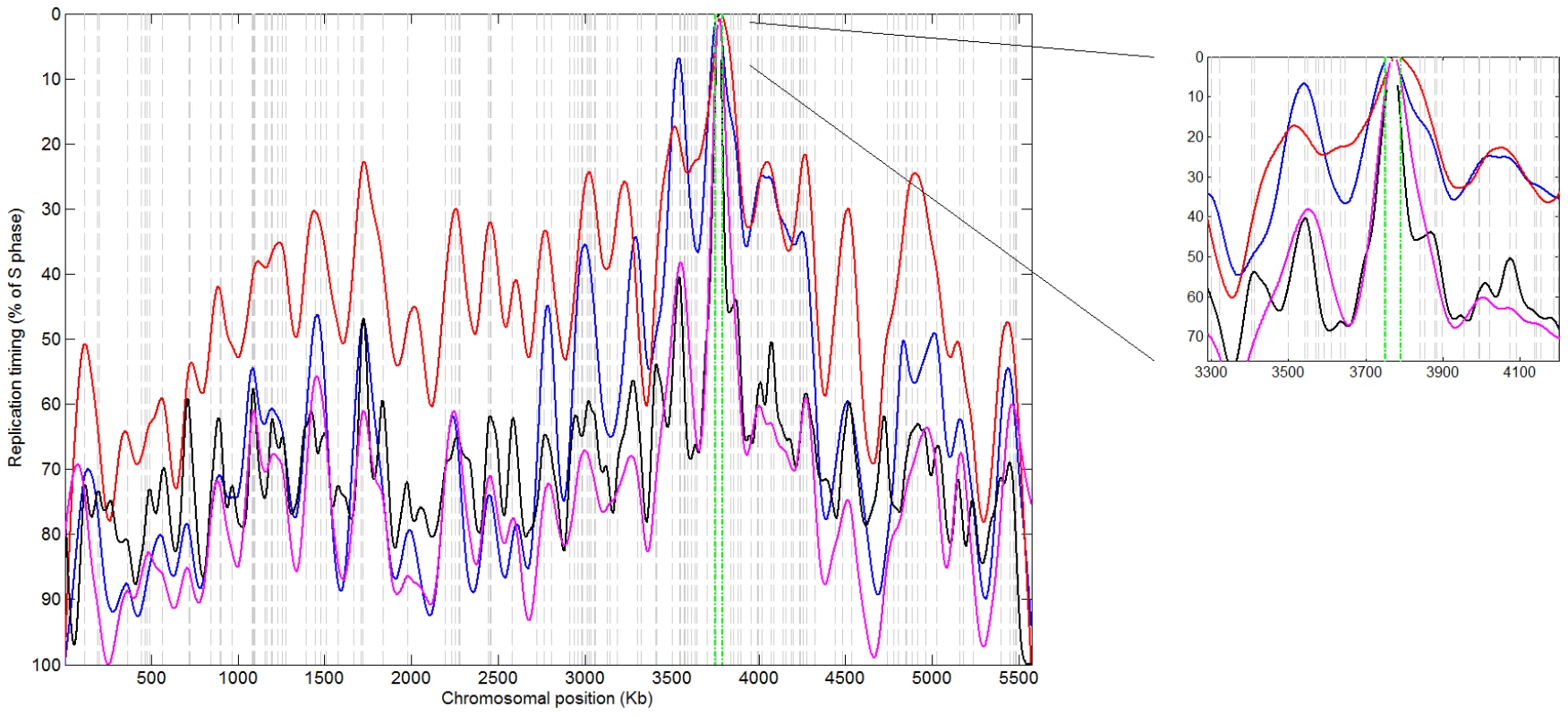
Replication timing microarray data for chromosome 1 from [35] (blue), [34] (red- time course experiment; black- HU experiment), and [36] (magenta) show early replication at CEN1. Green lines: centromere location; dashed grey lines: ORC binding sites that also incorporated BrdU [38]. Discussion
This study provides the first comprehensive analysis of DNA replication of epigenetically-inherited centromeres. Our results show that centromeres replicate at a distinct time from the rest of the chromosome, centromeric determinants can change the replication time of the loci at which they reside, and the DNA replication properties of centromeres are linked to their epigenetic inheritance over evolutionary time scales. Based on this, we propose a self-reinforcing, positive-feedback loop model, in which centromeric determinants affect DNA replication timing and in turn, a distinct replication time facilitates the recruitment of centromeric determinants to that specific locus (Figure 5). Specifically, centromere site specification by Cse4 nucleosomes recruits factors that mediate replication initiation at a distinct time at the very beginning of S-phase (Figure 2). Furthermore, we speculate that early, distinctive replication timing, in turn enables subsequent deposition of centromere-specific proteins such as Cse4 nucleosomes at the region that is first to replicate, for instance due to elevated levels of these proteins in the very beginning of S-phase [11]–[14]. In particular, it has previously been shown in S. pombe that expression of Cse4 occurs from late-M to G1/S phase, and precedes maximal expression of histone H3 in S-phase [11]. Our calculations suggest that centromeric chromatin is replicated very rapidly - within the first <20 seconds of S-phase in C. albicans (Materials and Methods). Further studies will be required to link the distinct timing of CENP-A deposition and the time of centromere replication.
