-
Články
Top novinky
Reklama- Vzdělávání
- Časopisy
Top články
Nové číslo
- Témata
Top novinky
Reklama- Videa
- Podcasty
Nové podcasty
Reklama- Kariéra
Doporučené pozice
Reklama- Praxe
Top novinky
ReklamaGenetical Genomics: Spotlight on QTL Hotspots
article has not abstract
Published in the journal: . PLoS Genet 4(10): e32767. doi:10.1371/journal.pgen.1000232
Category: Perspective
doi: https://doi.org/10.1371/journal.pgen.1000232Summary
article has not abstract
Genetical genomics aims at identifying quantitative trait loci (QTLs) for molecular traits such as gene expression or protein levels (eQTL and pQTL, respectively). One of the central concepts in genetical genomics is the existence of hotspots [1], where a single polymorphism leads to widespread downstream changes in the expression of distant genes, which are all mapping to the same genomic locus. Several groups have hypothesized that many genetic polymorphisms—e.g., in major regulators or transcription factors—would lead to large and consistent biological effects that would be visible as eQTL hotspots.
Rather surprisingly, however, there have been only very few verified hotspots in published genetical genomics studies to date. In contrast to local eQTLs, which coincide with the position of the gene and are presumably acting in cis—e.g., by polymorphisms in the promoter region—distant eQTLs have been found to be more elusive. They seem to show smaller effect sizes and are less consistent, perhaps due to the indirect regulation mechanism, resulting in lower statistical power to detect them and, consequently, an inability to reliably delimit hotspots [2]. While there are typically hundreds to thousands of strong local eQTLs per study, the number of associated hotspots is much lower. For example, a recent very large association study in about 1,000 humans did not find a single significant hotspot [3]. Other studies have reported up to about 30 hotspots, far less than the number of significant local eQTLs (Table 1). The molecular basis is known for less than a handful of cases. An example is the Arabidopsis ERECTA locus, which leads to a drastic phenotypic change in the plant and has broad pleiotropic effects on many molecular (and morphological) traits [4].
Tab. 1. eQTL Hotspots Reported in Selected Genetical Genomics Studies. 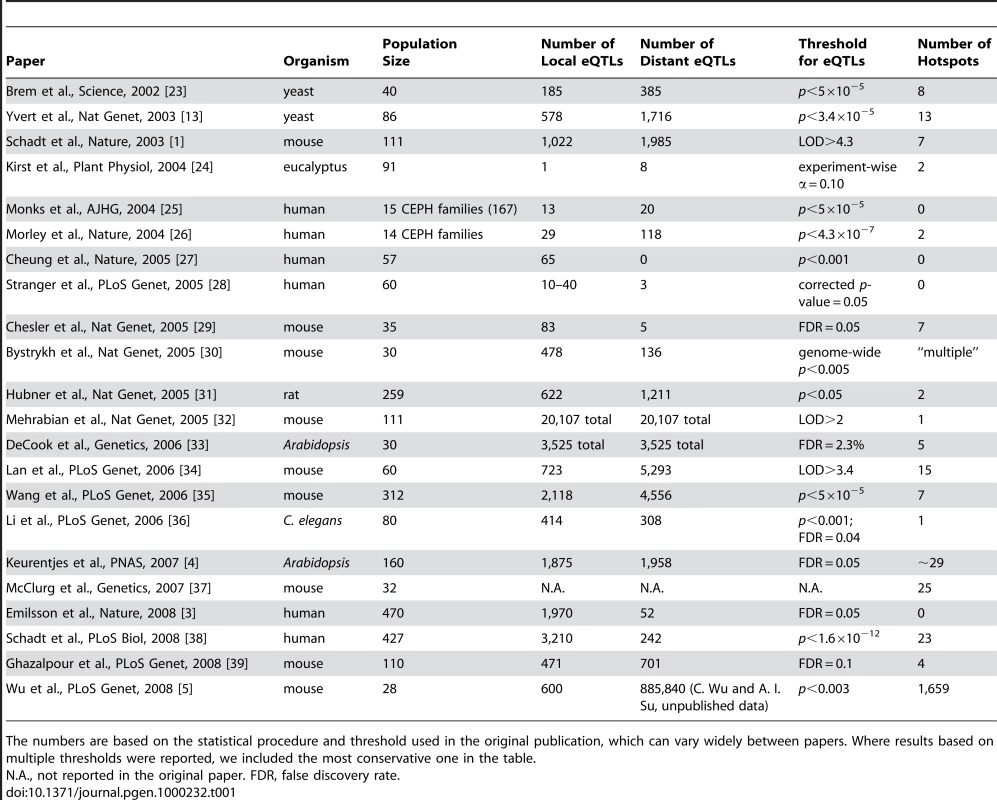
The numbers are based on the statistical procedure and threshold used in the original publication, which can vary widely between papers. Where results based on multiple thresholds were reported, we included the most conservative one in the table. Recently, Wu et al. [5] reported the large-scale identification of hotspots. They studied gene expression in adipose tissue of 28 inbred mouse strains and performed eQTL analysis by genome-wide association analysis. The paper reports the identification of over 1,600 candidate hotspots, each with a minimum hotspot size of 50 target genes. Furthermore, they demonstrated that these hotspots are biologically coherent by showing that in about 25% of cases, the hotspot targets are enriched for functional gene sets derived from Gene Ontology, the KEGG pathways database, and the Ingenuity Pathways Knowledge Base. These findings suggested that genetic polymorphisms can indeed lead to large and consistent biological effects that are visible as eQTL hotspots.
However, the authors chose a relatively permissive threshold of p = 0.003 for QTL detection, uncorrected for multiple testing. In total, 886,440 eQTLs were identified at this threshold, i.e., 134 per gene. A permutation test (C. Wu and A. I. Su, unpublished data) shows that this results in a false discovery rate of 64%, largely resulting from multiple testing across 157,000 SNPs and 6,601 probe sets. This relatively permissive threshold was chosen because the focus of the analysis was on patterns of eQTL hotspots and not on individual eQTL associations. Analysis of eQTL patterns is relatively robust to individual false positives, and a permissive threshold allows for relatively greater sensitivity in detecting signal [6]. The authors observed an enrichment of specific biological functions among the genes in the reported hotspots. The study also reported that enriched categories tended to match the annotation of candidate regulators. Moreover, one predicted regulator was experimentally validated. In sum, these data seem to support the hypothesis that hotspots are downstream of a common master regulator linked to the eQTL.
However, we suggest here that these observations may also be explained by clusters of genes with highly correlated expression. If one gene shows a spurious eQTL, many correlated genes will show the same spurious eQTL, in particular if the false discovery rate for individual eQTLs is very high [2], [7]–[9]. There are many nongenetic mechanisms that can create strongly correlated clusters of functionally related genes. On the one hand, such clusters may be a result of a concerted response to some uncontrolled environmental factor. On the other hand, dissected tissue samples can contain slightly varying fractions of individual cell types, leading to cell-type–specific gene clusters, which vary in a correlated manner. The resulting correlation patterns represent potentially confounding effects, both for the correct determination of a significance threshold and for the biological interpretation of the resulting hotspots.
Consequently, a key consideration in eQTL analysis is in the effective design of a permutation strategy to assess statistical significance. The approach used in [5] permuted the observed eQTLs among genes (Figure 1B). However, this approach has the disadvantage of ignoring the expression correlation between genes so that their spurious eQTLs no longer cluster along the genome. This permutation strategy leads to a potentially severe underestimate of the null distribution of the size of hotspots, when there are correlated clusters as described above.