Fig. 5. A model of DNA replication-timing–dependent epigenetic centromere inheritance. 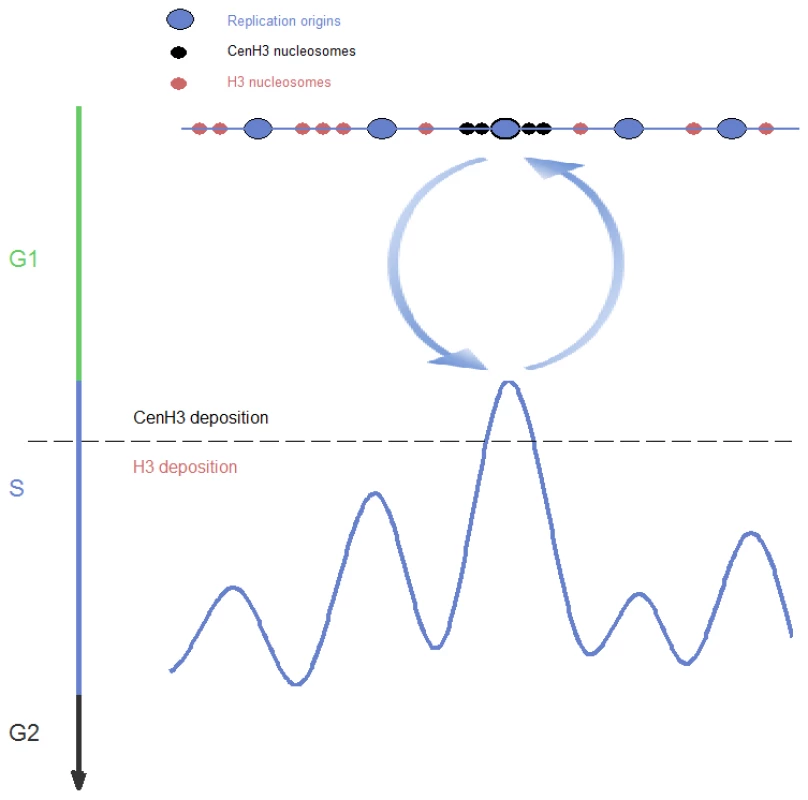
Two processes contribute to a self-reinforcing feedback loop: replication origin sites embedded within centromeric chromatin (upper part) are activated earlier than other origins (lower part); and assembly of centromeric chromatin occurs in very early S-phase, while canonical chromatin is replicated later. This self-reinforcing association between centromere determinants and early replication timing does not require a particular underlying DNA sequence, establishing it as an epigenetic system. This epigenetic system determines centromere position, as well as replication timing and the constitutive nature of centromeric replication origins, which our results show for the first time can be determined epigenetically. Early and constitutive replication are directly related to each other, as a locus that replicates first will never be passively replicated and hence will be active in every cell cycle. The combination of constitutive origin activity and the relaxation of sequence constraints that stems from the epigenetic nature of this system, enables the underlying DNA to mutate at elevated rates, explaining previous observations that centromeric DNA mutates at high rates while centromere position remains unaltered [21], [30]. We now show that centromeric DNA in several yeast species mutates with a specific sequence bias due to the influence of a centromere-associated replication origin.
We describe a novel type of epigenetic inheritance mechanism that is directly related to the fundamental mechanism of genetic inheritance in that both depend on DNA replication. Centromeres are maintained at specific chromosomal positions in a highly robust manner that is crucial for ensuring high-fidelity chromosome segregation; thus, this replication-timing-dependent epigenetic inheritance mechanism may confer a level of stability unprecedented for an epigenetic system. Finally, another attractive aspect of this model is that, instead of requiring a specialized mechanism for inheritance of centromeric nucleosomes outside of S-phase [11], [17], it directly connects centromeric nucleosome inheritance with DNA replication, similar to the inheritance of canonical nucleosomes that occurs following passage of the replication fork.
Materials and Methods
Strains and media
Strains SC5314 (wild type; diploid) and YJB9929s [7] (homozygosed for chromosome 5 subsequent to neocentromere formation) were grown in YPAD media at 30°C. Strain CAKS3b in which Cse4 expression is under the control of the glucose-repressible PCK1 promoter [31] was grown in YPA-succinate to induce Cse4 overexpression and diluted into fresh YPA-succinate or into YPAD to repress expression and grown for 6 hours at 30°C before harvesting cells.
Replication timing
FACS and replication timing microarray experiments and analysis were as previously described [22]. Agilent arrays (2×105 format) were custom designed with probes spaced every 140 bp, on average, across the entire genome (assembly21). The experiment was repeated four times with correlation and autocorrelation values comparable to those previously obtained [22] (data not shown), validating the high quality of the data. Strain YJB9929s was repeated twice with a microarray that included the same probes for chromosomes 2, 3 and 5 only. Microarray data have been submitted to GEO (http://www.ncbi.nlm.nih.gov/geo/) with accession number GSE17963.
The experimental repeats were weighted-averaged using Tukey's biweight method [22] and the averaged data was smoothed using a smoothing spline (as implicated in the Matlab function Csaps), which optimizes the degree of curvature versus deviance from the data, with the parameter chosen determining the weight of these two criteria. The parameters chosen for the different chromosomes were as follows (numbers are the -log10 of the parameters provided to Csaps): ChrR: 16; Chr1,2 : 15.75; Chr3 : 15.25; Chr4; 15; Chr5,6,7 : 14.75 (generally a function of chromosome length). This choice of parameters maximized the similarity to the S. cerevisiae data with respect to the significance of frequency components retained (data not shown), enabling valid comparison of the number of replication origins between S. cerevisiae and C. albicans (Figure S3).
The rDNA and chromosome 5 Major Repeat Sequence (MRS; which is larger than 50 Kb on chromosome 5) loci were treated as data gaps; the corresponding chromosomes were smoothed separately on each side of these gaps (with the same parameter). Scaling the entire genome, rather than individual chromosomes, to 0–100, had little if any effect on the data.
Origin analysis
2D gels were performed as previously described [32]. Chromatin immunoprecipitation was as previously described [7] with polyclonal antibodies against the entire ORC complex [33] kindly provided by Stephen P. Bell. PCR was performed with 33 primer pairs interrogating the CEN5 region or 24 primer pairs for the neocentromere region; data was averaged over two experimental repetitions and three consecutive primer pairs. RT-PCR was performed in duplicates using the LightCycler 480 system according to the manufacturer's instructions. ChIP-chip was performed in nine biological repeats according to Agilent protocols and will be described in detail elsewhere together with a complete list of replication origin sites (A.K., H-J.T, L.B. and J.B, manuscript in preparation). Results remained unaltered when normalizing to probe GC content.
Analysis of S. pombe replication microarray data
Datasets used were: a 65, 75, 85, 95 minutes S-phase time course experiment [34]; a replication timing in HU experiment performed in three repeats[34]; a single experiment using ssDNA mapping [35]; and a two time point (2 h, 4 h) S-phase time course experiment [36]. Chromosomes 2 and 3 had probe gaps of ∼50–80 Kb in the centromere region and thus were not amenable to centromere replication timing analysis. Data from each dataset was weighted-averaged over three consecutive probes and all the time points/repetitions of that experiment and smoothed using spline parameters (as above) of 17 for all experiments and 16 for the HU experiment (use of a parameter of 17 did not alter the result for the centromere). This data analysis approach effectively removes outlier data points that prevented the previous identification of CEN1 as the first locus to replicate on chromosome 1.
Time to replicate centromeric DNA
To avoid influences of the data smoothing on replication fork progression near origins [22], we linearly interpolated the replication profiles from peaks to valleys. Replication timing was converted to minutes by multiplying replication time in percent by the total duration of S-phase in minutes (Figure 1). The latest replication timing of the loci 3 Kb from each side of the centromere-proximal origin on each chromosome (excluding chromosome 6 as indicated above) was the time to replicate each centromere.