Fig. 1. Alternative Permutation Strategies for Determining the Significance of eQTL Hotspots in Linkage and Association Studies. 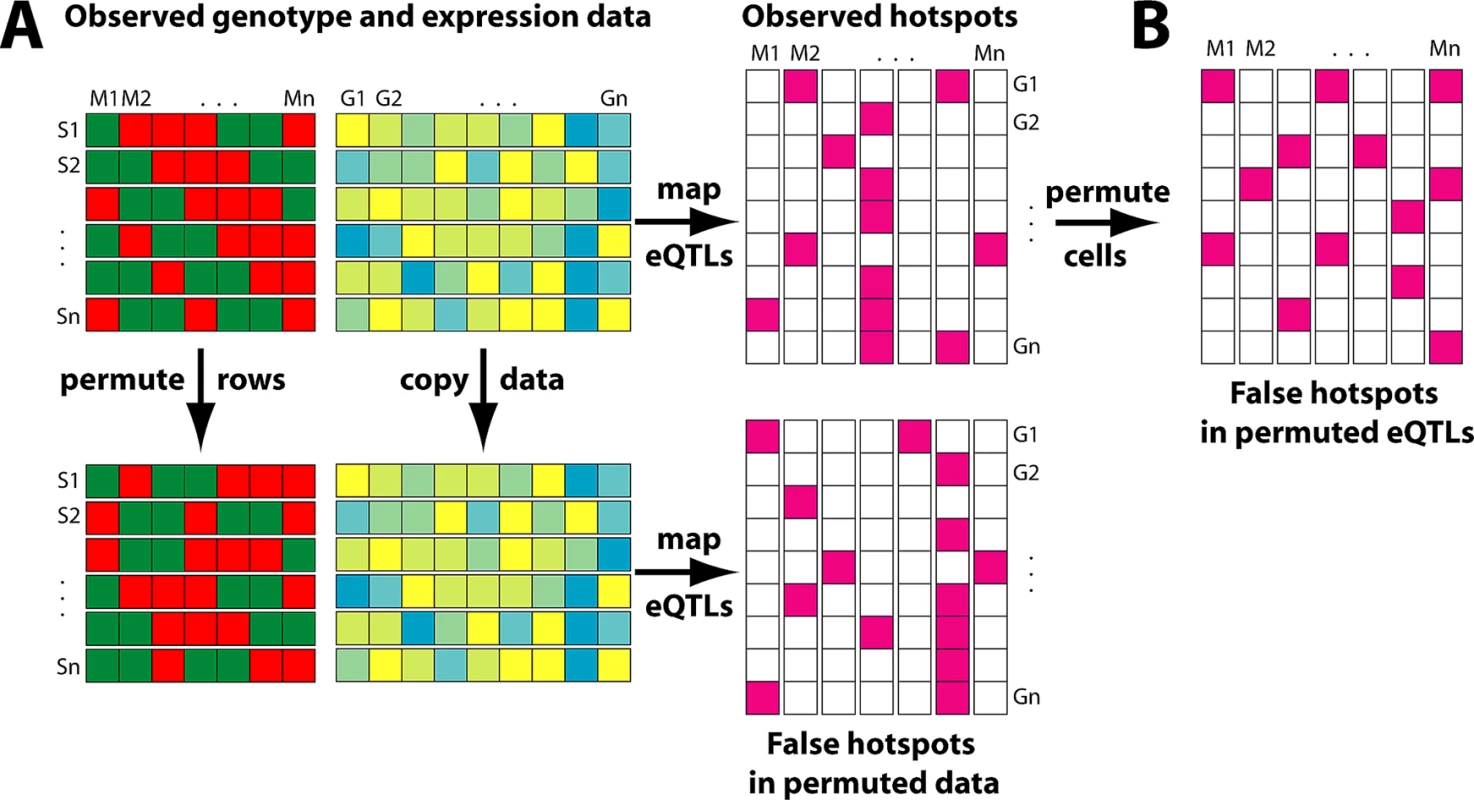
(A) The top panel shows the original data. The genotype matrix contains information about the genotype of each strain (S1…Sn) at each marker position along the genome (M1…Mn). For each strain, the expression of genes G1…Gn is measured. Linkage or association mapping combines these two sources of information to yield the eQTL matrix, where each purple entry indicates a significant linkage or association for a gene at a particular locus. The bottom panel illustrates the permutation strategy advocated here, where the strain labels are permuted, so that each strain is assigned the genotype vector of another random strain, while the expression matrix is unchanged. When the mapping is repeated on these permuted data, the correlation structure of gene expression is maintained, leading to an accurate estimate of the clustered distribution of false eQTLs along the genome. (B) shows the permutation strategy used in [5], where the original eQTL matrix is permuted by assigning the same number of eQTLs to genes randomly. The correlation of gene expression is lost, leading to an underestimate of the clustered pattern of spurious eQTLs. An alternative strategy would have been to permute the strain labels as shown in Figure 1A, maintaining the correlation of the expression traits while destroying any genetic association [2],[10]. As discussed above, it is expected that this would result in a more realistic significance threshold and a much smaller number of significant hotspots. Reanalysis of the data from [5] confirmed this idea: when permuting the strain labels (i.e., randomly swapping the genotypes between animals), the average maximum size of hotspots in the permuted data increases from less than 50 to 986. Consequently, even the largest hotspot in the real data only has a multiple testing corrected p-value of 0.23. This reanalysis demonstrates that expression correlation can indeed explain a large part of the co-mapping between genes. Such effects may also underlie some of the higher numbers of hotspots reported by some earlier studies (Table 1), especially where no appropriate permutation tests were applied to determine the statistical significance of hotspots [2].
Of course, this does not imply that all hotspots are necessarily false positives. As described above, about 5% of the co-mapping clusters in [5] are not only functionally coherent but also map to a locus that contains a gene of the same functional class. This number is not statistically significant, but it is still suggestive of an enrichment of functional associations (p<0.16, false discovery rate = 67%; C. Wu and A. I. Su, unpublished data). Some of these prioritized hotspots could correspond to true hotspots, and indeed one of them has been verified experimentally: cyclin H was validated as a new upstream regulator of cellular oxidative phosphorylation, as well as a transcriptional regulator of genes composing a hotspot [5].