Sequences and gene annotations
Genome sequences were obtained from the following sources: Candida albicans (assembly 21): from CGD (Candida Genome Database; http://www.candidagenome.org/); Saccharomyces cerevisiae: from SGD (Saccharomyces Genome Database; http://www.yeastgenome.org/); Candida dubliniensis, Schizosaccharomyces pombe: from The Wellcome Trust Sanger Institute (http://www.sanger.ac.uk/sequencing/Candida/dubliniensis/), with gene annotation from GeneDB (http://www.genedb.org/genedb/); Candida albicans strain WO-1, Candida guilliermondii, Candida lusitaniae, Candida parapsilosis, Candida tropicalis, Debaryomyces hansenii, Lodderomyces elongisporus: from the Broad Institute (http://www.broadinstitute.org/annotation/genome/candida_group/MultiHomehtml); Pichia stipitis: from JGI (http://genomejgi-psf.org/Picst3/Picst3homehtml); Yarrowia lipolytica: from Genolevures (http://www.genolevures.org/indexhtml) (with centromere positions from [37]).
Sequence skew analysis
GC skew was calculated as (G−C)/(G+C), AT skew as (A−T)/(A+T), and each was smoothed using a smoothing spline with parameters equivalent to smoothing by sliding windows of ∼1.5 Kb. For identifying strong skew patterns in the genome (see Figure S5), we categorized intergenic regions by the combination of length, GC content, and either of several parameters of: GC skew, AT skew, GC content, proximity of the latter to each other (of zero-intersection points for skews and extrema for GC content), and combinations thereof. We then searched for a discrete cluster in any of these parameters.
Supporting Information
Zdroje
1. PanchenkoT
BlackBE
2009 The epigenetic basis for centromere identity. Prog Mol Subcell Biol 48 1 32
2. MorrisCA
MoazedD
2007 Centromere Assembly and Propagation. Cell 128 647 650
3. EkwallK
2007 Epigenetic control of centromere behavior. Annu Rev Genet 41 63 81
4. AllshireRC
KarpenGH
2008 Epigenetic regulation of centromeric chromatin: old dogs, new tricks? Nature Reviews Genetics 9 923 937
5. MalikHS
HenikoffS
2009 Major evolutionary transitions in centromere complexity. Cell 138 1067 1082
6. SullivanBA
BlowerMD
KarpenGH
2001 Determining centromere identity: cyclical stories and forking paths. Nat Rev Genet 2 584 596
7. KetelC
WangHS
McClellanM
BouchonvilleK
SelmeckiA
2009 Neocentromeres form efficiently at multiple possible loci in Candida albicans. PLoS Genet 5 e1000400 doi:10.1371/journal.pgen.1000400
8. MarshallOJ
ChuehAC
WongLH
ChooKH
2008 Neocentromeres: new insights into centromere structure, disease development, and karyotype evolution. Am J Hum Genet 82 261 282
9. ShelbyRD
MonierK
SullivanKF
2000 Chromatin Assembly at Kinetochores Is Uncoupled from DNA Replication. J Cell Biol 151 1113 1118
10. HenikoffS
AhmadK
PlateroJS
van SteenselB
2000 Heterochromatic deposition of centromeric histone H3-like proteins. Proceedings of the National Academy of Sciences USA 97 716 721
11. TakahashiK
ChenES
YanagidaM
2000 Requirement of Mis6 Centromere Connector for Localizing a CENP-A-Like Protein in Fission Yeast. Science 288 2215 2219
12. TakayamaY
SatoH
SaitohS
OgiyamaY
MasudaF
2008 Biphasic Incorporation of Centromeric Histone CENP-A in Fission Yeast. Mol Biol Cell 19 682 690
13. DuPrawEJ
1968 Cell and Molecular Biology. Academic Press
14. CsinkAK
HenikoffS
1998 Something from nothing: the evolution and utility of satellite repeats. Trends in Genetics 14 200 204
15. AhmadK
HenikoffS
2001 Centromeres Are Specialized Replication Domains in Heterochromatin. J Cell Biol 153 101 110
16. SullivanB
KarpenG
2001 Centromere identity in Drosophila is not determined in vivo by replication timing. J Cell Biol 154 683 690
17. Weidtkamp-PetersS
RahnHP
CardosoMC
HemmerichP
2006 Replication of centromeric heterochromatin in mouse fibroblasts takes place in early, middle, and late S phase. Histochem Cell Biol 125 91 102
18. SanyalK
BaumM
CarbonJ