Other studies, which used much stricter thresholds for defining their hotspots, also demonstrated the potential of interpreting putative hotspots by a closer study of the associated genetic locus [11],[12]. An example is the recent work of Zhu et al. [12]: by combining eQTL information, transcription factor binding sites, and protein–protein interaction data in a Bayesian network approach, they were able to predict causal regulators for nine out of the 13 hotspots (69%) originally reported in [13]. With integrated methods like these, it should be possible to identify those hotspots that are more than just clusters of co-expressed genes. As a result, the number of identified, functionally relevant hotspots could ultimately increase beyond the small numbers reported in Table 1. This would create new opportunities for gene regulatory network reconstruction.
In any case, for the time being it seems that distant eQTLs and their hotspots are still scarce and hard to find, and that those that are reported should be interpreted with caution. This rarity of convincing hotspots in genetical genomics studies is intriguing. It could be due to the limited power of the initial studies, but it could also have a more profound reason. For example, it might well be that biological systems are so robust against subtle genetic perturbations that the majority of heritable gene expression variation is effectively “buffered” and does not lead to downstream effects on other genes, protein, metabolites, or phenotypes [14]–[17]. Experimental evidence for phenotypic buffering of protein coding polymorphisms is well established [18],[19].
In fact, it has been shown that phenotypic buffering is a general property of complex gene-regulatory networks [20]. Also, if small heritable changes in transcript levels were transmitted unbuffered throughout the system, there would be a grave danger that genetic recombination would lead to unhealthy combinations of alleles and, consequently, to systems failure. Hotspots with large pleiotropic effects are thus more likely to be removed by purifying selection. If, as thus expected, common alleles are predominantly buffered by the robust properties of the system and hence largely inconsequential for the rest of the molecules in the system, this will have profound consequences for the design and interpretation of genetical genomics studies of complex diseases. Most importantly, it could turn out that even so-called common diseases—like diabetes, asthma, or rheumatoid arthritis—are not necessarily the result of common, small-effect variants in a large number of genes, but are rather caused by changes at a few crucial fragile points of the system (hotspots), which cause large, system-wide disturbances [21],[22]. Future studies in genetical genomics should aim at further elucidating the striking rarity of eQTL hotspots.
Zdroje
1. SchadtEE
MonksSA
DrakeTA
LusisAJ
CheN
2003 Genetics of gene expression surveyed in maize, mouse and man. Nature 422 297 302
2. de KoningDJ
HaleyCS
2005 Genetical genomics in humans and model organisms. Trends Genet 21 377 381
3. EmilssonV
ThorleifssonG
ZhangB
LeonardsonAS
ZinkF
2008 Genetics of gene expression and its effect on disease. Nature 452 423 428
4. KeurentjesJJ
FuJ
TerpstraIR
GarciaJM
van den AckervekenG
2007 Regulatory network construction in Arabidopsis by using genome-wide gene expression quantitative trait loci. Proc Natl Acad Sci U S A 104 1708 1713
5. WuC
DelanoDL
MitroN
SuSV
JanesJ
2008 Gene set enrichment in eQTL data identifies novel annotations and pathway regulators. PLoS Genet 4(5) e1000070 doi:10.1371/journal.pgen.1000070
6. WesselJ
ZapalaMA
SchorkNJ
2007 Accommodating pathway information in expression quantitative trait locus analysis. Genomics 90 132 142
7. PengJ
WangP
TangH
2007 Controlling for false positive findings of trans-hubs in expression quantitative trait loci mapping. BMC Proc 1Suppl 1 S157
8. Perez-EncisoM
2004 In silico study of transcriptome genetic variation in outbred populations. Genetics 166 547 554
9. WangS
ZhengT
WangY
2007 Transcription activity hot spot, is it real or an artifact? BMC Proc 1Suppl 1 S94
10. ChurchillGA
DoergeRW
2008 Naive application of permutation testing leads to inflated type I error rates. Genetics 178 609 610
11. StylianouIM
AffourtitJP
ShockleyKR
WilpanRY
AbdiFA
2008 Applying gene expression, proteomics and single-nucleotide polymorphism analysis for complex trait gene identification. Genetics 178 1795 1805
12. ZhuJ
ZhangB
SmithEN
DreesB
BremRB
2008 Integrating large-scale functional genomic data to dissect the complexity of yeast regulatory networks. Nat Genet 40 854 861
13. YvertG
BremRB
WhittleJ
AkeyJM
FossE
2003 Trans-acting regulatory variation in Saccharomyces cerevisiae and the role of transcription factors. Nat Genet 35 57 64
14. Le RouzicA
CarlborgO
2008 Evolutionary potential of hidden genetic variation. Trends Ecol Evol 23 33 37
15. GibsonG
WagnerG
2000 Canalization in evolutionary genetics: a stabilizing theory? Bioessays 22 372 380
16. GibsonG
DworkinI
2004 Uncovering cryptic genetic variation. Nat Rev Genet 5 681 690
17. CarlborgO
HaleyCS
2004 Epistasis: too often neglected in complex trait studies? Nat Rev Genet 5 618 625
18. QueitschC
SangsterTA
LindquistS
2002 Hsp90 as a capacitor of phenotypic variation. Nature 417 618 624
19. RutherfordSL
LindquistS
1998 Hsp90 as a capacitor for morphological evolution. Nature 396 336 342
20. BergmanA
SiegalML
2003 Evolutionary capacitance as a general feature of complex gene networks. Nature 424 549 552
21. IyengarSK
ElstonRC
2007 The genetic basis of complex traits: rare variants or “common gene, common disease”? Methods Mol Biol 376 71 84
22. BodmerW
BonillaC
2008 Common and rare variants in multifactorial susceptibility to common diseases. Nat Genet 40 695 701
23. BremRB
YvertG
ClintonR
KruglyakL
2002 Genetic dissection of transcriptional regulation in budding yeast. Science 296 752 755
24. KirstM
MyburgAA
De LeonJP
KirstME
ScottJ
2004 Coordinated genetic regulation of growth and lignin revealed by quantitative trait locus analysis of cDNA microarray data in an interspecific backcross of eucalyptus. Plant Physiol 135 2368 2378