2004 Centromeric DNA sequences in the pathogenic yeast Candida albicans are all different and unique. Proceedings of the National Academy of Sciences USA 101 11374 11379
19. BaumM
SanyalK
MishraPK
ThalerN
CarbonJ
2006 Formation of functional centromeric chromatin is specified epigenetically in Candida albicans. Proceedings of the National Academy of Sciences USA 103 14877 14882
20. MishraPK
BaumM
CarbonJ
2007 Centromere size and position in Candida albicans are evolutionarily conserved independent of DNA sequence heterogeneity. Mol Genet Genomics 278 455 465
21. PadmanabhanS
ThakurJ
SiddharthanR
SanyalK
2008 Rapid evolution of Cse4p-rich centromeric DNA sequences in closely related pathogenic yeasts, Candida albicans and Candida dubliniensis. Proceedings of the National Academy of Sciences USA 105 19797 19802
22. KorenA
SoiferI
BarkaiN
2010 MRC1-dependent scaling of the budding yeast DNA replication timing program. Genome Res 20 781 790
23. RaghuramanMK
WinzelerEA
CollingwoodD
HuntS
WodickaL
2001 Replication Dynamics of the Yeast Genome. Science 294 115 121
24. SernovaNV
GelfandMS
2008 Identification of replication origins in prokaryotic genomes. Brief Bioinform 9 376 391
25. PavlovYI
NewlonCS
KunkelTA
2002 Yeast origins establish a strand bias for replicational mutagenesis. Mol Cell 10 207 213
26. VernisL
PoljakL
ChaslesM
UchidaK
CasaregolaS
2001 Only centromeres can supply the partition system required for ARS function in the yeast Yarrowia lipolytica. J Mol Biol 305 203 217
27. FournierP
AbbasA
ChaslesM
KudlaB
OgrydziakDM
1993 Colocalization of centromeric and replicative functions on autonomously replicating sequences isolated from the yeast Yarrowia lipolytica. Proceedings of the National Academy of Sciences USA 90 4912 4916
28. KimS-M
DubeyDD
HubermanJA
2003 Early-replicating heterochromatin. Genes & Development 17 330 335
29. MeraldiP
McAinshA
RheinbayE
SorgerP
2006 Phylogenetic and structural analysis of centromeric DNA and kinetochore proteins. Genome Biology 7 R23
30. HenikoffS
AhmadK
MalikHS
2001 The centromere paradox: stable inheritance with rapidly evolving DNA. Science 293 1098 1102
31. SanyalK
CarbonJ
2002 The CENP-A homolog CaCse4p in the pathogenic yeast Candida albicans is a centromere protein essential for chromosome transmission. Proc Natl Acad Sci U S A 99 12969 12974
32. BrewerBJ
FangmanWL
1987 The localization of replication origins on ARS plasmids in S. cerevisiae. Cell 51 463 471
33. WyrickJJ
AparicioJG
ChenT
BarnettJD
JenningsEG
2001 Genome-wide distribution of ORC and MCM proteins in S. cerevisiae: high-resolution mapping of replication origins. Science 294 2357 2360
34. HeichingerC
PenkettCJ
BahlerJ
NurseP
2006 Genome-wide characterization of fission yeast DNA replication origins. EMBO J 25 5171 5179
35. FengW
CollingwoodD
BoeckME
FoxLA
AlvinoGM
2006 Genomic mapping of single-stranded DNA in hydroxyurea-challenged yeasts identifies origins of replication. Nature Cell Biology 8 148 155
36. MickleK
RamanathanS
RosebrockA
OlivaA
ChaudariA
2007 Checkpoint independence of most DNA replication origins in fission yeast. BMC Molecular Biology 8 112
37. DujonB
ShermanD
FischerG
DurrensP
CasaregolaS
2004 Genome evolution in yeasts. Nature 430 35 44
38. HayashiM
KatouY
ItohT
TazumiM
YamadaY
2007 Genome-wide localization of pre-RC sites and identification of replication origins in fission yeast. EMBO J 26 1327 1339
Štítky
Genetika Reprodukční medicína
Článek vyšel v časopisePLOS Genetics
Nejčtenější tento týden
2010 Číslo 8
-
Všechny články tohoto čísla
- A Model for Damage Load and Its Implications for the Evolution of Bacterial Aging
- Mutation in the Gene Encoding Ubiquitin Ligase LRSAM1 in Patients with Charcot-Marie-Tooth Disease
- Identification of the Bovine Arachnomelia Mutation by Massively Parallel Sequencing Implicates Sulfite Oxidase (SUOX) in Bone Development
- Did Genetic Drift Drive Increases in Genome Complexity?