25. MonksSA
LeonardsonA
ZhuH
CundiffP
PietrusiakP
2004 Genetic inheritance of gene expression in human cell lines. Am J Hum Genet 75 1094 1105
26. MorleyM
MolonyCM
WeberTM
DevlinJL
EwensKG
2004 Genetic analysis of genome-wide variation in human gene expression. Nature 430 743 747
27. CheungVG
SpielmanRS
EwensKG
WeberTM
MorleyM
2005 Mapping determinants of human gene expression by regional and genome-wide association. Nature 437 1365 1369
28. StrangerBE
ForrestMS
ClarkAG
MinichielloMJ
DeutschS
2005 Genome-wide associations of gene expression variation in humans. PLoS Genet 1 e78 doi:10.1371/journal.pgen.0010078
29. CheslerEJ
LuL
ShouS
QuY
GuJ
2005 Complex trait analysis of gene expression uncovers polygenic and pleiotropic networks that modulate nervous system function. Nat Genet 37 233 242
30. BystrykhL
WeersingE
DontjeB
SuttonS
PletcherMT
2005 Uncovering regulatory pathways that affect hematopoietic stem cell function using ‘genetical genomics’. Nat Genet 37 225 232
31. HubnerN
WallaceCA
ZimdahlH
PetrettoE
SchulzH
2005 Integrated transcriptional profiling and linkage analysis for identification of genes underlying disease. Nat Genet 37 243 253
32. MehrabianM
AllayeeH
StocktonJ
LumPY
DrakeTA
2005 Integrating genotypic and expression data in a segregating mouse population to identify 5-lipoxygenase as a susceptibility gene for obesity and bone traits. Nat Genet 37 1224 1233
33. DeCookR
LallS
NettletonD
HowellSH
2006 Genetic regulation of gene expression during shoot development in Arabidopsis. Genetics 172 1155 1164
34. LanH
ChenM
FlowersJB
YandellBS
StapletonDS
2006 Combined expression trait correlations and expression quantitative trait locus mapping. PLoS Genet 2 e6 doi:10.1371/journal.pgen.0020006
35. WangS
YehyaN
SchadtEE
WangH
DrakeTA
2006 Genetic and genomic analysis of a fat mass trait with complex inheritance reveals marked sex specificity. PLoS Genet 2 e15 doi:10.1371/journal.pgen.0020015
36. LiY
AlvarezOA
GuttelingEW
TijstermanM
FuJ
2006 Mapping determinants of gene expression plasticity by genetical genomics in C. elegans. PLoS Genet 2 e222 doi:10.1371/journal.pgen.0020222
37. McClurgP
JanesJ
WuC
DelanoDL
WalkerJR
2007 Genomewide association analysis in diverse inbred mice: power and population structure. Genetics 176 675 683
38. SchadtEE
MolonyC
ChudinE
HaoK
YangX
2008 Mapping the genetic architecture of gene expression in human liver. PLoS Biol 6 e107 doi:10.1371/journal.pbio.0060107
39. GhazalpourA
DossS
KangH
FarberC
WenPZ
2008 High-resolution mapping of gene expression using association in an outbred mouse stock. PLoS Genet 4 e1000149 doi:10.1371/journal.pgen.1000149
Štítky
Genetika Reprodukční medicína
Článek vyšel v časopisePLOS Genetics
Nejčtenější tento týden
2008 Číslo 10
Nejčtenější v tomto čísle- Mutation and Evolutionary Rates in Adélie Penguins from the Antarctic
- Leaving the Past Behind
- Genetical Genomics: Spotlight on QTL Hotspots
Kurzy
Zvyšte si kvalifikaci online z pohodlí domova
Současné možnosti léčby obezity
nový kurzAutoři: MUDr. Martin Hrubý
Všechny kurzyPřihlášení#ADS_BOTTOM_SCRIPTS#Zapomenuté hesloZadejte e-mailovou adresu, se kterou jste vytvářel(a) účet, budou Vám na ni zaslány informace k nastavení nového hesla.
- Vzdělávání