- The 5p15.33 Locus Is Associated with Risk of Lung Adenocarcinoma in Never-Smoking Females in Asia
- An Alpha-Catulin Homologue Controls Neuromuscular Function through Localization of the Dystrophin Complex and BK Channels in
- Epigenetically-Inherited Centromere and Neocentromere DNA Replicates Earliest in S-Phase
- Survival and Growth of Yeast without Telomere Capping by Cdc13 in the Absence of Sgs1, Exo1, and Rad9
- Tuberous Sclerosis Complex 1 Regulates dE2F1 Expression during Development and Cooperates with RBF1 to Control Proliferation and Survival
- Disease-Associated Mutations That Alter the RNA Structural Ensemble
- The Transcriptomes of Two Heritable Cell Types Illuminate the Circuit Governing Their Differentiation
- Inactivation of VCP/ter94 Suppresses Retinal Pathology Caused by Misfolded Rhodopsin in
- Multiple Independent Loci at Chromosome 15q25.1 Affect Smoking Quantity: a Meta-Analysis and Comparison with Lung Cancer and COPD
- Transcriptional Regulation by CHIP/LDB Complexes
- Conserved Role of in Ethanol Responses in Mutant Mice
- A Global Overview of the Genetic and Functional Diversity in the Pathogenicity Island
- Common Inherited Variation in Mitochondrial Genes Is Not Enriched for Associations with Type 2 Diabetes or Related Glycemic Traits
- Extracellular Dopamine Potentiates Mn-Induced Oxidative Stress, Lifespan Reduction, and Dopaminergic Neurodegeneration in a BLI-3–Dependent Manner in
- Genetic Analysis of Baker's Yeast Msh4-Msh5 Reveals a Threshold Crossover Level for Meiotic Viability
- Genome-Wide Association Studies of Serum Magnesium, Potassium, and Sodium Concentrations Identify Six Loci Influencing Serum Magnesium Levels
- Something New: An Interview with Radoje Drmanac
- The Extinction Dynamics of Bacterial Pseudogenes
- Microtubule Actin Crosslinking Factor 1 Regulates the Balbiani Body and Animal-Vegetal Polarity of the Zebrafish Oocyte
- Consistent Association of Type 2 Diabetes Risk Variants Found in Europeans in Diverse Racial and Ethnic Groups
- Transmission of Mitochondrial DNA Diseases and Ways to Prevent Them
- Telomere Disruption Results in Non-Random Formation of Dicentric Chromosomes Involving Acrocentric Human Chromosomes
- Chromosome Axis Defects Induce a Checkpoint-Mediated Delay and Interchromosomal Effect on Crossing Over during Drosophila Meiosis
- Dynamic Chromatin Organization during Foregut Development Mediated by the Organ Selector Gene PHA-4/FoxA
- Ancient Protostome Origin of Chemosensory Ionotropic Glutamate Receptors and the Evolution of Insect Taste and Olfaction
- A Wnt-Frz/Ror-Dsh Pathway Regulates Neurite Outgrowth in
- PLOS Genetics
- Archiv čísel
- Aktuální číslo
- Informace o časopisu
Nejčtenější v tomto čísle- Identification of the Bovine Arachnomelia Mutation by Massively Parallel Sequencing Implicates Sulfite Oxidase (SUOX) in Bone Development
- Common Inherited Variation in Mitochondrial Genes Is Not Enriched for Associations with Type 2 Diabetes or Related Glycemic Traits
- A Model for Damage Load and Its Implications for the Evolution of Bacterial Aging
- Did Genetic Drift Drive Increases in Genome Complexity?
Kurzy
Zvyšte si kvalifikaci online z pohodlí domova
Současné možnosti léčby obezity
nový kurzAutoři: MUDr. Martin Hrubý
Všechny kurzyPřihlášení#ADS_BOTTOM_SCRIPTS#Zapomenuté hesloZadejte e-mailovou adresu, se kterou jste vytvářel(a) účet, budou Vám na ni zaslány informace k nastavení nového hesla.
- Vzdělávání



